Research Articles
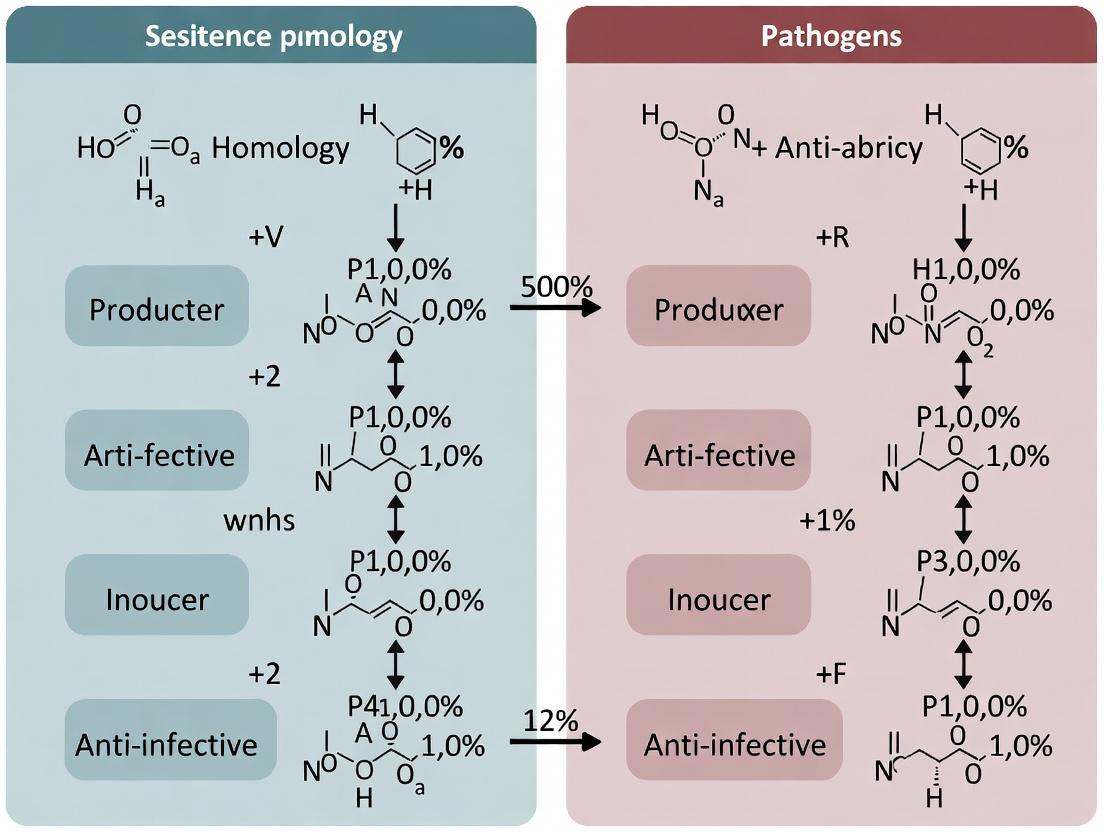
Decoding Resistance: Comparative Sequence Homology Analysis of Antimicrobial Genes in Producing Organisms vs. Pathogens
This article provides a comprehensive guide for researchers on the comparative sequence homology analysis of antimicrobial resistance (AMR) genes shared between antibiotic-producing environmental bacteria (the producers) and clinically relevant pathogens.
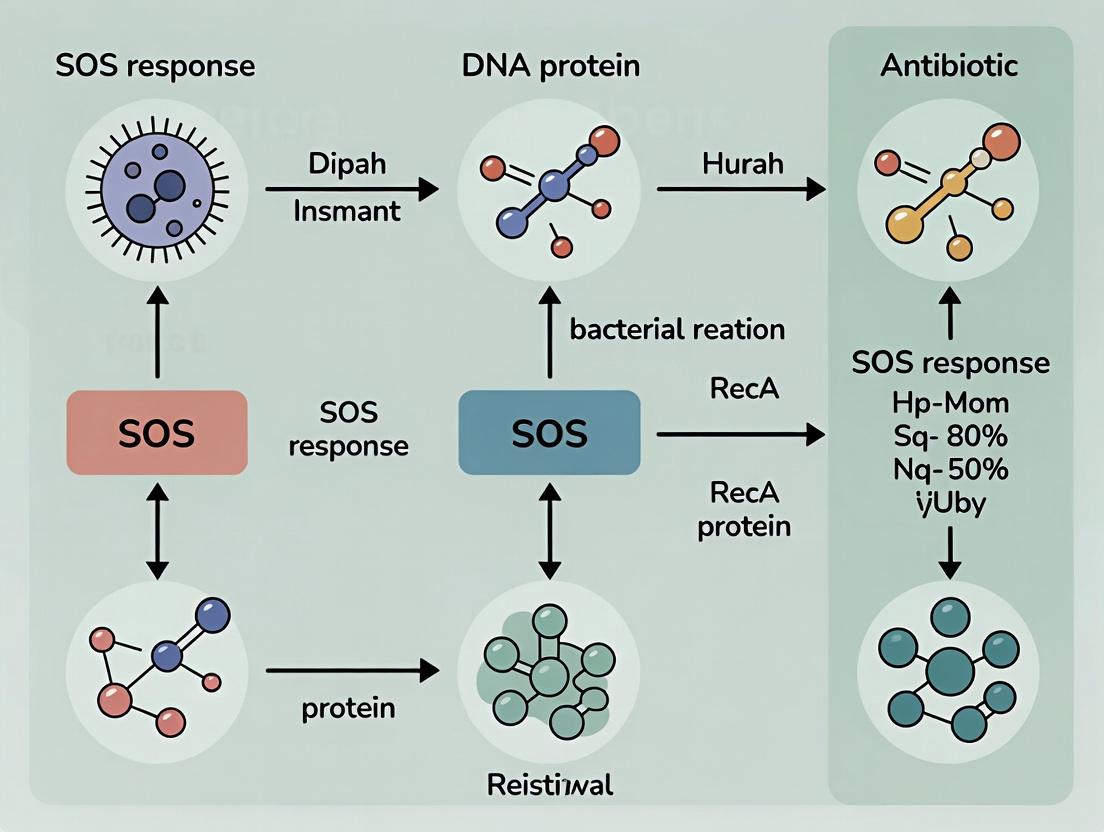
SOS Response Inhibition: A Novel Strategy to Combat Antibiotic Resistance and Potentiate Existing Drugs
This article comprehensively examines the bacterial SOS response as a high-priority target for combating antibiotic resistance.
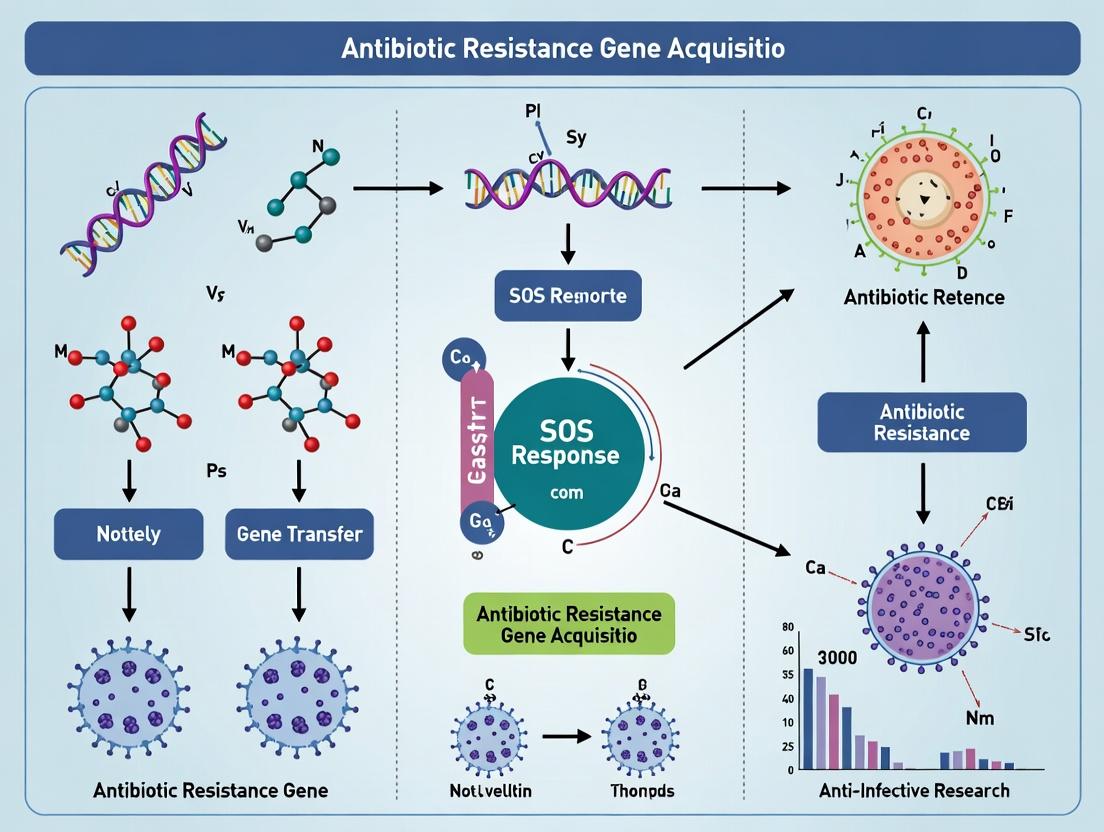
SOS Response: The Bacterial Stress Mechanism Fueling Antibiotic Resistance Gene Acquisition
This article examines the critical link between the bacterial SOS response and the accelerated acquisition of antibiotic resistance genes.
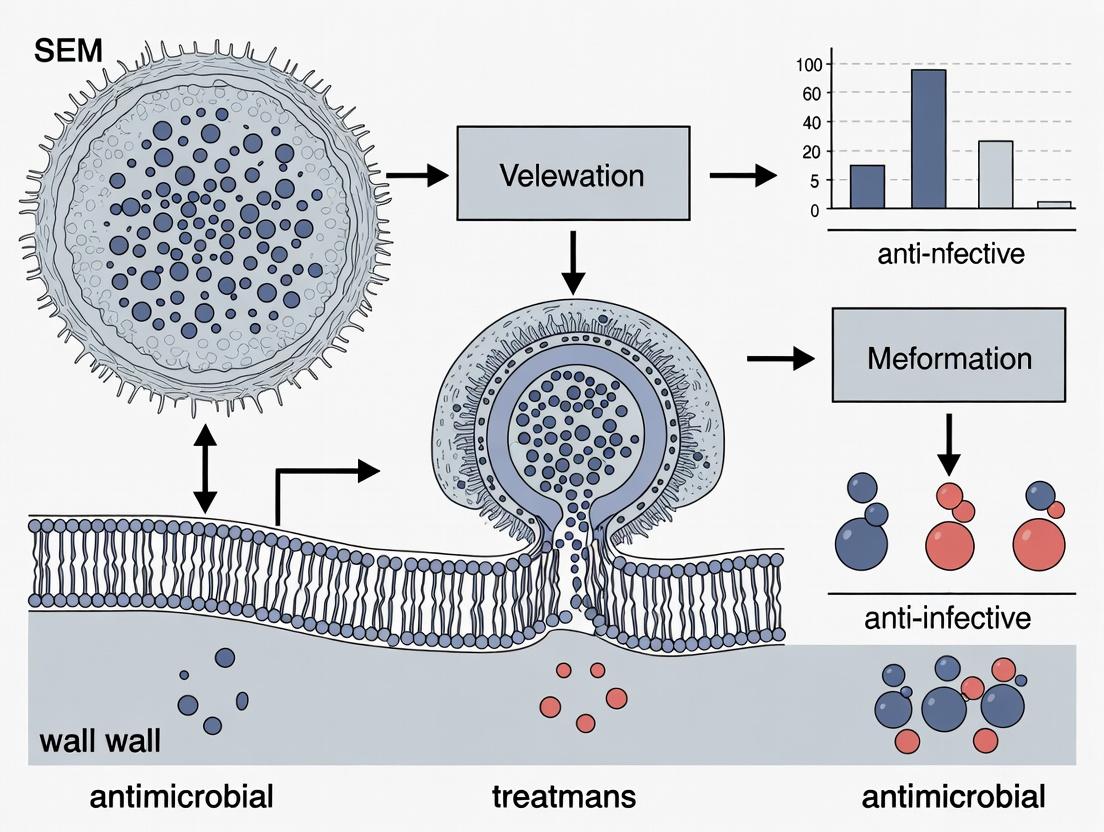
Visualizing the Kill: A Guide to SEM Analysis of Microbial Cell Deformation by Purified Antimicrobial Compounds
This comprehensive guide details the application of Scanning Electron Microscopy (SEM) for analyzing microbial cell structural damage and deformation following treatment with purified antimicrobials.
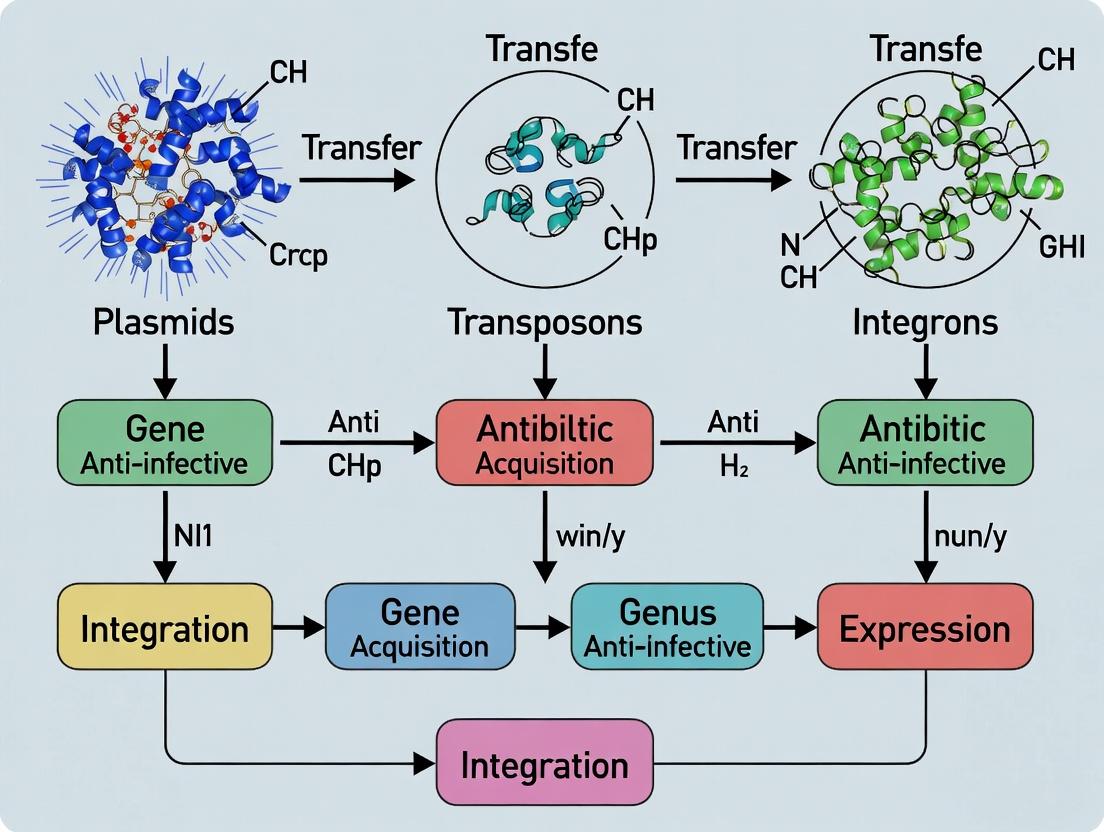
Mobile Genetic Elements as Superhighways: How Plasmids, Transposons, and Integrons Drive Antibiotic Resistance
This article provides a comprehensive overview for researchers and drug development professionals on the critical role of mobile genetic elements (MGEs) in disseminating antibiotic resistance genes (ARGs).
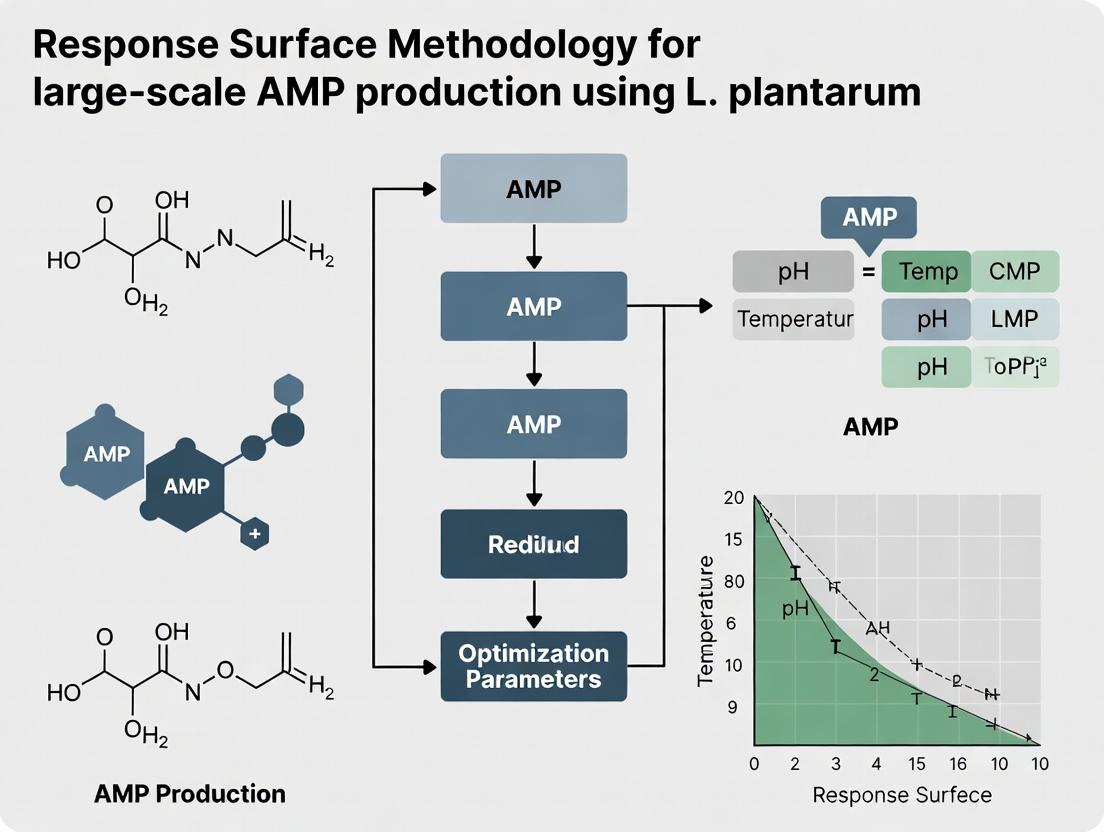
Optimizing Large-Scale AMP Production from Lactobacillus plantarum Using Response Surface Methodology: A Complete Guide
This article provides a comprehensive guide for researchers and bioprocess scientists on applying Response Surface Methodology (RSM) to optimize the large-scale production of Antimicrobial Peptides (AMPs) from Lactobacillus plantarum.
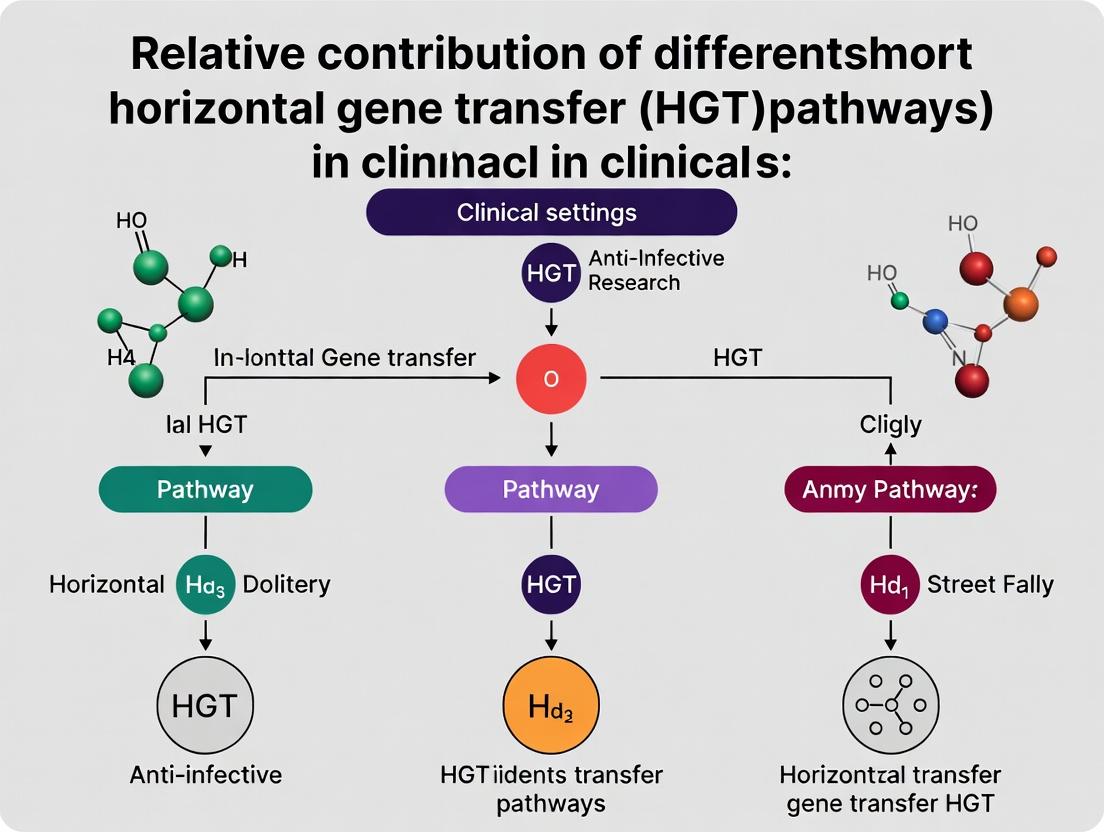
HGT Pathways in Antibiotic Resistance: Mechanisms, Clinical Impact, and Detection Strategies for Healthcare
This article provides a comprehensive analysis of the relative contributions of different Horizontal Gene Transfer (HGT) pathways—conjugation, transformation, and transduction—in clinical and hospital settings, with a focus on antimicrobial resistance...
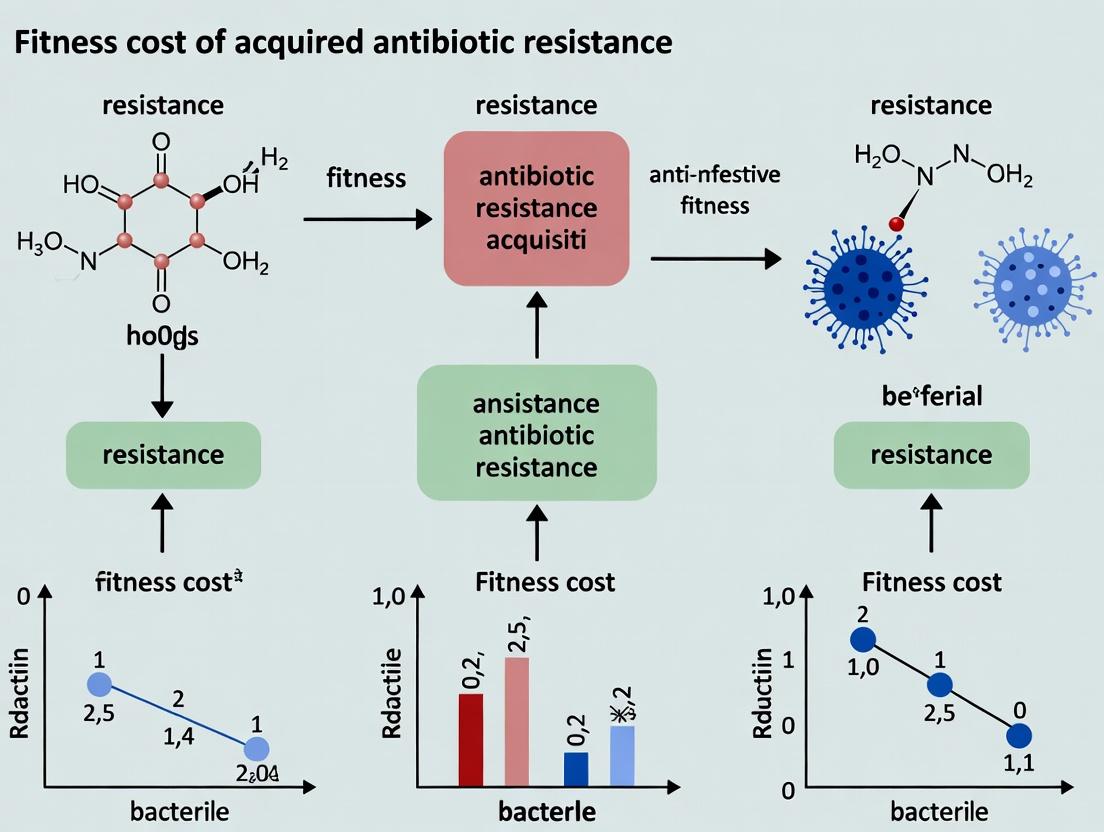
Compensatory Evolution: Strategies to Reduce Fitness Costs in Antibiotic-Resistant Pathogens
This review addresses the critical challenge of fitness costs associated with acquired antibiotic resistance, exploring how resistance mechanisms burden bacterial physiology and reduce competitiveness.
Optimizing Nature's Defense: RSM-Enhanced Bacteriocins vs. Chemical Preservatives in Biomedical Applications
This article presents a comprehensive analysis of Response Surface Methodology (RSM) for optimizing bacteriocin production and efficacy as natural antimicrobials, directly comparing their performance against conventional chemical preservatives.
Response Surface Methodology (RSM) in Antibacterial R&D: A Systematic Approach for Validating Efficacy Against Multidrug-Resistant Pathogens
This article provides a comprehensive framework for employing Response Surface Methodology (RSM) to validate the antibacterial efficacy of novel compounds and formulations against multidrug-resistant (MDR) pathogens.