Vesiduction in Gram-Negative Bacteria: A Comprehensive Guide to Monitoring Mechanisms and Methods
This article provides a detailed framework for researchers, scientists, and drug development professionals aiming to monitor vesiduction—the cargo-selective extracellular vesicle biogenesis pathway—in Gram-negative bacteria.
Vesiduction in Gram-Negative Bacteria: A Comprehensive Guide to Monitoring Mechanisms and Methods
Abstract
This article provides a detailed framework for researchers, scientists, and drug development professionals aiming to monitor vesiduction—the cargo-selective extracellular vesicle biogenesis pathway—in Gram-negative bacteria. We first explore the foundational biology of vesiduction and its role in virulence and communication. We then detail current methodological approaches, from genetic reporters to proteomic and lipidomic analyses. The guide further addresses common troubleshooting and optimization strategies for experimental challenges. Finally, we compare and validate different monitoring techniques, discussing their strengths and applications in antimicrobial and therapeutic development. This comprehensive resource synthesizes the latest research to advance the study of this critical bacterial process.
What is Vesiduction? Unveiling the Mechanism of Selective Vesicle Biogenesis in Gram-Negative Pathogens
Within the thesis context of monitoring vesiduction in Gram-negative bacteria, this document defines the cargo-selective vesiduction pathway. Vesiduction is the active, genetically regulated production of outer membrane vesicles (OMVs) for specific cargo export, distinct from stochastic blebbing due to membrane stress. This pathway is crucial in bacterial communication, virulence, and antibiotic resistance.
Application Notes:
- Drug Discovery: Targeting vesiduction machinery disrupts virulence factor and toxin delivery, offering novel anti-infective strategies.
- Vaccine Development: Engineered OMVs from cargo-selective pathways are potent, natural antigen delivery platforms.
- Diagnostics: Vesiduction-specific biomarkers in patient samples can indicate active, virulent infection stages.
- Fundamental Research: Monitoring cargo-selective vesiduction reveals novel mechanisms of intercellular signaling and metabolic regulation in microbial communities.
Table 1: Key Quantitative Markers Differentiating Cargo-Selective Vesiduction from Random Blebbing
| Parameter | Cargo-Selective Vesiduction | Random Membrane Blebbing | Measurement Method |
|---|---|---|---|
| OMV Diameter | 20 - 100 nm (narrow, uniform distribution) | 50 - 250 nm (broad, variable distribution) | Dynamic Light Scattering (DLS), TEM |
| Specific Cargo Enrichment Ratio (e.g., Toxin) | 5 - 20x (enriched vs. cytoplasm) | ~1x (reflects cytoplasmic concentration) | Immunoblot, Mass Spectrometry |
Dependence on tol-pal System |
High (disruption reduces yield >70%) | Low (disruption may increase yield) | Genetic knockout OMV quantification |
| Response to DNA-damaging Agents | Induced (2-5x increase in OMV production) | Minimally affected | OMV protein yield post-induction |
| Energy Requirement | ATP-dependent (inhibited by cyanide/azide) | Energy-independent | OMV yield with metabolic inhibitors |
Table 2: Essential Genes and Proteins in Gram-negative Bacterial Vesiduction
| Gene/Protein Complex | Function in Vesiduction Pathway | Phenotype of Deletion/Mutation |
|---|---|---|
tol-pal System |
Maintains OM-PM linkage; controlled destabilization enables vesiduction. | Hypervesiculation, but cargo is non-selective (random blebbing). |
vacJ/Yrb ABC Transporter |
Regulates phospholipid asymmetry in OM; essential for cargo loading. | Altered OMV phospholipid & protein content; reduced virulence. |
| σE Stress Response | Upregulates vesiduction-related genes in response to periplasmic stress. | Attenuated vesiduction under stress conditions. |
| Outer Membrane Proteins (e.g., OmpA, OmpC) | Curvature induction; potential cargo selection gates. | Altered OMV size and morphology. |
Detailed Experimental Protocols
Protocol 3.1: Isolation and Purification of OMVs for Cargo Analysis
Objective: To separate OMVs from culture supernatant with high purity. Reagents: See Section 4.
- Culture: Grow Gram-negative bacterium (e.g., Pseudomonas aeruginosa) to late-log phase (OD600 ~1.0) in appropriate medium.
- Harvest: Centrifuge culture at 10,000 x g for 20 min at 4°C to remove cells.
- Filtration: Filter supernatant through a 0.45 μm PVDF filter, then a 0.22 μm filter.
- Ultracentrifugation: Pellet OMVs by centrifuging filtered supernatant at 150,000 x g for 2 hours at 4°C.
- Wash: Resuspend pellet gently in sterile PBS or HEPES buffer.
- Second Ultracentrifugation: Repeat step 4. Resuspend final OMV pellet in a small volume (e.g., 100 μL) of buffer.
- Storage: Aliquot and store at -80°C. Avoid repeated freeze-thaw cycles.
Protocol 3.2: Assessing Cargo Selectivity via Comparative Proteomics
Objective: To quantify enrichment of specific proteins in OMV versus cell envelope. Reagents: See Section 4.
- Sample Preparation: Isplicate OMV (Protocol 3.1) and prepare total cell envelope fraction (cell lysis followed by differential centrifugation).
- Protein Digestion: Quantify protein content (BCA assay). Denature, reduce, alkylate, and digest equal protein amounts from each fraction with trypsin.
- Mass Spectrometry: Analyze peptides by LC-MS/MS using a high-resolution instrument (e.g., Q-Exactive).
- Data Analysis: Use label-free quantification (LFQ) software (e.g., MaxQuant). Calculate enrichment ratio: (LFQ intensity in OMV) / (LFQ intensity in cell envelope).
- Validation: Confirm hits for highly enriched proteins (>5x) via immunoblot.
Protocol 3.3: Chemical Inhibition of ATP-Dependent Vesiduction
Objective: To demonstrate energy dependence of cargo-selective pathway.
- Culture Setup: Inoculate triplicate cultures. Grow to mid-log phase (OD600 ~0.5).
- Inhibition: Add sodium azide (10 mM final concentration) or DMSO (vehicle control) to cultures.
- Incubation: Continue incubation for 60 minutes.
- OMV Isolation: Harvest OMVs from each culture using Protocol 3.1, Steps 2-7.
- Quantification: Perform BCA assay on final OMV suspensions.
- Analysis: Normalize OMV protein yield to culture OD600. Compare azide-treated yield to control.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Vesiduction Research
| Item | Function/Application | Key Consideration |
|---|---|---|
| Polycarbonate Ultracentrifuge Tubes | For high-speed pelleting of OMVs. | Compatible with >150,000 x g; non-binding surface. |
| 0.22 μm PVDF Syringe Filters | Sterile filtration of supernatant to remove residual cells. | Low protein binding minimizes cargo loss. |
| Protease Inhibitor Cocktail (e.g., EDTA-free) | Prevents degradation of OMV cargo proteins during isolation. | EDTA-free to avoid destabilizing OM integrity. |
| OptiPrep or Sucrose Density Gradient Media | For further purification of OMVs away from soluble proteins/contaminants. | Essential for downstream omics applications. |
| Anti-LPS Antibody (Core or Lipid A) | Positive control for OMV presence via immunoblot/ELISA. | Confirms successful isolation of membrane material. |
| Sodium Azide (NaN3) | Inhibitor of cytochrome oxidases; depletes ATP. | Used to test energy dependence of vesiduction. |
| Mitomycin C | DNA-damaging agent; induces the SOS response and vesiduction. | Positive control for stimulating cargo-selective pathway. |
Visualization Diagrams
Diagram 1 Title: Cargo-Selective Vesiduction vs. Random Blebbing Pathways
Diagram 2 Title: Experimental Workflow for OMV Isolation and Analysis
Diagram 3 Title: Regulatory Network Inducing Cargo-Selective Vesiduction
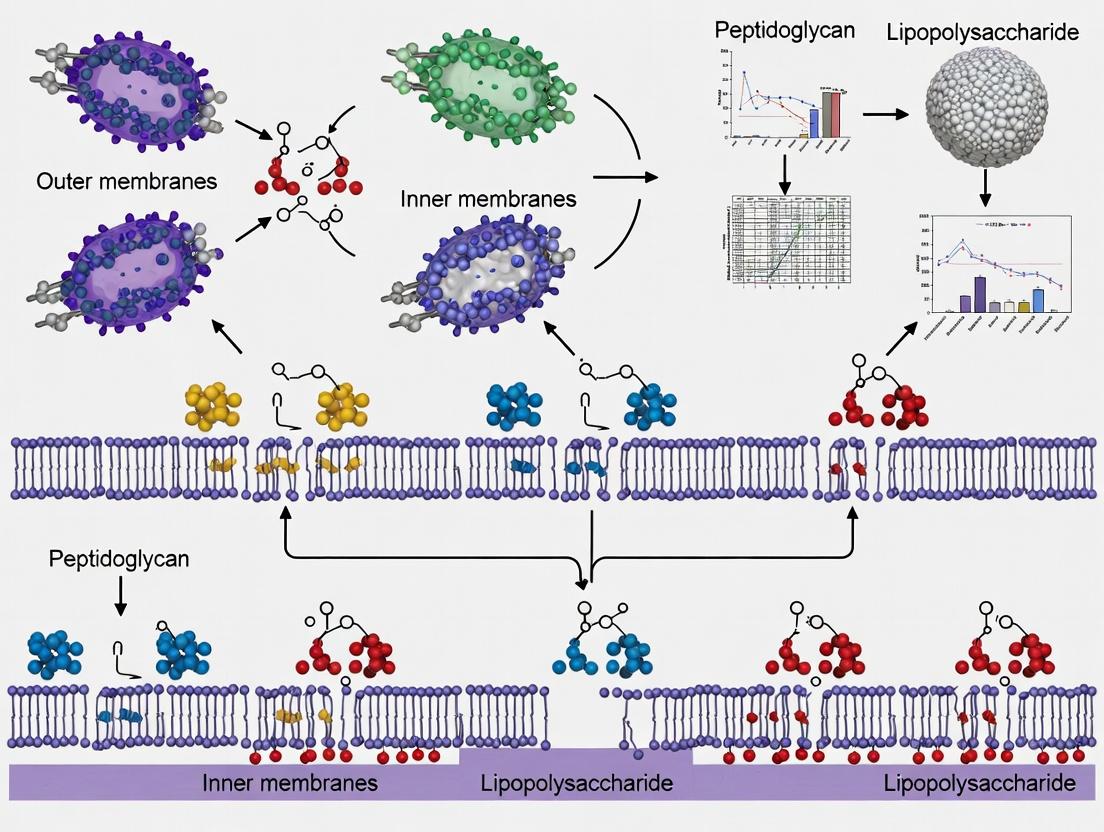
Maintaining outer membrane (OM) asymmetry is critical for Gram-negative bacterial fitness and virulence. Disruption of this asymmetry, through mislocalization of phospholipids (PLs) to the outer leaflet, triggers a stress response leading to the production of outer membrane vesicles (OMVs). This regulated process, termed "vesiduction," is a key area of study for understanding bacterial pathogenesis, intercellular communication, and potential antimicrobial targets. The VacJ/Yrb ABC transporter complex, the Mla (Maintenance of Lipid Asymmetry) system, and associated phospholipid turnover enzymes are central molecular players in monitoring and regulating OM lipid homeostasis, thereby directly influencing vesiduction.
Table 1: Key Phenotypic Outcomes of System Disruption in E. coli and Haemophilus influenzae
| Molecular System/ Gene Disrupted | Observed Phenotype (Quantitative Change) | Measured Parameter | Implication for Vesiduction |
|---|---|---|---|
| VacJ/Yrb (H. influenzae) | ↑ OM vesiculation (2-3 fold) | OMV protein yield | Loss of retrograde transport ↑ OM PL accumulation, driving vesiculation. |
| MlaFEDCB (E. coli) | ↑ OM permeability (4-8 fold) | Nile Red uptake / SDS sensitivity | Compromised OM barrier; PL accumulation in OM outer leaflet. |
| MlaA (OM lipoprotein) (E. coli) | ↑ OM PL content (~1.5-2 fold) | Phospholipid: LPS ratio (OM extracts) | Defect in retrograde PL removal; primary OM site for PL "vacuuming". |
| PldA (OM phospholipase) (E. coli) | ↓ OM vesiculation upon stress | OMV count (NTA) | Cleaves mislocalized PLs, reducing OM curvature stress and vesiculation. |
| YhjJ (Inner membrane lyso-PE acyltransferase) (E. coli) | ↑ Sensitivity to OM stress | Growth in presence of EDTA/antibiotics | Disruption of PL recycling loop impacts overall homeostasis. |
Table 2: Representative Genetic Interactions Affecting Vesiculation
| Genetic Background | Second-site Modification | Effect on Vesiculation | Interpretation |
|---|---|---|---|
| ΔmlaA | ΔpldA | Synergistic increase | MlaA removal and PldA removal independently promote PL accumulation. |
| ΔvacJ (H. influenzae) | Complemented with vacJ gene | Reversion to wild-type levels | Confirms VacJ is directly responsible for phenotype. |
| Wild-type | Overexpression of pldA | Decreased baseline OMV release | Enhanced hydrolysis of mislocalized PLs stabilizes OM. |
Experimental Protocols
Protocol 3.1: Assessing Outer Membrane Permeability via Nile Red Uptake Assay
Purpose: To quantify the loss of OM asymmetry and barrier function in Mla/VacJ mutants. Reagents: Nile Red stain (1 mg/mL in DMSO), PBS (pH 7.4), bacterial culture in mid-log phase (OD600 ~0.5). Procedure:
- Harvest 1 mL of bacterial culture by centrifugation (8,000 x g, 2 min).
- Wash cell pellet twice with 1 mL PBS.
- Resuspend pellet in 1 mL PBS containing 5 µM Nile Red (final concentration).
- Incubate in the dark at 37°C for 30 minutes.
- Wash cells twice with PBS to remove unincorporated dye.
- Resuspend final pellet in 200 µL PBS and transfer to a black-walled 96-well plate.
- Measure fluorescence (Ex/Em: 552/636 nm) using a plate reader. Normalize fluorescence values to cell density (OD600).
Protocol 3.2: Quantification of Outer Membrane Vesicle (OMV) Production
Purpose: To directly measure vesiduction changes in mutant strains. Reagents: Supernatant from bacterial culture grown to stationary phase in desired medium, DNase I (100 µg/mL), RNase A (50 µg/mL), Protease Inhibitor Cocktail, 0.45 µm filter. Procedure:
- Culture bacteria to stationary phase (OD600 ~2.0). Remove cells by centrifugation (10,000 x g, 15 min, 4°C).
- Treat supernatant with DNase I and RNase A for 30 min at 37°C to degrade nucleic acids.
- Filter supernatant through a 0.45 µm PES filter.
- Concentrate OMVs by ultracentrifugation (150,000 x g, 3 h, 4°C).
- Gently resuspend OMV pellet in sterile PBS or buffer.
- Quantify OMV yield via:
- Protein content: Bradford or BCA assay.
- Particle count/size: Nanoparticle Tracking Analysis (NTA). Dilute sample 1:1000 in filtered PBS and run on NTA system.
Protocol 3.3: Analysis of Outer Membrane Phospholipid Content
Purpose: To determine the phospholipid-to-LPS ratio in purified outer membranes. Reagents: Lysozyme, EDTA, Sucrose gradient solutions, Proteinase K, LPS extraction kit, Phospholipid assay kit. Procedure:
- Prepare spheroplasts using lysozyme/EDTA treatment.
- Lyse spheroplasts by sonication. Remove unbroken cells by low-speed centrifugation.
- Separate total membranes by ultracentrifugation (100,000 x g, 1 h).
- Fractionate inner and outer membranes on a discontinuous sucrose density gradient (e.g., 35%, 45%, 55% w/v).
- Harvest the dense OM fraction. Validate purity by SDS-PAGE (presence of OmpF/C, absence of cytoplasmic markers).
- Split OM sample for parallel analysis:
- LPS Quantification: Use a commercial endotoxin (LAL) assay or measure KDO content.
- Phospholipid Quantification: Extract lipids via Bligh-Dyer method. Measure inorganic phosphate after acid digestion.
Visualizations
Diagram 1: Mla-mediated retrograde PL transport & vesiduction trigger.
Diagram 2: Experimental workflow for linking PL transport to vesiduction.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Studying PL Transport and Vesiduction
| Reagent / Material | Function / Application | Key Consideration |
|---|---|---|
| Nile Red Dye | Fluorescent probe for hydrophobic environments; increased uptake indicates OM permeability defects. | Prepare fresh stock in DMSO; optimize concentration for each strain. |
| Protease Inhibitor Cocktail (EDTA-free) | Protects OMV and membrane protein integrity during purification. | Use EDTA-free versions to preserve OM integrity and metal-dependent enzymes. |
| Polymyxin B Agarose | Binds and removes LPS from samples, aiding in pure phospholipid analysis. | Critical for accurate lipidomics; removes major contaminant. |
| Anti-MlaA or Anti-VacJ Antibodies | Immunodetection for monitoring protein localization and expression levels. | Validate cross-reactivity for species of interest. |
| C11-BODIPY 581/591 FL | Oxidation-sensitive fluorescent fatty acid analog; reports on PL transport to OM. | Used in kinetic assays to track asymmetric insertion and flipping. |
| Sucrose (Ultra Pure) | For constructing density gradients for inner/outer membrane fractionation. | Essential for obtaining pure OM samples for lipid ratio analysis. |
| Nanoparticle Tracking Analyzer (NTA) | Instrument for sizing and counting purified OMVs in suspension. | Requires careful sample dilution and calibration for reproducibility. |
| Phospholipase A1/A2 (e.g., from Thermus thermophilus) | Control enzyme to hydrolyze outer leaflet PLs, mimicking PldA activity. | Useful as an in vitro tool to manipulate OM asymmetry. |
1. Introduction & Context This Application Note is framed within a doctoral thesis investigating the monitoring of vesiduction—the production and function of bacterial extracellular vesicles (BEVs)—in Gram-negative bacteria. This document details protocols and analyses for elucidating how BEVs contribute to virulence factor delivery, horizontal gene transfer (HGT), and immune modulation, providing a toolkit for researchers in pathogenesis and therapeutic development.
2. Key Quantitative Data Summary
Table 1: BEV-Associated Virulence Factors in Select Gram-negative Pathogens
| Pathogen | BEV Cargo Identified | Demonstrated Effect on Host Cells | Citation (Example) |
|---|---|---|---|
| Pseudomonas aeruginosa | Alkaline phosphatase, hemolytic phospholipase C, Cif toxin | Epithelial cell cytotoxicity, immunosuppression | (Bomberger et al.) |
| Escherichia coli (UPEC) | Cytolysin A (ClyA), heat-labile enterotoxin | Pro-inflammatory cytokine release, pore formation | (Bielaszewska et al.) |
| Salmonella Typhi | Typhoid toxin components (PltA, CdtB) | DNA damage in target cells, systemic intoxication | (Hoshino et al.) |
Table 2: Quantification of BEV-Mediated Horizontal Gene Transfer Efficiency
| Donor Strain (Plasmid) | Recipient Strain | Transfer Method | BEV-Mediated Transfer Frequency (CFU/µg DNA) | Free DNA Transfer Frequency | Fold Increase |
|---|---|---|---|---|---|
| E. coli (pUC19 - AmpR) | E. coli Δcompetence | BEV incubation | (3.2 ± 0.4) x 10² | < 10⁰ | >300 |
| Acinetobacter baumannii (pAB5) | A. baumannii | BEV incubation | (1.5 ± 0.2) x 10³ | (5.0 ± 1.0) x 10¹ | 30 |
3. Experimental Protocols
Protocol 3.1: Isolation of BEVs from Gram-negative Bacterial Culture Supernatant (Density Gradient Ultracentrifugation)
- Materials: Late-log phase bacterial culture, 0.22 µm filter, ultracentrifuge with fixed-angle and swinging-bucket rotors, OptiPrep density gradient medium, PBS, sterile syringes and needles.
- Procedure:
- Culture bacteria to late-log phase (OD₆₀₀ ~1.0). Pellet cells at 10,000 x g for 20 min at 4°C.
- Filter supernatant through a 0.22 µm PES membrane.
- Concentrate filtrate 100-fold using centrifugal filters (100 kDa MWCO) or by ultracentrifugation at 150,000 x g for 2h at 4°C.
- Resuspend crude vesicle pellet in 1 mL PBS.
- Prepare a discontinuous OptiPrep gradient (e.g., 40%, 20%, 10% in PBS) in an ultracentrifuge tube. Layer the resuspended BEV sample on top.
- Centrifuge at 200,000 x g for 18h at 4°C in a swinging-bucket rotor.
- Fractionate the gradient from the top. Analyze fractions for protein content (Bradford) and density. Pool BEV-rich fractions (typically at ~1.15 g/cm³).
- Dilute pooled fractions in PBS and pellet BEVs at 150,000 x g for 2h. Resuspend in PBS, aliquot, and store at -80°C.
- Validation: Nanoparticle Tracking Analysis (NTA) for size/concentration, TEM for morphology, immunoblot for vesicle markers (e.g., OmpA).
Protocol 3.2: Assessing BEV-Mediated Host Cell Modulation (Cytokine Profiling)
- Materials: Cultured mammalian immune cells (e.g., THP-1 macrophages), purified BEVs, LPS (positive control), cell culture reagents, cytokine ELISA or multiplex assay kits.
- Procedure:
- Differentiate THP-1 cells into macrophages using PMA.
- Seed cells in 24-well plates at 2.5 x 10⁵ cells/well. Stimulate with purified BEVs (e.g., 1-10 µg protein/mL), PBS (negative control), or LPS (positive control) for 6-24h.
- Collect cell-free supernatants by centrifugation.
- Quantify cytokine levels (e.g., IL-1β, IL-6, IL-8, TNF-α) using commercial ELISA kits per manufacturer's instructions.
- Normalize data to cell viability (assessed via MTT or ATP-based assay). Perform statistical analysis comparing BEV-treated to control groups.
Protocol 3.3: Measuring BEV-Facilitated Horizontal Gene Transfer
- Materials: Donor strain harboring selectable plasmid, antibiotic-free recipient strain, BEVs purified from donor, appropriate agar plates with/without antibiotics, DNase I.
- Procedure:
- Purify BEVs from donor strain culture following Protocol 3.1. Treat half of the BEV sample with DNase I (1 U/µg DNA, 37°C, 30 min) to degrade external DNA.
- Grow recipient strain to mid-log phase. Wash cells 2x with PBS.
- Co-incubate recipient cells (10⁸ CFU) with BEVs (5-20 µg protein) in a 1 mL mixture for 2h at 37°C. Include controls: recipient cells alone, recipient + free plasmid, recipient + DNase-treated BEVs.
- Wash cells to remove BEVs/antibiotics. Serially dilute and plate on selective (recipient + BEV plasmid resistance) and non-selective media to determine total CFU.
- Calculate transfer frequency as (CFU on selective media) / (Total CFU on non-selective media).
4. Diagrams
BEV-Mediated Pathogenesis
BEV Isolation & Characterization Workflow
5. The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions for Vesiduction Research
| Item | Function & Application | Example/Notes |
|---|---|---|
| OptiPrep (Iodixanol) | Inert, iso-osmotic density gradient medium for high-purity BEV isolation. | Critical for separating BEVs from protein aggregates and non-vesicular contaminants. |
| Protease & DNase/RNase Inhibitors | Cocktails added during BEV purification to preserve and define native cargo. | Determines if an associated factor is intra-vesicular or externally attached. |
| Nanoparticle Tracking Analyzer (NTA) | Instrument for real-time analysis of BEV particle size distribution and concentration. | Provides essential quantitative physical data (e.g., particles/mL, mean diameter). |
| Lipopolysaccharide (LPS) ELISA | Quantifies endotoxin levels in BEV preparations. | Distinguishes BEV effects from soluble LPS contamination. |
| Cytochalasin D / Dynasore | Pharmacological inhibitors of host cell actin-dependent endocytosis and dynamin. | Used to interrogate BEV entry mechanisms into eukaryotic cells. |
| Anti-OmpA / Anti-BamA Antibodies | Western blot markers for outer membrane vesicles (OMVs) of Gram-negative bacteria. | Confirms vesicular origin and purity of preparations. |
| SYBR Gold Nucleic Acid Stain | Fluorescent stain for visualizing DNA/RNA cargo within purified BEVs via microscopy. | Essential for HGT and cargo localization studies. |
Within the context of monitoring vesiduction in Gram-negative bacteria research, distinguishing between Outer Membrane Vesicle (OMV) biogenesis pathways is critical. This application note differentiates the selective process of "vesiduction" from constitutive OMV release. Vesiduction refers to the stress-induced, cargo-specific formation of OMVs, often involving the recruitment of specific proteins and lipids. In contrast, constitutive biogenesis is a baseline, continuous process driven by inherent membrane instability. Understanding these pathways is essential for research in bacterial communication, pathogenesis, and the development of novel antimicrobials or vaccine platforms.
Key Comparative Data
Table 1: Distinguishing Features of Vesiduction vs. Constitutive OMV Biogenesis
| Feature | Vesiduction (Selective) | Constitutive OMV Biogenesis |
|---|---|---|
| Primary Trigger | Specific environmental stresses (e.g., antibiotics, DNA damage, nutrient starvation). | Baseline membrane dynamics and perturbations (e.g., lipid accumulation, peptidoglycan defects). |
| Regulation | Tightly regulated, often involving specific stress response pathways (e.g., SOS response, σE). | Largely unregulated, stochastic process. |
| Cargo Selection | Highly selective; specific enrichment of DNA, toxins, signaling molecules, or misfolded proteins. | Relatively non-selective; reflects outer membrane and periplasmic composition. |
| Key Molecular Actors | Stress response regulators (e.g., RecA, σE), cross-linking enzymes (e.g., DsbA), ToIC. | LPS structure, phospholipid accumulation, VacJ/Yrb system, outer membrane protein complexes. |
| Vesicle Size/Quantity | Variable size; often produces a distinct sub-population in response to the trigger. | More uniform size distribution; constant low-level production. |
| Biological Role | Stress response, targeted toxin delivery, horizontal gene transfer, immune evasion. | Waste removal, membrane homeostasis, decoy production. |
Table 2: Quantitative Metrics for Monitoring Vesiduction vs. Constitutive Release
| Metric | Typical Constitutive Levels | Indicative Vesiduction Response | Common Measurement Technique |
|---|---|---|---|
| OMV Yield (μg protein/mL culture) | 10-50 μg/mL | 2-5 fold increase (e.g., 100-250 μg/mL) | BCA/Lowry assay on purified OMVs |
| Specific Cargo Enrichment (Fold-Change) | 1x (Baseline) | >5x enrichment of specific cargo (e.g., DNA, toxin) | qPCR, Immunoblot, Proteomics |
| Expression of Stress Regulators | Basal level | Upregulation (e.g., >10x increase in recA transcription) | RT-qPCR, Reporter fusions |
| Vesicle Sub-population Size (nm) | 20-100 nm (median ~80 nm) | Emergence of a distinct population (e.g., 50-150 nm) | Nanoparticle Tracking Analysis (NTA) |
Detailed Experimental Protocols
Protocol 1: Induction and Quantification of Vesiduction Aim: To trigger and measure selective vesiduction in E. coli using ciprofloxacin-induced DNA damage. Materials: Wild-type E. coli, ciprofloxacin stock, appropriate growth medium, sterile filters (0.22 μm), ultracentrifuge. Procedure:
- Grow E. coli to mid-log phase (OD600 ~0.6) in liquid culture.
- Split culture into two flasks: Treatment and Control.
- Add ciprofloxacin to Treatment flask at sub-lethal concentration (e.g., 0.1 μg/mL). Add equal volume solvent to Control.
- Incubate for 2 hours with shaking.
- Collect cells by centrifugation at 10,000 x g for 15 min at 4°C. Retain supernatants.
- Filter supernatants through 0.22 μm filters to remove residual cells.
- Ultracentrifuge filtered supernatants at 150,000 x g for 2 hours at 4°C to pellet OMVs.
- Resuspend OMV pellets in sterile PBS or Tris buffer.
- Quantify total OMV protein yield via BCA assay (Table 2).
- Analyze specific cargo (e.g., genomic DNA via qPCR for a conserved gene, toxin via immunoblot) to determine enrichment factors.
Protocol 2: Differential OMV Sub-population Analysis via NTA Aim: To identify and characterize vesiduction-specific vesicle sub-populations. Materials: Purified OMV samples from Protocol 1, Nanoparticle Tracking Analyzer (e.g., Malvern NanoSight), sterile syringes. Procedure:
- Dilute purified OMV samples in filtered PBS to achieve 20-100 particles per frame for optimal NTA analysis.
- Load sample into the NTA sample chamber using a sterile syringe.
- Capture three 60-second videos for each sample (Control and Treatment) under constant flow conditions.
- Use NTA software to analyze particle size and concentration for each video.
- Compare the size distribution profiles. A vesiduction response is indicated by a significant shift in median size or the emergence of a secondary peak in the 50-150 nm range (Table 2).
- Export concentration data to calculate fold-increase in total particle release.
Signaling & Workflow Visualizations
Diagram 1: Vesiduction via SOS Response Pathway (79 chars)
Diagram 2: OMV Isolation & Analysis Workflow (62 chars)
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function & Application |
|---|---|
| Sub-lethal Antibiotics (e.g., Ciprofloxacin, Mitomycin C) | Induce specific stress responses (SOS) to trigger vesiduction for study. |
| Protease Inhibitor Cocktails | Preserve protein cargo during OMV purification and storage. |
| DNase I / RNase A | Treat OMVs to distinguish surface-associated from packaged nucleic acid cargo. |
| Anti-LPS Antibodies | Standard markers for normalizing OMV yields or for capture assays. |
| Lipid-specific Dyes (e.g., FM4-64) | Fluorescently label OMVs for imaging or flow cytometry analysis. |
| Protein Cross-linkers (e.g., DSP, Formaldehyde) | Stabilize transient protein complexes involved in cargo selection prior to lysis. |
| NTA Calibration Beads | Standardize Nanoparticle Tracking Analysis for accurate size/concentration data. |
| Density Gradient Media (e.g., OptiPrep) | Isolate specific OMV sub-populations via buoyant density centrifugation. |
The study of vesiduction—the production and function of outer membrane vesicles (OMVs) in Gram-negative bacteria—is crucial for understanding bacterial communication, pathogenesis, and host interaction. Escherichia coli, Salmonella enterica, Pseudomonas aeruginosa, and Vibrio cholerae serve as premier model organisms due to their genetic tractability, well-characterized biology, and relevance to human disease. Their distinct vesiduction profiles and regulatory networks provide comparative insights into this conserved yet heterogeneous process, offering targets for therapeutic intervention and diagnostic tool development.
Comparative Analysis of Vesiduction Characteristics
Table 1: Quantitative Vesiduction Profile of Model Organisms
| Organism | Average OMV Diameter (nm) | Typical Yield (μg protein / mL culture, OD600=1.0) | Key Enriched Cargo | Primary Induction Signal(s) |
|---|---|---|---|---|
| E. coli (K-12) | 20-100 | 10-25 | OmpA, OmpC, OmpF, SurA | SOS response, envelope stress (σᴱ, CpxR) |
| S. enterica (Typhimurium) | 50-150 | 30-60 | SPI-1/2 effectors, LPS, flagellin | PhoP/Q, low Mg²⁺, host cell contact |
| P. aeruginosa (PAO1) | 50-200 | 50-100 | Alkaline phosphatase, hemolysin, Cif | Quorum sensing (LasR, RhIR), aminoglycosides |
| V. cholerae (O1 El Tor) | 40-120 | 20-50 | CTXϕ phage, cholera toxin, hemagglutinin | Quorum sensing (HapR), bile salts |
Table 2: Key Regulatory Genes Influencing Vesiduction
| Organism | Positive Regulators | Negative Regulators | Stress Pathways Linked to Vesiduction |
|---|---|---|---|
| E. coli | degP, baeR, rob | nlpI, ppGpp | σᴱ, CpxAR, Rcs |
| Salmonella | phoP, ssrB, tolR | yfgL, ompA | PhoP/Q, PmrA/B |
| Pseudomonas | lasR, rhIR, mexI | mvfr, psrA | Las/Rhl QS, AlgU |
| Vibrio | vrrA, toxR, hapR | vpsR, hns | Quorum Sensing, ToxR/S, VarS/A |
Protocols for OMV Isolation and Characterization
Protocol 3.1: Standardized OMV Purification (Ultracentrifugation Method)
Principle: Sequential centrifugation steps to remove cells and debris, followed by ultracentrifugation to pellet OMVs. Materials: Bacterial culture, centrifuge, ultracentrifuge, fixed-angle rotor, 0.22 μm filter, PBS buffer. Procedure:
- Grow bacterial culture to desired phase (e.g., late-log for P. aeruginosa, stationary for V. cholerae).
- Centrifuge culture at 4°C, 10,000 × g for 20 min to pellet cells.
- Filter supernatant through 0.22 μm pore-size membrane.
- Ultracentrifuge filtered supernatant at 4°C, 150,000 × g for 2-3 h.
- Discard supernatant, gently resuspend OMV pellet in PBS or suitable buffer.
- Optional: Purify further via density gradient centrifugation (e.g., OptiPrep). Note: For Salmonella, include protease inhibitors to prevent degradation of effector proteins.
Protocol 3.2: Quantification of OMV Yield and Size Distribution
Principle: Use BCA for protein content and Nanoparticle Tracking Analysis (NTA) for size/concentration. Materials: BCA Protein Assay Kit, Nanosight NS300, PBS. Procedure:
- Protein Yield: Perform BCA assay on purified OMV suspension per manufacturer's instructions. Relate to original culture OD600.
- Size Distribution: Dilute OMVs in filtered PBS to ~10⁸ particles/mL. Inject into NTA chamber. Record 60-second videos in triplicate. Analyze using NTA software to determine mean/median diameter and particle concentration.
Experimental Protocol: Monitoring Vesiduction via Fluorescent Reporter
Title: Protocol for Real-Time Monitoring of Vesiduction Using a degP-GFP Reporter Fusion. Background: The degP gene, encoding a periplasmic protease, is upregulated by σᴱ envelope stress and correlates with increased vesiduction in E. coli and Salmonella. Objective: Quantify vesiduction induction in live cells under stress conditions. Reagent Solutions:
- LB Broth: Standard growth medium.
- 1M IPTG: Inducer for plasmid-based reporters.
- Stressors: 2 μg/mL Cephalexin (SOS inducer), 0.5 mM EDTA (envelope disruptor).
- PBS Buffer (pH 7.4): For washing and dilution.
- 96-well Black Wall/Clear Bottom Plate: For fluorescence readings.
- Microplate Reader: Capable of fluorescence (Ex/Em 488/510 nm) and OD600 measurement.
Procedure:
- Strain Preparation: Transform E. coli MG1655 with plasmid pDegP-GFP (PdegP fused to gfp).
- Culture Growth: Inoculate 5 mL LB with antibiotic. Grow overnight at 37°C, 200 rpm.
- Induction & Stress: Dilute culture 1:100 in fresh LB + antibiotic. Grow to OD600 ~0.3. Split culture. Treat one with stressor (e.g., Cephalexin), keep one as control.
- Real-Time Monitoring: Transfer 200 μL aliquots to 96-well plate. Place in pre-warmed (37°C) microplate reader.
- Data Acquisition: Cycle every 10 min for 6-8 h: orbital shaking for 5 sec, OD600 measurement, fluorescence measurement (Ex/Em 488/510 nm, gain optimized).
- Data Analysis: Normalize fluorescence of each well to its OD600 (RFU/OD). Plot normalized fluorescence vs. time. Correlate fluorescence peaks with OMV yield measured in parallel cultures.
Diagram: Key Vesiduction Signaling Pathways in Model Organisms
Diagram Title: Vesiduction Signaling Pathways in Key Gram-Negative Models
Diagram: OMV Isolation & Characterization Workflow
Diagram Title: Workflow for OMV Isolation and Characterization
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Vesiduction Research
| Item | Function in Vesiduction Research | Example/Supplier Notes |
|---|---|---|
| OptiPrep Density Gradient Medium | Separates OMVs from protein aggregates and other contaminants in a density gradient ultracentrifugation step. | Useful for high-purity OMV preps for proteomics. |
| BCA Protein Assay Kit | Colorimetric quantification of total protein in OMV preparations; correlates with yield. | More compatible with detergents than Bradford assay. |
| Nanosight NS300 / Particle Metrix ZetaView | Nanoparticle Tracking Analysis (NTA) instruments for real-time size distribution and concentration of OMVs. | Critical for standardized vesicle quantification. |
| Polymyxin B Affinity Resin | Binds to LPS; used to deplete or capture OMVs for functional studies or to confirm OMV identity. | Confirms vesicle origin from outer membrane. |
| Ciprofloxacin / Cephalexin | Antibiotic stressors that induce the SOS response and/or envelope stress, increasing vesiduction. | Used as positive controls in induction experiments. |
| AHL Quorum Sensing Molecules (e.g., 3O-C12-HSL) | Synthetic autoinducers used to directly stimulate vesiduction pathways in Pseudomonas and Vibrio. | For dissecting QS-specific effects. |
| Anti-OmpA / Anti-LPS Antibodies | Western blot validation of OMV markers from E. coli and Salmonella. | Confirms presence of outer membrane components. |
| Protease Inhibitor Cocktail (EDTA-free) | Added during OMV purification to prevent cargo degradation, especially critical for Salmonella. | Maintains integrity of protein cargo. |
How to Monitor Vesiduction: Essential Techniques from Genetics to Advanced Imaging
Application Notes
Genetic reporter systems, specifically fluorescent and luminescent fusion proteins, are indispensable tools for visualizing and quantifying dynamic cellular processes in real-time. Within the context of a broader thesis on monitoring vesiduction (vesicle production) in Gram-negative bacteria, these constructs enable the direct observation of protein localization, secretion, and membrane remodeling events. The fusion of reporters like Green Fluorescent Protein (GFP) to bacterial outer membrane lipoproteins, such as VacJ, allows for the precise tracking of protein function within the vesiculation pathway. This facilitates the dissection of molecular mechanisms, identification of regulatory checkpoints, and screening for compounds that modulate vesicle biogenesis—a process linked to virulence, biofilm formation, and intercellular communication.
Table 1: Common Genetic Reporters for Bacterial Vesiduction Studies
| Reporter Protein | Excitation/Emission (nm) or λmax | Brightness (Relative to GFP) | Maturation Half-time (min) | Primary Application in Vesiduction |
|---|---|---|---|---|
| GFP (avGFP) | 488/509 | 1.0 | ~40 | General protein fusion, localization |
| mCherry | 587/610 | 0.47 | ~40 | Dual-color imaging, tracking |
| NanoLuc | N/A (bioluminescent) | ~100x Renilla luciferase | <5 min | High-sensitivity quantification |
| sfGFP (superfolder GFP) | 485/510 | 1.2 | ~10 | Fusions to difficult-to-fold proteins |
| GFP-VacJ Fusion | 488/509 | N/A | Dependent on VacJ folding | Specific tracking of outer membrane asymmetry |
Table 2: Measured Impact of VacJ Dysfunction on Vesicle Production
| Bacterial Strain | Vesicle Yield (μg protein/mL/OD unit) | Mean Vesicle Diameter (nm) | Notes |
|---|---|---|---|
| Wild-Type (e.g., A. baumannii) | 5.2 ± 0.8 | 65 ± 15 | Baseline vesiduction |
| ΔvacJ mutant | 12.5 ± 1.5 | 85 ± 20 | ~2.4x increase in yield |
| Complementation (GFP-VacJ) | 6.0 ± 1.0 | 70 ± 18 | Partial functional restoration |
| Drug-Treated (Inhibitor X) | 3.1 ± 0.5 | 60 ± 10 | Suppression of vesiculation |
Experimental Protocols
Protocol 1: Construction of a GFP-VacJ Fusion Plasmid for Gram-Negative Bacteria
Objective: To generate a functional, in-frame fusion of superfolder GFP to the N-terminus of the VacJ lipoprotein.
Materials:
- Research Reagent Solutions Toolkit:
- sfGFP Amplification Primers: Forward primer with linker (e.g., GGSGGS) and restriction site (e.g., BamHI); Reverse primer with no stop codon.
- VacJ Gene Template: Genomic DNA from target Gram-negative bacterium (e.g., Acinetobacter baumannii).
- Expression Vector: Medium-copy plasmid with inducible promoter (e.g., pBAD/ara) and antibiotic resistance (e.g., ampicillin).
- Restriction Enzymes & T4 DNA Ligase: For cloning (e.g., BamHI, XhoI).
- Gibson Assembly Master Mix: For seamless, restriction-site independent cloning.
- Electrocompetent E. coli DH5α: For plasmid propagation.
- Chemically Competent Target Bacterium: For final transformation.
- LB Agar/Broth with Selective Antibiotic: For selection of transformants.
- Arabinose (20% w/v stock): For inducible expression of the fusion construct.
- PCR Purification & Gel Extraction Kits: For DNA clean-up.
Methodology:
- Amplify DNA Fragments: Perform PCR to amplify the sfgfp gene (with forward primer containing flexible linker sequence) and the vacJ gene (without its native signal peptide if desired, and without stop codon).
- Purify and Assemble: Gel-purify both PCR products. Use Gibson Assembly to insert the fused sfgfp-linker-vacJ fragment into the linearized expression vector backbone.
- Transform and Screen: Transform the assembly reaction into E. coli DH5α, plate on selective LB agar. Screen colonies by colony PCR and verify the plasmid sequence by Sanger sequencing.
- Transform Target Bacterium: Isolate the verified plasmid and transform into the chemically competent target Gram-negative strain (e.g., A. baumannii).
- Induce and Validate: Grow a positive clone to mid-log phase, induce with 0.2% arabinose for 2 hours. Validate expression and localization via SDS-PAGE (check for ~50 kDa fusion protein) and fluorescence microscopy.
Protocol 2: Live-Cell Imaging and Quantification of GFP-VacJ Localization
Objective: To visualize the subcellular localization of GFP-VacJ and correlate it with sites of vesicle budding.
Materials:
- Bacterial strain expressing GFP-VacJ.
- Microscopy-Grade Agarose: For preparing live-cell imaging pads.
- Imaging Medium: Minimal medium or PBS.
- Confocal or Epifluorescence Microscope with a 100x oil-immersion objective and appropriate GFP filter set.
- Membrane Stain (e.g., FM4-64 or FM5-95): To label the cytoplasmic membrane.
- Image Analysis Software (e.g., Fiji/ImageJ): For quantification.
Methodology:
- Sample Preparation: Grow the GFP-VacJ strain to an OD600 of ~0.5 under inducing conditions. Concentrate cells gently by centrifugation.
- Slide Preparation: Create an agarose pad (1-2%) on a microscope slide. Apply 2-5 μL of cell concentrate to the pad and cover with a coverslip.
- Image Acquisition: Using a fluorescence microscope, capture both phase-contrast and GFP-channel images. For dual-color, stain with FM4-64 (2 μg/mL) for 5 mins, wash, and image using separate channels.
- Colocalization Analysis: Use software to measure fluorescence intensity profiles along the cell membrane. Calculate Pearson's correlation coefficient between GFP-VacJ and membrane stain signals.
- Vesicle Association: Look for punctate GFP-VacJ foci at the cell surface. Correlate these foci with budding vesicles visible in phase-contrast or by supplementing the medium with a non-membrane-permeant DNA stain (e.g., propidium iodide) to label extruded DNA in outer membrane vesicles.
Protocol 3: Quantifying Vesiduction Using a Luminescent Reporter (NanoLuc-VacJ)
Objective: To quantify changes in VacJ localization or abundance as a proxy for vesicle production using a highly sensitive luminescent reporter.
Materials:
- Plasmid encoding a NanoLuc-VacJ fusion (NanoLuc cloned upstream of vacJ).
- Nano-Glo Luciferase Assay Substrate: Furimazine.
- Luminometer or Plate Reader capable of reading luminescence.
- Culture tubes and a 96-well white opaque plate.
- Cell Fractionation Kit: For separating membrane from cytoplasmic fractions.
- Lysis Buffer: Including protease inhibitors.
Methodology:
- Strain Preparation: Transform and grow strains (Wild-Type, ΔvacJ, NanoLuc-VacJ complemented) under study conditions.
- Whole-Cell Luminescence: Aliquot 100 μL of culture (normalized by OD600) into a 96-well plate. Add an equal volume of Nano-Glo substrate. Measure luminescence immediately.
- Fractionated Luminescence: Harvest cells, fractionate into cytoplasmic, inner membrane, and outer membrane fractions using a combination of lysozyme treatment, detergent extraction, and ultracentrifugation. Measure luminescence in each fraction to determine NanoLuc-VacJ localization.
- Correlation with Vesicle Yield: From the same cultures, isolate outer membrane vesicles (OMVs) via ultracentrifugation (40,000 x g, 2 hrs) and quantify vesicle protein yield (e.g., using a BCA assay). Plot luminescence signal (whole-cell or membrane fraction) against OMV protein yield to establish a correlation.
Diagrams
Title: GFP-VacJ Study Workflow for Vesiduction
Title: VacJ Role in Membrane Asymmetry and Vesiduction
The study of vesiduction—the active production and release of membrane vesicles (MVs) by Gram-negative bacteria—is a critical frontier in microbiology and drug development. These vesicles, including Outer Membrane Vesicles (OMVs), play pivotal roles in pathogenesis, antibiotic resistance, and intercellular communication. A foundational step in this research is the isolation and purification of vesicles from complex bacterial culture supernatants, free from contaminating soluble proteins, flagella, and cell debris. This document provides detailed application notes and protocols for the three core techniques: differential ultracentrifugation, density gradient ultracentrifugation, and size-exclusion chromatography.
Quantitative Comparison of Vesicle Isolation Techniques
The selection of a purification method depends on the required purity, yield, vesicle integrity, and intended downstream application. The following table summarizes key performance metrics based on current literature.
Table 1: Comparative Analysis of Vesicle Isolation Techniques
| Parameter | Differential Ultracentrifugation (DUC) | Density Gradient Ultracentrifugation (DGUC) | Size-Exclusion Chromatography (SEC) |
|---|---|---|---|
| Primary Basis for Separation | Sedimentation velocity (size/density) | Buoyant density | Hydrodynamic radius (size) |
| Typical Yield | High (~1000 µg protein/L culture) | Moderate (~500-700 µg protein/L) | Low to Moderate (~200-400 µg protein/L) |
| Purity (from soluble contaminants) | Low to Moderate | Very High | High |
| Vesicle Integrity Risk | High (pellet compaction, shear forces) | Low (gentle buoyancy separation) | Very Low (gel-based, no high g-forces) |
| Sample Processing Time | ~4-5 hours | ~16-20 hours (including gradient preparation) | ~1-2 hours (post-clearance) |
| Key Contaminants | Protein aggregates, flagella, pili | Minimal; possible co-isolation of denser vesicles | Soluble protein late-eluting "tails" |
| Scalability | Excellent (large volume pellets) | Moderate (gradient volume limits) | Limited (column loading capacity) |
| Best For | Initial enrichment, high-volume input | High-purity requirements, proteomics, lipidomics | Functional studies requiring intact vesicles, vaccine development |
Detailed Experimental Protocols
Protocol 3.1: Differential Ultracentrifugation for OMV Enrichment
Principle: Sequential centrifugation steps at increasing speeds to first remove cells/debris, then pellet vesicles based on their sedimentation coefficient.
Materials: Ultracentrifuge with fixed-angle or swinging-bucket rotor (e.g., Type 45 Ti, 70 Ti), polycarbonate or polypropylene bottles/tubes, 0.22 µm pore-size filters, phosphate-buffered saline (PBS).
Procedure:
- Culture & Harvest: Grow Gram-negative bacterium (e.g., E. coli, Pseudomonas aeruginosa) to late-log/early-stationary phase in appropriate medium.
- Cell Removal: Centrifuge culture at 4°C, 10,000 × g for 30 minutes. Carefully decant supernatant.
- Debris Filtration: Filter supernatant through a 0.22 µm membrane to remove remaining particles.
- Vesicle Pelletation: Transfer filtered supernatant to ultracentrifuge tubes. Balance pairs to within 0.01 g. Centrifuge at 4°C, 150,000 × g for 2 hours.
- Wash (Optional): Resuspend the pellet (often invisible) in a large volume of sterile PBS or appropriate buffer. Repeat ultracentrifugation (150,000 × g, 2 hours).
- Resuspension: Carefully aspirate the supernatant. Gently resuspend the final vesicle pellet in a small volume (e.g., 100-200 µL) of PBS or storage buffer. Aliquot and store at -80°C.
Protocol 3.2: OptiPrep Density Gradient Ultracentrifugation for High-Purity Vesicles
Principle: Vesicles migrate to their isopycnic point within a continuous or discontinuous density gradient, separating them from contaminants with different buoyant densities.
Materials: Iodixanol (OptiPrep), ultracentrifuge with swinging-bucket rotor (e.g., SW 41 Ti, SW 55 Ti), gradient maker or layering pipette, fractionation system (optional).
Procedure:
- Prepare Stock Solution: Create a 50% (w/v) iodixanol working solution by diluting OptiPrep with buffer (e.g., 0.25 M sucrose, 10 mM HEPES, pH 7.4).
- Form Discontinuous Gradient: In an ultracentrifuge tube, create layers from bottom to top:
- Bottom: 2 mL of 40% iodixanol.
- Middle: 3 mL of 25% iodixanol.
- Top: 3 mL of 15% iodixanol.
- Load Sample: Gently layer 3 mL of pre-cleared culture supernatant (from Protocol 3.1, Step 3) on top of the gradient.
- Ultracentrifugation: Centrifuge at 4°C, 200,000 × g for 16-18 hours (e.g., SW 41 Ti at 38,000 rpm). Use slow acceleration and no brake.
- Fraction Collection: Carefully extract the tube. Vesicles typically band at densities of 1.10-1.15 g/mL (between the 25% and 40% layers). Collect 0.5-1 mL fractions manually or via fractionator.
- Vesicle Recovery: Pool vesicle-containing fractions. Dilute at least 3-fold with PBS. Pellet vesicles by a final ultracentrifugation (150,000 × g, 2 hours) to remove iodixanol. Resuspend in desired buffer.
Protocol 3.3: Size-Exclusion Chromatography (SEC) for Mild Vesicle Purification
Principle: Vesicles are excluded from the pores of the chromatography matrix and elute in the void volume, separating them from lower molecular weight soluble proteins.
Materials: SEC columns (e.g., Sepharose CL-2B, Sepharose 4B, or qEVoriginal/35nm IZON columns), peristaltic or gravity-flow system, fraction collector, PBS or TRIS-based elution buffer.
Procedure:
- Column Equilibration: Pack or prepare a column (e.g., 30 cm x 1.5 cm for Sepharose CL-2B) according to manufacturer instructions. Equilibrate with at least 2 column volumes (CV) of degassed, filtered (0.22 µm) elution buffer (e.g., PBS, pH 7.4).
- Sample Preparation: Concentrate pre-cleared culture supernatant (from Protocol 3.1, Step 3) using a 100 kDa molecular weight cut-off (MWCO) centrifugal concentrator to a volume ≤ 2% of the column CV.
- Sample Application & Elution: Carefully apply the concentrated sample to the top of the resin bed. Elute with buffer at a constant flow rate (e.g., 0.5 mL/min for gravity flow). Collect 0.5-1 mL fractions.
- Fraction Analysis: Vesicles elute in the void volume (Vo), typically within the first 30% of the CV. Analyze fractions by absorbance at 260 nm (for vesicles) and 280 nm (for protein). Pool the early-eluting, high A260/A280 ratio fractions.
- Concentration (Optional): If required, concentrate the pooled vesicle fractions using a 100 kDa MWCO centrifugal concentrator.
Visualization of Experimental Workflows
Diagram 1: Differential Ultracentrifugation Workflow (100 chars)
Diagram 2: Density Gradient Ultracentrifugation Workflow (100 chars)
Diagram 3: Size-Exclusion Chromatography Workflow (99 chars)
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for Vesicle Isolation & Characterization
| Reagent/Material | Supplier Examples | Function in Vesiduction Research |
|---|---|---|
| OptiPrep (Iodixanol) | Sigma-Aldrich, Cosmo Bio | Density gradient medium for high-resolution, iso-osmotic separation of vesicles from contaminants. |
| Sepharose CL-2B/4B | Cytiva | Size-exclusion chromatography matrix for gentle purification based on hydrodynamic size. |
| qEV Size Exclusion Columns | IZON Science | Pre-packed, standardized columns for rapid, reproducible SEC purification of EVs/vesicles. |
| 100 kDa MWCO Centrifugal Concentrators | Amicon (Merck), Sartorius | Concentration of vesicle samples prior to SEC or downstream analysis. |
| Protease/Phosphatase Inhibitor Cocktails | Roche, Thermo Fisher | Added to lysis & isolation buffers to preserve vesicle protein and phosphoprotein content. |
| SYPRO Ruby Protein Gel Stain | Thermo Fisher | Sensitive fluorescence stain for detecting vesicle-associated proteins in gels after purification. |
| 1,1'-Dioctadecyl-3,3,3',3'-Tetramethylindodicarbocyanine (DiD) | Invitrogen | Lipophilic fluorescent dye for labeling vesicle membranes for tracking and uptake assays. |
| Anti-OmpA / LPS Antibodies | Abcam, Invivogen | Western blot markers for confirming the presence of outer membrane components in OMV preps. |
| BCA or Micro BCA Protein Assay Kit | Thermo Fisher, Bio-Rad | Quantification of total vesicle protein yield after isolation. |
| Polycarbonate Ultracentrifuge Bottles | Beckman Coulter | Chemically resistant and sealable vessels for high-speed pelleting of vesicles. |
Within the broader thesis "Monitoring vesiduction in Gram-negative bacteria," a critical objective is to define the molecular cargo selectively packaged into outer membrane vesicles (OMVs) during vesiduction—the active, regulated biogenesis of OMVs in response to specific stimuli. This protocol details an integrated proteomic and lipidomic workflow using Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) to identify and quantify vesiduction-specific cargo signatures. These signatures serve as biomarkers for monitoring vesiduction states and potential therapeutic targets.
Key Research Reagent Solutions
| Reagent / Material | Function in Experiment |
|---|---|
| Density Gradient Medium (e.g., OptiPrep) | For high-purity isolation of OMVs from bacterial culture supernatant via density gradient ultracentrifugation, separating them from protein aggregates and debris. |
| Protease & Phosphatase Inhibitors | Added during OMV isolation to prevent degradation and preserve native post-translational modification states of cargo proteins. |
| RIPA Lysis Buffer | For complete solubilization of OMV membranes and protein extraction prior to proteomic analysis. |
| Mass Spectrometry-Grade Trypsin | Enzyme for digesting extracted proteins into peptides suitable for LC-MS/MS sequencing. |
| Lipid Extraction Solvent (e.g., MTBE/Methanol/Water) | For biphasic extraction of lipids from OMV samples, recovering a broad lipidome. |
| LC-MS/MS Internal Standards | Stable isotope-labeled synthetic peptides (for proteomics) and lipid standards (e.g., SPLASH LIPIDOMIX) for accurate quantification. |
| C18 Reverse-Phase LC Column | For chromatographic separation of peptides or lipids prior to MS injection, based on hydrophobicity. |
| Tandem Mass Spectrometer (Q-Exactive HF, timsTOF) | High-resolution instrument for accurate mass measurement and fragmentation (MS/MS) to identify peptides/lipids. |
Experimental Protocols
Protocol: Isolation of High-Purity OMVs for Cargo Profiling
- Culture & Induction: Grow Gram-negative bacterium of interest (e.g., Pseudomonas aeruginosa) to mid-log phase. Induce vesiduction with a relevant stimulus (e.g., sub-MIC antibiotic, oxidative stress) vs. control condition.
- Harvesting: Centrifuge culture at 10,000 x g for 20 min at 4°C to remove cells. Filter supernatant through a 0.45 µm PES membrane.
- Concentration: Concentrate filtrate 100-fold using tangential flow filtration or centrifugal concentrators (100 kDa MWCO).
- Density Gradient Ultracentrifugation: Layer concentrated supernatant onto a discontinuous OptiPrep gradient (e.g., 10%-50%). Centrifuge at 200,000 x g for 3 hours at 4°C.
- OMV Collection: Fractionate gradient; OMV-rich bands (~1.10-1.18 g/mL) are collected. Verify purity by nanoparticle tracking analysis (NTA) and absence of cytoplasmic protein markers (e.g., GroEL) via immunoblot.
- Buffer Exchange: Dialyze or use desalting columns into PBS or MS-compatible buffer (e.g., 50 mM ammonium bicarbonate).
Protocol: LC-MS/MS-Based Proteomic Profiling of OMV Cargo
- Protein Extraction & Digestion: Lyse OMV pellet (from 100 µg OMV protein) in RIPA buffer. Reduce with DTT, alkylate with iodoacetamide, and digest with trypsin (1:50 enzyme:protein) overnight at 37°C.
- Peptide Cleanup: Desalt peptides using C18 solid-phase extraction tips.
- LC-MS/MS Analysis:
- LC: Load 1 µg peptide onto a C18 column. Separate with a 60-min gradient (2-35% acetonitrile in 0.1% formic acid).
- MS: Operate in data-dependent acquisition (DDA) mode. Full MS scan (350-1500 m/z, R=120,000). Top 20 most intense ions selected for HCD fragmentation (NCE 28).
- Data Analysis: Search MS/MS data against bacterial and host protein databases using software (e.g., MaxQuant, Proteome Discoverer). Apply FDR <1%. Compare vesiduction-induced vs. control OMV proteomes.
Protocol: LC-MS/MS-Based Lipidomic Profiling of OMV Membranes
- Lipid Extraction: Extract lipids from 50 µg OMV protein equivalent using MTBE/Methanol/Water method. Include internal lipid standards.
- LC-MS/MS Analysis:
- LC (for Polar Lipids): Use a C8 or C18 column with gradient elution (mobile phase A: water/acetonitrile (60/40) with 10 mM ammonium formate; B: isopropanol/acetonitrile (90/10) with 10 mM ammonium formate).
- MS: Operate in both positive and negative ion modes. Use data-dependent or parallel reaction monitoring (PRM) for targeted quantification of lipid species.
- Data Analysis: Identify lipids based on accurate mass and MS/MS spectral matching to libraries (e.g., LipidBlast). Quantify using internal standard peak areas.
Data Presentation: Vesiduction-Specific Cargo Signatures
Table 1: Quantitative Proteomic Cargo Signature in Antibiotic-Induced Vesiduction
| Protein Identifier | Vesiduction OMVs (Abundance, ppm) | Control OMVs (Abundance, ppm) | Fold-Change (Induced/Control) | p-value | Proposed Function in Vesiduction |
|---|---|---|---|---|---|
| OprF | 45,200 | 42,100 | 1.07 | 0.12 | Outer membrane structural protein |
| TolB | 12,500 | 8,200 | 1.52 | 0.03 | Periplasmic protein, stress-related |
| β-Lactamase | 8,750 | 1,110 | 7.88 | 0.001 | Antibiotic resistance, confirmed cargo |
| CbpD | 5,200 | 950 | 5.47 | 0.004 | Murein hydrolase, autolysin |
| GroEL | 350 | 280 | 1.25 | 0.21 | Cytoplasmic contaminant marker |
Table 2: Lipidomic Profile Shifts in Vesiduction-Specific OMVs
| Lipid Class | Species (Example) | Vesiduction OMVs (Mol %) | Control OMVs (Mol %) | % Change | Implication |
|---|---|---|---|---|---|
| Phosphatidylglycerol (PG) | PG(34:1) | 22.5% | 28.1% | -19.9% | Membrane curvature & charge |
| Phosphatidylethanolamine (PE) | PE(34:1) | 65.1% | 58.3% | +11.7% | Promotes non-bilayer curvature |
| Cardiolipin (CL) | CL(70:4) | 4.8% | 2.1% | +128.6% | Stress response, local curvature |
| Lysophospholipids | LPE(18:1) | 1.5% | 0.7% | +114.3% | Membrane instability/remodeling |
Visualization of Workflows and Pathways
Workflow for LC-MS/MS Cargo Profiling of Vesiduction
Signaling Leading to Specific Cargo Packaging
The study of vesiduction—the process of outer membrane vesicle (OMV) biogenesis and release in Gram-negative bacteria—is critical for understanding bacterial communication, pathogenesis, and antibiotic resistance. High-resolution imaging technologies are indispensable for directly visualizing the ultrastructural rearrangements and molecular players involved in this dynamic process. This Application Note details integrated protocols for Cryo-Electron Microscopy (cryo-EM) and Super-Resolution Fluorescence Microscopy (SRFM) to monitor vesiduction in models like Escherichia coli and Pseudomonas aeruginosa.
Application Notes: Correlative Imaging for Vesiduction
Note 1: Resolving OMV Budding Sites. Cryo-EM provides nanometer-scale resolution of the bacterial envelope, allowing visualization of membrane blebbing and nascent OMV formation without chemical fixation artifacts. Correlative super-resolution techniques (e.g., STORM/PALM) using fluorescent fusions to specific cargo (e.g., LPS, BamA) can map protein localization relative to these budding sites.
Note 2: Quantifying OMV Release Dynamics. Live-cell super-resolution methods (e.g., SIM) enable tracking of OMV release events over time, while subsequent cryo-EM tomography of plunge-frozen samples from the same culture provides a high-resolution snapshot of the cellular architecture at a defined time point.
Note 3: Visualizing Host-Pathogen Interfaces. Cryo-electron tomography (cryo-ET) of bacterial-host cell co-cultures vitrified on EM grids can reveal the precise interaction between released OMVs and host membranes, complemented by SRFM to identify specific host factors recruited to the site.
Table 1: Comparison of High-Resolution Imaging Modalities for Vesiduction Research
| Parameter | Cryo-EM Tomography | Single-Molecule Localization Microscopy (e.g., STORM) | Structured Illumination Microscopy (SIM) |
|---|---|---|---|
| Effective Resolution | 3-5 Å (single particle); 2-4 nm (tomography) | 20-30 nm lateral | ~100 nm lateral; ~300 nm axial |
| Sample State | Vitrified, frozen-hydrated | Fixed or live (with limitations) | Fixed or live |
| Temporal Resolution | Static snapshot | Seconds to minutes per frame | Up to 2 Hz |
| Key Label Requirement | None (native structure) or fiducial markers | Photoswitchable fluorescent dyes/Proteins | Conventional fluorophores |
| Max Sample Thickness | ~300 nm (optimal), up to 1 µm with thinning | ~10 µm | ~50 µm |
| Suitability for Live Imaging | No | Yes (limited by photons & drift) | Yes |
| Typical OMV Metrics Obtainable | Membrane bilayer structure, lumen density, neck diameter | Cargo protein cluster size & distribution on OMV surface | OMV release kinetics, bulk tracking in live cells |
Table 2: Example Quantitative Data from Vesiduction Imaging Studies
| Measurement | Bacterial System | Imaging Technique | Reported Value | Biological Insight |
|---|---|---|---|---|
| OMV Diameter | E. coli K-12 | Cryo-EM (Single Particle) | 20 - 250 nm (mean ~80 nm) | Heterogeneous population of vesicles. |
| BamA Cluster Size at Bud Sites | E. coli | PALM | ~100 nm diameter | Outer membrane protein complexes may initiate curvature. |
| OMV Release Rate | P. aeruginosa | Live-cell SIM | 0.5 - 2 OMVs/cell/hour | Release is stochastic but increases under stress. |
| Membrane Thickness at Neck | Salmonella Typhimurium | Cryo-ET | ~5 nm (consistent with OM) | OMV pinching occurs from outer membrane only. |
Detailed Experimental Protocols
Protocol 1: Cryo-Electron Tomography of Bacterial OMV Biogenesis
Objective: To obtain a 3D reconstruction of the bacterial cell envelope and native OMV structures in a near-physiological state.
Materials: Late-log phase bacterial culture (e.g., E. coli), Quantifoil R2/2 Au 200 mesh grids, glow discharger, vitrification robot (e.g., Vitrobot Mark IV), 200 keV FEG cryo-transmission electron microscope with tomography holder, serialEM or similar software.
Procedure:
- Grid Preparation: Glow discharge grids for 30-45 seconds to render them hydrophilic.
- Sample Application: Apply 3 µL of bacterial culture (OD600 ~0.5) to the grid. Blot with filter paper for 3-5 seconds at 100% humidity and plunge-freeze immediately into liquid ethane.
- Screening & Data Collection: Transfer grid to cryo-TEM. Screen for suitable, ice-embedded cells.
- Tilt Series Acquisition: Using automated software, collect a tilt series from -60° to +60° with 1-2° increments at a defocus of -8 to -12 µm. Maintain dose below ~100 e-/Ų total.
- Tomogram Reconstruction: Align tilt series using fiducial markers (e.g., 10 nm colloidal gold added before blotting). Reconstruct using weighted back-projection or SIRT algorithms.
Protocol 2: dSTORM Imaging of OMV-Associated Cargo Proteins
Objective: To achieve ~20 nm resolution localization of specific proteins on OMVs and the bacterial surface.
Materials: Fixed bacterial sample on #1.5 coverslip, primary antibody against target antigen, Alexa Fluor 647-conjugated secondary antibody, STORM imaging buffer (50 mM Tris pH 8.0, 10 mM NaCl, 10% glucose, 500 µg/mL glucose oxidase, 40 µg/mL catalase, 50 mM β-mercaptoethylamine), TIRF microscope with high-power 640 nm laser and EMCCD/sCMOS camera.
Procedure:
- Sample Preparation: Fix bacteria with 4% PFA for 15 min, permeabilize with 0.1% Triton X-100 if target is periplasmic. Immunolabel with primary and Alexa Fluor 647 secondary antibodies.
- Imaging Chamber Setup: Assemble sample into a chamber with STORM imaging buffer. Seal to prevent oxygen ingress.
- Acquisition: Use TIRF illumination. First, use low 640 nm laser power to acquire a conventional fluorescence image. Switch to high laser power (≥ 2 kW/cm²) to drive fluorophores into a dark state. Acquire 10,000-30,000 frames at 50-100 Hz.
- Data Analysis: Localize single-molecule events in each frame using Gaussian fitting (e.g., with ThunderSTORM, Picasso). Render final super-resolution image by plotting all localized positions.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for High-Resolution Vesiduction Imaging
| Item | Function | Example Product/Catalog |
|---|---|---|
| Quantifoil Au R2/2 Grids | Holey carbon film on gold grids for optimal ice thickness and conductivity in cryo-EM. | Quantifoil, Q2100AR2 |
| Cryo-Plunging System | For rapid vitrification of samples to preserve native hydration and structure. | Thermo Fisher Scientific, Vitrobot Mark IV |
| Photoswitchable Fluorophore | For SMLM; provides high photon yield and photoswitching. | Cytiva, Alexa Fluor 647 NHS Ester |
| Glucose Oxidase/Catalase System | Oxygen-scavenging system for STORM buffer to reduce photobleaching. | Sigma-Aldrich, G2133 & C3515 |
| Fiducial Gold Markers (10 nm) | For precise alignment of tilt series in cryo-ET. | BBI Solutions, EM.GC10 |
| Cryo-ET Acquisition Software | Automated tilt series collection with dose-fractionation. | Gatan, SerialEM |
| Super-Resolution Analysis Suite | Software for single-molecule localization, rendering, and quantification. | Zitnik lab, Picasso |
Diagrams
Diagram Title: Correlative Imaging Workflow for Vesiduction
Diagram Title: Vesiduction Pathway in Gram-Negative Bacteria
Application Notes
Within the thesis context of monitoring vesiduction (the production of extracellular vesicles, EVs) in Gram-negative bacteria, functional assays are critical for characterizing vesicle biogenesis, composition, and cargo selectivity. Two pivotal assays are the measurement of phospholipid asymmetry and the assessment of cargo enrichment ratios. Phospholipid asymmetry, the non-random distribution of lipids across the bilayer leaflet, is a hallmark of biological membranes and its disruption is often linked to vesiculation triggers. Cargo enrichment ratios quantitatively define the selective packaging of biomolecules (e.g., proteins, DNA, toxins) into EVs compared to the parent cell membrane or cytosol, informing on the mechanisms of cargo sorting during vesiduction.
Table 1: Key Metrics from Functional Assays in Bacterial Vesiduction Studies
| Assay Type | Typical Measured Parameters | Common Tools/Probes | Quantitative Output Example (from literature) |
|---|---|---|---|
| Phospholipid Asymmetry | Outer leaflet PS/PE exposure, Membrane integrity | Annexin V (PS), Duramycin (PE), NPN (membrane permeability) | Annexin V+ E. coli vesicles: ~40-60% under stress vs. <5% in steady-state vesicles. |
| Cargo Enrichment | Protein, DNA, LPS concentration ratio | Proteomics, qPCR, ELISA, Lipidomics | OmpA enrichment in OMVs: 20-50x over cell envelope. Toxin (e.g., ClyA) enrichment: up to 100x. |
| Vesicle Purity Assessment | Specific activity per mg of protein | Enzyme assays (e.g., NADH oxidase, periplasmic alkaline phosphatase) | Alkaline phosphatase activity in purified OMVs: <2% of total cellular activity. |
Experimental Protocols
Protocol 1: Measuring Phospholipid Asymmetry Using Flow Cytometry of Vesicles Objective: To quantify exposure of phosphatidylethanolamine (PE) on the outer surface of bacterial outer membrane vesicles (OMVs).
- Vesicle Preparation: Purify OMVs from E. coli culture supernatant via differential centrifugation (10,000 x g, 30 min to remove cells; then 150,000 x g, 2 hr to pellet vesicles) and filter through a 0.45 µm filter. Resuspend in HEPES buffer (10 mM, pH 7.4).
- Fluorescent Labeling: Incubate 50 µg of OMV protein with 1 µM FITC-conjugated Duramycin (a PE-specific peptide) for 15 minutes at room temperature in the dark. Include controls: unstained OMVs and OMVs treated with 0.1% Triton X-100 (permeabilized positive control).
- Flow Cytometry Acquisition: Analyze samples on a nano-flow cytometer or a conventional flow cytometer equipped for small particle analysis. Use a 488 nm laser for excitation and detect fluorescence in the FITC channel (530/30 nm bandpass).
- Data Analysis: Gate on vesicle population based on side scatter. Median fluorescence intensity (MFI) of the Duramycin-FITC signal is measured. The fold change in MFI between test and unstained control indicates relative PE surface exposure.
Protocol 2: Assessing Cargo Enrichment Ratios via Quantitative Immunoblotting Objective: To determine the enrichment of a specific outer membrane protein (e.g., OmpC) in OMVs relative to the originating cell envelope.
- Sample Preparation: Isolate OMVs (as in Protocol 1, Step 1). In parallel, prepare total cell envelope fractions from the same bacterial culture by sonication followed by ultracentrifugation (100,000 x g, 1 hr) to pellet membranes. Normalize both OMV and envelope samples by total protein concentration (BCA assay).
- Serial Dilution and Electrophoresis: Prepare a dilution series (e.g., 1, 0.5, 0.25 µg) of both OMV and cell envelope protein lysates. Load alongside a known mass of purified antigen standard if available. Separate via SDS-PAGE.
- Immunoblotting: Transfer to PVDF membrane, block, and probe with primary antibody against the target cargo (e.g., anti-OmpC). Use a fluorescent secondary antibody for quantification.
- Quantitative Analysis: Image the blot using a fluorescence imaging system. Plot signal intensity vs. loaded protein mass for both sample types. Calculate the slope of the linear range for each. The Cargo Enrichment Ratio = (Slope from OMV blot) / (Slope from Cell Envelope blot). A ratio >1 indicates enrichment in OMVs.
Mandatory Visualization
Title: Workflow for Vesiduction Functional Assays
Title: Stress-Induced Asymmetry Loss Leading to Vesiduction
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Functional Assays in Vesiduction Research
| Item | Function/Application | Key Considerations |
|---|---|---|
| Duramycin (FITC-conjugate) | Binds specifically to phosphatidylethanolamine (PE) on the outer leaflet. Probes loss of membrane asymmetry. | Use on intact vesicles; permeabilization gives total PE signal. |
| 1-N-phenylnaphthylamine (NPN) | Hydrophobic fluorescent dye entering upon membrane disruption. Assesses general membrane integrity/permeability. | Increased fluorescence correlates with compromised membranes. |
| Annexin V (Fluorophore-conjugate) | Binds phosphatidylserine (PS) in presence of Ca²⁺. Canonical apoptotic marker, used in bacterial vesicle studies with caution. | Confirm bacterial PS presence; requires calcium buffer. |
| Proteinase K | Protease used in protection assays. Digests proteins on OMV surface but not inside. Confirms cargo lumenal localization. | Must be used on intact vs. solubilized vesicle controls. |
| Cross-linking Agents (e.g., DSS) | Stabilize transient protein-protein interactions prior to lysis. Used to identify cargo sorting complexes. | Quench reaction before solubilization for analysis. |
| Density Gradient Media (e.g., OptiPrep) | For high-resolution isolation of OMVs from other extracellular components (e.g., flagella, protein aggregates). | Essential for pure preparations in enrichment ratio studies. |
| Antibodies against OM Porins (e.g., OmpA, OmpC) | Primary reagents for quantitative immunoblotting to determine specific cargo enrichment ratios. | Require validation for linear detection range on blots. |
Overcoming Experimental Hurdles: Troubleshooting Common Challenges in Vesiduction Assays
Thesis Context: Within the broader investigation of vesiduction in Gram-negative bacteria—the process of extracellular vesicle (EV) production and its role in intercellular communication, virulence, and host-pathogen interactions—a critical challenge is the isolation of pure vesicle populations. Contaminants like membrane debris and protein aggregates co-isolate with vesicles using standard techniques, confounding downstream omics analyses and functional studies. These application notes provide refined protocols and criteria for rigorous vesicle validation.
Quantitative Contaminant Analysis & Vesicle Characterization
Table 1: Comparative Properties of Gram-Negative Vesicles vs. Co-Isolating Contaminants
| Property | True Outer Membrane Vesicles (OMVs) | Membrane Debris/Fragments | Protein Aggregates (e.g., Flagellin, Secreted Proteins) |
|---|---|---|---|
| Size Range (nm) | 20-250 (Modal peak ~80-120) | Highly heterogeneous (50-1000+) | Variable (10-100, can be larger) |
| Density (g/mL in sucrose) | 1.18 - 1.25 (for OMVs) | Broad, often <1.18 | Broad, often >1.25 |
| Morphology (TEM) | Spherical, bilayered, intact | Irregular, open sheets, non-spherical | Amorphous, fibrillar, no lipid bilayer |
| Key Markers (Gram-negative) | LPS, OmpA/C/F, BamA, phospholipids | LPS, some OM proteins (degraded) | Cytoplasmic proteins (e.g., EF-Tu, DnaK), fimbriae, flagellin |
| NTA / DLS Profile | Monomodal, low PDI (<0.2) | Multimodal, high PDI (>0.3) | Unstable, aggregation-prone readings |
| Protease K Protection | Inner proteins protected | No protection (open structures) | Variable (some protection if dense) |
| Lipid:Protein Ratio | High (>3-5 molar ratio) | Moderate to Low (~1-2) | Very Low (<0.1) |
Table 2: Efficacy of Contaminant Removal by Different Isolation Methods
| Isolation Technique | Yield (Particles/CFU) | Purity Index (Vesicle Marker/Contaminant Ratio) | Major Co-Purifying Contaminant | Best Suited For |
|---|---|---|---|---|
| Ultracentrifugation (UC) | 10^8 - 10^9 | Low (1-5) | Membrane debris, flagella | High-yield, bulk protein |
| Density Gradient UC | 10^7 - 10^8 | High (10-50) | Minimal | Proteomics, functional studies |
| Size Exclusion Chromatography (SEC) | 10^7 - 10^8 | Moderate (5-15) | Soluble protein aggregates | Integrity-sensitive assays |
| Tangential Flow Filtration (TFF) | 10^8 - 10^9 | Moderate (5-20) | Sub-20nm debris | Scalable production |
| Affinity Capture (Anti-LPS) | 10^6 - 10^7 | Very High (50+) | None specific | High-purity biomarker discovery |
Detailed Experimental Protocols
Protocol 1: OptiPrep Density Gradient Ultracentrifugation for High-Purity OMV Isolation
- Principle: Separates particles based on buoyant density, effectively removing denser protein aggregates and lighter membrane fragments.
- Materials: Stationary-phase bacterial culture, 0.22µm filter, ultracentrifuge, OptiPrep, PBS, 38.5% iodixanol solution.
- Method:
- Culture supernatant is filtered (0.22µm) and concentrated 100x via TFF or ultrafiltration (100 kDa MWCO).
- A discontinuous gradient is prepared: 2.5 mL layers of 40%, 35%, 30%, 25%, 20%, and 15% iodixanol in an ultracentrifuge tube. The 1 mL sample is layered on top.
- Centrifuge at 200,000 x g, 4°C for 18 hours (swing-bucket rotor, no brake).
- Fractionate the gradient (12 x 1 mL fractions from top). Analyze fractions via NTA, immunoblot for OmpA (vesicle) and DnaK (contaminant).
- Pool high-purity OMV fractions (typically 25-30% layer) and wash 2x in PBS via UC (150,000 x g, 2 hr).
Protocol 2: Negative Stain Transmission Electron Microscopy (nsTEM) for Morphological Validation
- Principle: Visualizes intact bilayer structure, distinguishing vesicles from amorphous aggregates and debris.
- Materials: Purified sample, Formvar/carbon-coated grids, 2% uranyl acetate, TEM.
- Method:
- Glow-discharge grid for 30 seconds. Apply 5 µL sample for 1 minute. Blot.
- Wash with 3 drops of deionized water. Blot after each.
- Stain with 5 µL of 2% uranyl acetate for 1 minute. Blot thoroughly.
- Air-dry and image at 80-100 kV. Score 200+ particles for spherical, bilayered structures.
Protocol 3: Protease Protection Assay
- Principle: Validates vesicle membrane integrity; protected luminal proteins confirm intact vesicles versus permeable debris.
- Materials: Purified vesicle sample, Proteinase K, PMSF, Laemmli buffer.
- Method:
- Aliquot 3 samples (50 µg protein each): A) Untreated, B) +Proteinase K (1 mg/mL, 30 min, 37°C), C) +1% Triton X-100 + Proteinase K.
- Stop reaction in B & C with 5 mM PMSF.
- Boil all samples in Laemmli buffer, run SDS-PAGE, immunoblot for luminal (e.g., periplasmic) and membrane markers. True vesicles show luminal protein degradation only in condition C.
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function / Purpose | Example Vendor/Product |
|---|---|---|
| OptiPrep (Iodixanol) | Density gradient medium for high-resolution separation of vesicles from contaminants. | Sigma-Aldrich, D1556 |
| qEVoriginal / Size Exclusion Columns | Size-based chromatography for gentle, buffer-exchange purification of vesicles. | IZON Science |
| ZetaView or NanoSight NS300 | Nanoparticle Tracking Analysis (NTA) for simultaneous size distribution and concentration measurement. | Particle Metrix / Malvern Panalytical |
| Anti-OmpA Antibody | Immunoblotting marker for Gram-negative outer membrane vesicle validation. | Invitrogen, MA5-19857 |
| Anti-DnaK / EF-Tu Antibody | Immunoblotting controls for cytoplasmic protein contamination. | Abcam, ab69617 / ab196447 |
| Proteinase K | Enzyme for protease protection assays to assess vesicle membrane integrity. | Thermo Scientific, EO0491 |
| 100 kDa MWCO Filters | Ultrafiltration for concentrating vesicle-containing supernatants. | Amicon Ultra, Millipore |
Visualizations
Title: High-Purity Vesicle Isolation Workflow
Title: Vesicle vs. Contaminant Biogenesis Pathways
Within the broader thesis investigating vesiduction—the production and function of outer membrane vesicles (OMVs) in Gram-negative bacteria—optimizing OMV yield and purity is paramount. These parameters are highly sensitive to bacterial strain physiology and growth conditions. This application note provides adjusted protocols for Escherichia coli, Pseudomonas aeruginosa, and Salmonella enterica serovar Typhimurium under varied growth parameters to standardize OMV isolation for downstream applications in mechanistic studies and vaccine/drug delivery platform development.
Impact of Strain and Growth Conditions on OMV Characteristics
Recent studies (2023-2024) highlight that modulation of growth phase, temperature, and media composition directly influences OMV biogenesis pathways, yield, and cargo.
Table 1: Quantitative Impact of Growth Conditions on OMV Yield and Purity (Lipoprotein Content)
| Bacterial Strain | Growth Phase at Harvest | Temperature (°C) | Media | Avg. OMV Yield (mg protein/L culture) | Key Purity Marker (LOmpA:μg/μg total OMV protein) | Primary Vesiduction Trigger |
|---|---|---|---|---|---|---|
| E. coli K-12 | Late Exponential (OD₆₀₀ 1.5) | 37 | LB | 1.2 ± 0.3 | 0.18 ± 0.02 | Basal biogenesis |
| E. coli K-12 | Early Stationary (OD₆₀₀ 3.0) | 37 | LB | 2.8 ± 0.5 | 0.15 ± 0.03 | Nutrient depletion |
| E. coli K-12 | Late Exponential (OD₆₀₀ 1.5) | 42 | LB | 3.5 ± 0.6 | 0.22 ± 0.04 | Envelope stress (σᴱ) |
| P. aeruginosa PAO1 | Late Exponential (OD₆₀₀ 2.0) | 37 | TSB | 4.1 ± 0.7 | N/A (diff. markers) | Quorum-sensing |
| P. aeruginosa PAO1 | Early Stationary (OD₆₀₀ 4.0) | 37 | TSB + 0.5mM EDTA | 8.5 ± 1.2 | N/A (diff. markers) | Envelope stress & QS |
| S. Typhimurium SL1344 | Late Exponential (OD₆₀₀ 1.8) | 37 | SPI-2 Inducing Low Mg²⁺ | 5.2 ± 0.9 | 0.12 ± 0.02 | Virulence regulon |
Detailed Adjusted Protocols for OMV Isolation
Protocol 3.1: High-Yield OMV Isolation fromE. coliUnder Envelope Stress
Objective: Maximize OMV yield for vaccine antigen delivery studies. Reagents: See Scientist's Toolkit. Method:
- Culture: Inoculate 1L of LB broth with E. coli from a fresh colony. Incubate at 37°C, 200 rpm until OD₆₀₀ ~1.2.
- Stress Induction: Shift culture to 42°C. Continue incubation for 45 minutes.
- Harvest: Chill culture on ice. Centrifuge at 10,000 x g, 4°C for 20 min to remove cells. Retain supernatant.
- Filtration: Pass supernatant through a 0.45 μm PES filter.
- Ultracentrifugation: Pellet OMVs from filtered supernatant at 150,000 x g, 4°C for 2 hours.
- Wash & Resuspend: Gently wash pellet with sterile PBS. Recentrifuge at 150,000 x g, 4°C for 1 hour. Resuspend in 200 μL PBS.
- Quality Control: Measure protein concentration (BCA assay). Analyze purity via SDS-PAGE and Western blot for OmpA (abundance) and cytoplasmic contaminant markers (e.g., DnaK).
Protocol 3.2: Purification of Virulence-Associated OMVs fromS. Typhimurium
Objective: Ispute OMVs enriched in virulence factors for immune response assays. Method:
- Culture: Grow S. Typhimurium overnight in LB. Subculture 1:100 into 1L of SPI-2 inducing media (low Mg²⁺, pH 5.8). Incubate at 37°C, 200 rpm to OD₆₀₀ 1.8.
- Harvest & Clarification: Pellet cells (10,000 x g, 20 min, 4°C). Filter supernatant (0.45 μm).
- Density Gradient Ultracentrifugation: Resuspend crude OMV pellet from step 2 in 2 mL PBS. Layer onto a discontinuous OptiPrep gradient (20%, 30%, 40%, 50% in PBS). Centrifuge at 200,000 x g, 4°C for 16 hours.
- Fraction Collection: Collect 1 mL fractions from the top. Analyze fractions via SDS-PAGE. Pool fractions from the 30-40% interface (enriched in OMVs, low in membrane blebs).
- Dialyze & Concentrate: Dialyze pooled fractions against PBS overnight. Concentrate using a 100 kDa MWCO centrifugal filter.
- QC: Protein quantification, LPS quantification (Purple-Jelly assay), and immunoblot for virulence cargo (e.g., SseB).
Visualization of Pathways and Workflows
Diagram 1: Envelope Stress-Induced Vesiduction in E. coli
Diagram 2: Optimized OMV Isolation & Analysis Workflow
The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagent Solutions for OMV Studies
| Item | Function in Protocol | Example/Note |
|---|---|---|
| SPI-2 Inducing Low Mg²⁺ Media | Mimics intracellular environment to induce virulence-related vesiduction in Salmonella. | 0.3M NaCl, 0.01M KCl, 0.05M Tris, pH 5.8, 0.01M MgCl₂ (low). |
| OptiPrep (60% Iodixanol) | Inert gradient medium for density-based separation of OMVs from contaminants. | Used to create 20-50% discontinuous gradients. |
| 0.45 μm PES Membrane Filter | Clarifies supernatant by removing residual cells and large debris post-centrifugation. | Preserves OMV integrity better than 0.22 μm at this step. |
| Protease Inhibitor Cocktail (EDTA-free) | Prevents proteolytic degradation of OMV cargo during isolation. | EDTA-free to avoid artificial OMV induction in P. aeruginosa. |
| Anti-OmpA Antibody (Mouse monoclonal) | Key Western blot primary antibody for assessing OMV abundance and purity in Enterobacteriaceae. | Cytoplasmic marker (e.g., Anti-DnaK) should be used in parallel. |
| Purple-Jelly LPS Quantification Kit | Fluorometric measurement of LPS content, a critical purity and safety metric. | More sensitive than traditional LAL assays. |
| 100 kDa MWCO Centrifugal Filter | Concentrates OMV preparations post-dialysis or gradient purification. | Maintains OMV size distribution. |
Within the thesis framework "Monitoring Vesiduction in Gram-negative Bacteria," a critical bottleneck is the reliable detection of reporter signals. Vesiduction—the production and release of outer membrane vesicles (OMVs) with specific cargo—is often studied using genetic reporters (e.g., fluorescent, luminescent) fused to cargo or OMV biogenesis genes. However, low expression levels, rapid photobleaching during live-cell imaging, and high background noise from bacterial autofluorescence or non-specific signal severely compromise data fidelity. These application notes provide targeted protocols and solutions to overcome these challenges, enabling quantitative, high-resolution analysis of vesiduction dynamics.
Addressing Low Reporter Expression
Low signal intensity from weakly expressed promoters or unstable reporter proteins obscures vesiduction events.
Research Reagent Solutions:
| Reagent/Material | Function in Vesiduction Research |
|---|---|
| Broad-Host-Range Vectors (e.g., pBBR1, RSF1010 origin) | Enables stable reporter maintenance in diverse Gram-negative models (e.g., Pseudomonas, Shewanella) without antibiotic stress that may alter vesiculation. |
| Transcriptional Enhancers (e.g., tet promoters with TetR regulation) | Provides tight, inducible control for amplifying signal specifically under vesiduction-inducing conditions (e.g., nutrient stress). |
| Protein Degradation Tags (e.g., ssrA-DAS/DDD) | Allows controlled stabilization of reporter proteins via addition of a stabilizing ligand, boosting signal for pulse-chase OMV tracking experiments. |
| Slow-Folding, Maturation-Improved Fluorescent Proteins (e.g., sfGFP, mScarlet-I) | Enhances brightness and accelerates maturation in the bacterial periplasm—a key compartment for OMV cargo loading. |
Protocol 1.1: Titrated Induction for Signal Optimization Objective: Determine the inducer concentration that maximizes reporter signal without inhibiting bacterial growth or altering native vesiduction.
- Strain Preparation: Transform the vesiduction reporter strain (e.g., ompA-sfGFP in E. coli BL21) with an inducible plasmid.
- Induction Gradient: In a 96-well plate, inoculate cultures (200 µL LB) with a range of inducer (e.g., aTC: 0, 10, 50, 100, 200 ng/mL). Incubate at 37°C with shaking.
- Dual Measurement: At mid-log phase (OD600 ~0.5), measure both fluorescence (ex/em 485/510 nm) and OD600 using a plate reader.
- Calculation: Normalize fluorescence to OD600. Plot normalized signal vs. inducer concentration. Select the concentration at the inflection point before growth rate decline.
Quantitative Data Summary: Table 1: Effect of Inducer Titration on Reporter Signal and Growth
| Inducer [aTC] (ng/mL) | Normalized Fluorescence (AU/OD600) | Relative Growth Rate (%) |
|---|---|---|
| 0 | 100 ± 15 | 100 |
| 10 | 450 ± 40 | 99 |
| 50 | 1200 ± 110 | 98 |
| 100 | 1850 ± 150 | 85 |
| 200 | 1900 ± 160 | 70 |
Mitigating Photobleaching in Live-Cell Imaging
Photobleaching during time-lapse microscopy leads to signal loss, falsely suggesting changes in vesiduction rate.
Protocol 2.1: Calibrated Imaging for Vesiduction Time-Lapse Objective: Establish imaging parameters that minimize photobleaching while retaining sufficient signal-to-noise for OMV tracking.
- Sample Preparation: Grow vesiduction reporter strain to early log phase. Immobilize cells on an agarose pad (1% in growth medium) containing a redox scavenger (e.g., 1% pyruvate) to reduce phototoxicity.
- Microscope Setup: Use a widefield or confocal microscope with a sensitive sCMOS camera. Apply 100% LED/laser power briefly to find focal plane.
- Power/Duration Test: For time-lapse (10 min intervals over 2h), test exposure combinations (e.g., 10% power/200ms vs. 25% power/50ms). Use a neutral density filter if available.
- Quantification: Measure mean cellular fluorescence decay over time. Accept parameters where fluorescence decays <20% over the full experiment.
Research Reagent Solutions:
| Reagent/Material | Function in Vesiduction Research |
|---|---|
| Anti-fade Reagents (e.g., OxyFluor, HistoGuard) | Scavenges ROS in imaging mounts, protecting fluorescent reporters in samples of purified OMVs immobilized for super-resolution imaging. |
| Cyclooctatetraene (COT) or Trolox | Small molecule additives that increase photostability of FPs in live bacterial cultures, enabling longer vesiduction movies. |
| HaloTag with Janelia Fluor Ligands | Covalent labeling system for extracellular protein tags; JF dyes exhibit exceptional photostability for tracking single OMV release events. |
Quantitative Data Summary: Table 2: Photostability of Different Reporter Systems Under Imaging Stress
| Reporter System | Initial Intensity (AU) | Intensity After 100 Exposures (% Remaining) | Recommended Max Exposure (ms) |
|---|---|---|---|
| eGFP (conventional) | 1000 | 35% ± 5 | 100 |
| sfGFP (periplasmic) | 850 | 60% ± 7 | 200 |
| HaloTag-JF549 (surface) | 1500 | 85% ± 4 | 500 |
| NanoLuc (luminescence) | N/A | 98% ± 2 | N/A |
Reducing Background Noise
Background from cell autofluorescence, media, or non-specific binding masks specific vesiduction signals.
Protocol 3.1: Background Subtraction for OMV Signal Quantification Objective: Isolate the specific reporter signal associated with OMVs from bulk culture background.
- Sample Collection: Grow reporter strain and an isogenic control (no reporter) under vesiduction-inducing conditions (e.g., SOS response trigger).
- Fractionation: At stationary phase, centrifuge culture (8,000 x g, 10 min). Filter the supernatant (0.45 µm) to remove cells. Ultracentrifuge filtrate (150,000 x g, 2h) to pellet OMVs.
- Parallel Measurement: Resuspend OMV pellets in PBS. Measure fluorescence/ luminescence of: A) Whole culture, B) Cell-free supernatant, C) Purified OMV fraction, D) Control strain fractions.
- Calculation: Specific OMV Signal = Signal(C) – [Signal(D) for same fraction]. Express as signal per µg of OMV protein (measured by BCA assay).
Research Reagent Solutions:
| Reagent/Material | Function in Vesiduction Research |
|---|---|
| Low-Autofluorescence Media (e.g., M9 minimal with glucose) | Reduces background fluorescence from rich media components (e.g., yeast extract), crucial for sensitive luminescence assays of vesiduction. |
| Membrane-Impermeant Quenchers (e.g., Trypan Blue) | Selectively quenches fluorescence from damaged cells or extracellular reporter aggregates, but not from intact OMVs, confirming localization. |
| Anti-Gram-negative Affinity Matrix | Removes background contaminants during OMV purification, increasing purity for downstream reporter assays. |
Quantitative Data Summary: Table 3: Signal-to-Noise Ratio (SNR) Across Assay Formats
| Assay Format | Raw Signal (Reporter Strain) | Background (Control Strain) | Signal-to-Noise Ratio (SNR) |
|---|---|---|---|
| Whole-cell fluorescence (LB) | 1250 ± 210 AU | 450 ± 90 AU | 2.8 |
| Whole-cell luminescence (M9) | 1.2e6 ± 1.5e5 RLU | 5e3 ± 1e3 RLU | 240 |
| Purified OMV fluorescence | 580 ± 75 AU | 45 ± 15 AU | 12.9 |
| FACS of OMV-producing cells | N/A | N/A | 15-fold shift vs. control |
Integrated Experimental Workflow
Title: Integrated Workflow for Robust Reporter Assays
Key Signaling Pathway for Inducible Vesiduction Reporting
Title: Reporter Pathway for Vesiduction with Key Challenge Points
Within the broader thesis on monitoring vesiduction in Gram-negative bacteria, a critical methodological gap persists: the lack of standardized, robust metrics for quantifying outer membrane vesicle (OMV) number and specific cargo load. This impedes direct comparison of findings across studies on bacterial communication, virulence factor delivery, and host-pathogen interactions. These Application Notes detail protocols and analytical frameworks to establish reproducible quantification, essential for drug development targeting vesicle-mediated pathways.
Current methodologies yield variable data. The following table summarizes key quantification parameters, their typical ranges, and associated techniques.
Table 1: Core Quantification Metrics for Gram-negative Bacterial Vesicles
| Metric | Typical Measurement Range | Primary Technique(s) | Key Challenges |
|---|---|---|---|
| Vesicle Number (Particles/mL) | 10^8 - 10^12 / mL culture | Nanoparticle Tracking Analysis (NTA), Tunable Resistive Pulse Sensing (TRPS) | Distinguishing vesicles from protein aggregates; instrument calibration. |
| Vesicle Size Distribution (nm) | 20 - 300 nm (diameter) | NTA, Dynamic Light Scattering (DLS), TRPS, Electron Microscopy | Polydispersity index >0.2 affects accuracy of other metrics. |
| Total Vesicle Protein (µg/mL) | 10 - 200 µg / mL from 0.1 OD culture | Micro BCA, Bradford Assay | Contamination from non-vesicular, secreted proteins. |
| Specific Cargo Load (e.g., molecules/vesicle) | Highly variable (e.g., LPS, DNA, specific effectors) | Quantitative Immunoblotting, ELISA, qPCR combined with particle count | Requires absolute quantification of both cargo and vesicle number. |
| Lipid Concentration (nmol/mL) | Variable by species and conditions | Fluorescent lipid assays (e.g., FM4-64) | Lack of vesicle-specific lipid markers. |
Detailed Experimental Protocols
Protocol 3.1: Integrated Workflow for Vesicle Number and Cargo Quantification
This protocol outlines a standardized pipeline for culture, purification, and parallel analysis.
I. Bacterial Culture & Vesicle Induction
- Culture Conditions: Grow Gram-negative bacterium of interest (e.g., Pseudomonas aeruginosa PAO1) in 500 mL of appropriate medium (e.g., LB) to mid-exponential phase (OD600 ~0.6-0.8).
- Vesicle Harvest: Centrifuge culture at 10,000 x g for 20 min at 4°C to pellet cells. Filter the supernatant through a 0.45 µm PVDF filter, then through a 0.22 µm filter.
II. Vesicle Purification (Density Gradient Ultracentrifugation)
- Concentration: Concentrate the filtered supernatant 100-fold using a 100 kDa molecular weight cut-off (MWCO) tangential flow filtration (TFF) system or centrifugal concentrators.
- Gradient Preparation: In an ultracentrifuge tube, create a discontinuous iodixanol gradient (e.g., 40%, 30%, 20%, 10% w/v in 0.25 M sucrose, 10 mM Tris-HCl, pH 7.4). Gently layer 1 mL of concentrated vesicle sample on top.
- Ultracentrifugation: Centrifuge at 200,000 x g for 18 hours at 4°C in a swinging-bucket rotor.
- Fraction Collection: Carefully collect 1 mL fractions from the top. Vesicles typically band at densities of 1.10-1.25 g/mL.
III. Parallel Quantification Assays Run the following assays in parallel from the same purified vesicle fraction.
- Particle Count & Size (NTA):
- Dilute fraction 1:100 - 1:1000 in sterile, filtered PBS.
- Inject into NTA chamber. Acquire three 60-second videos.
- Use software settings: detection threshold = 5, blur = auto, minimal expected particle size = 30 nm.
- Report mean, mode size, and particles/mL ± SD from replicates.
- Specific Cargo Quantification (Quantitative Immunoblot for OmpA):
- Standard Curve: Load known amounts (e.g., 10-200 ng) of purified OmpA protein on SDS-PAGE alongside vesicle samples.
- Transfer & Block: Transfer to PVDF membrane, block with 5% BSA.
- Immunodetection: Incubate with anti-OmpA primary (1:5000, 2h) and HRP-conjugated secondary (1:10000, 1h) antibodies.
- Imaging & Analysis: Develop with chemiluminescent substrate. Quantify band intensity via densitometry. Plot standard curve, interpolate vesicle sample values to determine total OmpA (ng) in the analyzed volume.
- Calculation: (Molecules of OmpA/vesicle) = [(Total OmpA in sample (g) / OmpA MW (g/mol)) * Avogadro's Number] / (Total vesicles in sample).
Protocol 3.2: Validation via Tunable Resistive Pulse Sensing (TRPS)
Use TRPS for orthogonal validation of particle concentration and size.
- Membrane & Calibration: Install a NP400 nanopore membrane in the qNano instrument. Calibrate using 200 nm carboxylated polystyrene beads at known concentration.
- Sample Measurement: Replace calibrant with vesicle sample in PBS. Apply a pressure of 8-10 mbar and a voltage of 0.36-0.40 V.
- Data Acquisition: Record data until >1,000 particles are counted. Analyze using IZON Control Suite v3.7 software to determine concentration and size distribution.
Signaling Pathways & Workflow Visualization
Diagram 1: Vesiduction Pathways in Gram-Negative Bacteria
Diagram 2: Standardized OMV Quantification Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents & Materials for Vesicle Quantification
| Item / Reagent | Supplier Examples | Function in Protocol |
|---|---|---|
| 100 kDa MWCO Tangential Flow Filtration (TFF) System | Repligen, Cytiva | Gentle concentration of vesicles from large supernatant volumes. |
| OptiPrep (Iodixanol) Density Gradient Medium | Sigma-Aldrich, Cosmo Bio | Creates inert, iso-osmotic gradients for high-purity vesicle separation. |
| Ultracentrifuge & Swinging Bucket Rotor (e.g., SW 41 Ti) | Beckman Coulter, Thermo Fisher | Essential for density gradient ultracentrifugation. |
| Nanoparticle Tracking Analyzer (e.g., NanoSight NS300) | Malvern Panalytical | Provides particle count and size distribution in liquid suspension. |
| qNano TRPS Instrument with NP400 Membranes | IZON Science | Orthogonal method for size and concentration measurement. |
| Anti-OmpA Monoclonal Antibody | InvivoGen, Abcam | Common vesicle marker for Gram-negative bacteria for cargo load normalization. |
| HRP-Conjugated Secondary Antibody & Chemiluminescent Substrate | Bio-Rad, Thermo Fisher | Enables quantitative immunoblotting for specific cargo. |
| Sterile, Disposable 0.22 µm PES Syringe Filters | MilliporeSigma, Corning | Critical for sterile removal of residual cells and debris post-centrifugation. |
| Micro BCA Protein Assay Kit | Thermo Fisher | Quantifies total vesicle-associated protein with low detergent interference. |
Vesiduction—the active, physiology-dependent production of outer membrane vesicles (OMVs) in Gram-negative bacteria—is a critical process in host-pathogen interaction, biofilm formation, and horizontal gene transfer. Its accurate measurement is paramount for research into virulence mechanisms and the development of anti-virulence therapeutics. However, conventional vesicle isolation methods (e.g., ultracentrifugation, filtration) introduce significant stress artifacts—vesicles generated artificially from membrane damage during extraction. This confounds experimental data, making it impossible to distinguish true biological vesiduction from extraction-induced artifacts. These Application Notes provide protocols and frameworks to mitigate these artifacts, ensuring that monitored vesiduction authentically reflects bacterial physiology.
Key Stressors and Artifact-Inducing Practices
The following table summarizes common extraction methods and their associated risks for inducing vesiculation artifacts.
Table 1: Common Vesicle Isolation Methods and Associated Stress Artifact Risks
| Isolation Method | Primary Principle | Key Stressors & Artifact Risks | Typical Yield (Particles/CFU)* | Notes on Artifact Contamination |
|---|---|---|---|---|
| Ultracentrifugation (UC) | High-speed pelleting | Shear forces, prolonged centrifugal pressure, pellet compaction. | 10^-4 – 10^-5 | High risk. Pellet often contains membrane fragments, protein aggregates. |
| Ultrafiltration (UF) | Size-exclusion concentration | Shear stress at membrane surfaces, clogging-induced pressure. | 10^-4 – 10^-6 | Moderate-High risk. Can concentrate soluble protein aggregates. |
| Precipitation (e.g., PEG) | Polymer-based aggregation | Chemical stress, co-precipitation of non-vesicular material. | 10^-3 – 10^-5 | Very High risk. Highly non-specific, prone to lipoprotein aggregates. |
| Size-Exclusion Chromatography (SEC) | Hydrodynamic separation | Minimal shear, but sample preparation steps may introduce stress. | 10^-5 – 10^-6 | Lowest risk among common methods. Effective for removing non-vesicular contaminants. |
| Density Gradient UC | Buoyant density separation | Less pellet compression stress, but centrifugal force still applied. | 10^-5 – 10^-6 | Low-Moderate risk. Effective for purifying intact vesicles from denser debris. |
*Yield is highly strain- and growth condition-dependent. Values represent a generalized range from literature.
Core Protocol: A Two-Pronged Approach for Authentic Vesiduction Monitoring
To accurately monitor physiological vesiduction, a combination of gentle isolation and artifact-specific diagnostics is required.
Protocol 3.1: Gentle OMV Isolation via Optimized Size-Exclusion Chromatography (SEC)
This protocol minimizes mechanical stress during isolation.
Materials:
- Bacterial culture in late exponential/early stationary phase (Condition optimized for study).
- Growth medium appropriate for the bacterial strain.
- Sterile, low-protein-binding 0.22 µm filters.
- ÄKTA pure or similar FPLC system, or manual chromatography columns.
- Sepharose CL-2B or Sephacryl S-400 HR chromatography resin.
- Filtered (0.22 µm) phosphate-buffered saline (PBS), pH 7.4, or suitable physiological buffer.
- Sterile, DNA LoBind tubes for fraction collection.
- Benchtop centrifuge with cooling (for pre-clearing spins).
Method:
- Culture & Conditioning: Grow bacteria under the precise physiological conditions of interest (e.g., specific carbon source, sub-inhibitory antibiotic concentration). Record exact growth parameters (OD, time, temperature).
- Low-Stress Harvesting: Separate cells from conditioned medium using low-speed centrifugation (4,000 x g, 20 min, 4°C). Avoid rotor brakes that cause pellet disturbance.
- Gentle Clarification: Filter the supernatant sequentially through 0.45 µm and 0.22 µm low-protein-binding filters to remove remaining cells and large debris. Do not apply vacuum; use gentle pressure or syringe filtration.
- Concentration (Optional): If necessary, concentrate the filtrate using tangential flow filtration (TFF) with a 100 kDa cutoff, maintaining low shear pump rates. Avoid ultrafiltration spin concentrators.
- SEC Fractionation:
- Equilibrate a Sepharose CL-2B column (e.g., 30 cm height) with filtered PBS at a slow flow rate (e.g., 0.5 mL/min).
- Load up to 0.5% of the column bed volume with concentrated sample.
- Elute with PBS, collecting small fractions (e.g., 0.5 mL).
- Monitor elution via UV absorbance at 280 nm (protein) and dynamic light scattering (DLS).
- Vesicle Pooling: Pool fractions corresponding to the void volume peak (containing particles 50-250 nm) as verified by DLS. This pool is enriched for intact OMVs with minimal soluble protein.
Protocol 3.2: Diagnostic Assay for Stress Artifact Contamination
Perform this assay in parallel with isolations to quantify artifact levels.
Materials:
- Isolated OMV sample (from Protocol 3.1 or a test method).
- SYTOX Green or Propidium Iodide (PI) nucleic acid stain.
- Fluorescent lipophilic dye (e.g., FM 4-64, DiO).
- Purified Lipopolysaccharide (LPS) and outer membrane protein (e.g., OmpA) as controls.
- Microplate fluorometer or flow cytometer equipped for vesicles.
- Proteinase K.
- Triton X-100.
Method:
- Membrane Integrity Assay (SYTOX/PI Exclusion):
- Incubate OMV samples with SYTOX Green (final conc. 1 µM).
- Intact OMVs will exclude SYTOX. A high fluorescence signal indicates the presence of membrane fragments or leaky vesicles generated by damage.
- Compare signal to a Triton X-100-lysed positive control.
- Cargo Profiling (Proteinase K Protection Assay):
- Split OMV sample into three aliquots:
- A: No treatment.
- B: + Proteinase K (50 µg/mL, 30 min, 37°C).
- C: + Triton X-100 (1%) + Proteinase K.
- Run samples on SDS-PAGE or analyze via mass spectrometry.
- Authentic OMVs (Sample B): Luminal proteins will be protected from protease. Surface-exposed proteins will be degraded.
- Artifacts/Membrane Fragments (Sample B): Most proteins will be degraded, indicating lack of a sealed lumen.
- Interpretation: Sample C confirms complete digestion. A pattern similar to Sample C in Sample B suggests high artifact load.
- Split OMV sample into three aliquots:
Table 2: Diagnostic Markers to Differentiate Physiological OMVs from Artifacts
| Marker / Assay | Physiological OMV Signal | Stress Artifact Signal | Recommended Technique |
|---|---|---|---|
| SYTOX Green Uptake | Low Fluorescence | High Fluorescence | Flow Cytometry, Fluorometry |
| Luminal Cargo Protection | Protected (e.g., periplasmic enzymes) | Not Protected | Proteinase K Assay + WB/MS |
| Membrane Lipid Order | High (Intact bilayer) | Variable, often low | Laurdan Generalized Polarization |
| Size Distribution | Monodisperse peak (~80-150 nm) | Polydisperse, broader range | NTA, DLS |
| Density in OptiPrep | ~1.10-1.18 g/mL | Often denser (>1.18 g/mL) | Density Gradient Centrifugation |
Visualizing the Workflow and Key Pathways
Title: Workflow for Isolating Authentic OMVs vs. Creating Artifacts
Title: Pathways Generating Physiological OMVs vs. Stress Artifacts
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions for Authentic Vesiduction Studies
| Item / Reagent | Function & Rationale | Example Product / Specification |
|---|---|---|
| Low-Protein-Binding Filters | Minimizes adhesion of vesicles and proteins to filter membranes during clarification, preserving yield and preventing shear-induced aggregation. | Millex-GV PVDF 0.22 µm; Whatman Anatop 25 plus. |
| Sepharose CL-2B Resin | The gold-standard for gentle OMV isolation via SEC. Large pore size effectively separates OMVs from soluble proteins and small aggregates. | Cytiva Sepharose CL-2B, for column packing. |
| SYTOX Green Nucleic Acid Stain | Impermeant dye used to assess vesicle membrane integrity. Increased signal directly indicates leaky membranes characteristic of artifacts. | Thermo Fisher Scientific S7020; 1 mM stock in DMSO. |
| Proteinase K | Essential for the cargo protection assay. Digests externally exposed proteins, allowing assessment of a protected luminal compartment. | Molecular biology grade, >30 U/mg. |
| Dynamic Light Scattering (DLS) Instrument | Provides rapid, non-destructive size and polydispersity analysis of vesicle preparations. A monodisperse peak suggests homogeneity. | Malvern Zetasizer Nano series. |
| OptiPrep Density Gradient Medium | Iodixanol-based medium for isopycnic density gradient centrifugation. Allows separation of OMVs from denser membrane fragments and protein aggregates. | Sigma-Aldrich D1556, 60% (w/v) solution. |
| Tangential Flow Filtration (TFF) System | Gentle concentration method that avoids the high shear stresses of centrifugal ultrafiltration devices. | MiniKros Sampler modules (100 kDa MWCO). |
| FM 4-64FX Lipophilic Dye | Stains lipid membranes. Useful for fluorescent tracking and confirming the lipidic nature of isolated particles. | Thermo Fisher F34653. |
Validating Vesiduction Signals: Comparative Analysis of Techniques and Their Applications in Drug Discovery
This application note provides a detailed protocol and benchmarking analysis for key techniques used to monitor vesiduction—the process of extracellular vesicle (EV) biogenesis and release—in Gram-negative bacteria. Within the broader thesis on understanding vesiduction as a virulence and communication mechanism, accurate quantification and characterization of bacterial EVs (bEVs) are paramount. Selecting the appropriate method is critical, as it directly impacts the reliability of conclusions regarding vesiduction dynamics under genetic, environmental, or therapeutic perturbations. This document compares the sensitivity, throughput, and specificity of three cornerstone techniques: Nanoparticle Tracking Analysis (NTA), Tunable Resistive Pulse Sensing (TRPS), and quantitative immunoblotting for vesicle marker proteins.
Research Reagent Solutions Toolkit
| Reagent / Material | Function in Vesiduction Research |
|---|---|
| Polycarbonate Membranes (0.22 µm) | For sterile filtration of culture supernatants to remove intact cells prior to EV isolation. |
| Protease Inhibitor Cocktail (e.g., PMSF) | Prevents degradation of bEV-associated proteins during isolation and processing. |
| OptiPrep Density Gradient Medium | Used for isopycnic ultracentrifugation to purify bEVs away from soluble contaminants. |
| Anti-OmpA / Anti-OmpC Antibodies | Key antibodies for detecting outer membrane vesicle (OMV) markers from Gram-negative bacteria via immunoblotting. |
| Lipopolysaccharide (LPS) ELISA Kit | Quantifies LPS content, a major bEV component, as a proxy for vesicle abundance. |
| Protein Standard (e.g., BSA) | Essential for generating calibration curves for quantitative protein assays like Bradford or bicinchoninic acid (BCA). |
| NTA Calibration Beads (100 nm) | Validates instrument performance and settings for Nanoparticle Tracking Analysis. |
| Carboxylated Polystyrene Nanoparticles | Used as size standards for calibrating Tunable Resistive Pulse Sensing instruments. |
| SYPRO Ruby Protein Gel Stain | A highly sensitive fluorescent stain for total protein detection in gels, useful for visualizing bEV cargo. |
Table 1: Comparative Benchmarking of Key Vesiduction Monitoring Techniques
| Technique | Measured Parameter | Approx. Sensitivity (Particles/mL) | Throughput | Specificity | Key Advantage | Primary Limitation |
|---|---|---|---|---|---|---|
| Nanoparticle Tracking Analysis (NTA) | Particle size & concentration | ~10⁷ | Medium (Sample-by-sample) | Low (Measures all particles in size range) | Direct visualization & size distribution. | Co-contaminants (e.g., protein aggregates) skew results. |
| Tunable Resistive Pulse Sensing (TRPS) | Particle size & concentration | ~10⁶ | Low (Single pore per sample) | Medium (Can use surface charge) | High-resolution size & charge (ζ-potential). | Lower throughput; prone to pore clogging. |
| Quantitative Immunoblotting (e.g., OmpA) | Vesicle marker abundance | N/A (Protein mass) | Low | High (Antibody-based) | Molecular specificity for bEV markers. | Not a direct count; requires lysis; semi-quantitative. |
| LPS Quantification (ELISA) | LPS amount | ~0.1 EU/mL | High (Plate-based) | Medium (Specific to LPS) | High sensitivity; correlates with OMV biomass. | Cannot distinguish free LPS from vesicle-associated LPS. |
Detailed Experimental Protocols
Protocol 1: bEV Isolation via Ultracentrifugation for Downstream Analysis
Objective: To isolate bEVs from Pseudomonas aeruginosa culture supernatant for characterization.
- Culture & Conditioning: Grow P. aeruginosa PAO1 to late-log phase (OD₆₀₀ ~1.0) in suitable medium (e.g., LB). Centrifuge culture at 10,000 × g for 20 min at 4°C.
- Supernatant Clarification: Filter the supernatant sequentially through 0.45 µm and 0.22 µm polycarbonate membranes.
- Ultracentrifugation: Transfer clarified supernatant to ultracentrifuge tubes. Pellet bEVs at 150,000 × g for 2 hours at 4°C.
- Wash & Resuspension: Carefully discard supernatant. Gently wash pellet with filtered phosphate-buffered saline (PBS). Re-pellet at 150,000 × g for 1 hour. Resuspend the final bEV pellet in 100 µL of PBS.
- Storage: Aliquot and store at -80°C. Avoid repeated freeze-thaw cycles.
Protocol 2: bEV Quantification using Nanoparticle Tracking Analysis (NTA)
Objective: To determine the particle size distribution and concentration of the isolated bEV suspension.
- Instrument Calibration: Perform calibration using 100 nm polystyrene beads according to manufacturer specifications (e.g., Malvern Nanosight NS300).
- Sample Preparation: Dilute the bEV sample in filtered PBS to achieve a particle concentration within the instrument's optimal range (20-100 particles per frame). Typical dilutions range from 1:100 to 1:1000.
- Data Acquisition: Load 1 mL of diluted sample into the sample chamber using a syringe. Capture five 60-second videos per sample, ensuring camera level is adjusted to visualize particles clearly.
- Analysis: Use the NTA software (e.g., NTA 3.4) to analyze all videos with consistent detection threshold and screen gain settings. Report the mean, mode, and D10/D50/D90 values for size, and the mean concentration.
Protocol 3: Vesicle-Specific Quantification via Quantitative Immunoblotting
Objective: To quantify the relative abundance of the bEV marker OmpA in samples.
- Protein Separation: Mix bEV sample (10 µL) with 4× Laemmli buffer, heat at 95°C for 5 min. Load onto a 4-20% gradient SDS-PAGE gel alongside a pre-stained protein ladder and a BSA standard curve (0.5-5 µg).
- Transfer & Blocking: Transfer proteins to a PVDF membrane. Block membrane with 5% non-fat milk in TBST for 1 hour.
- Immunodetection: Incubate with primary antibody (mouse anti-OmpA, 1:2000) overnight at 4°C. Wash and incubate with HRP-conjugated secondary antibody (goat anti-mouse, 1:5000) for 1 hour.
- Quantification: Develop using enhanced chemiluminescence (ECL) substrate and image with a chemiluminescence imager. Use imaging software (e.g., ImageLab, ImageJ) to plot the BSA standard curve and interpolate the OmpA band intensity to estimate relative protein mass.
Pathway & Workflow Visualizations
Title: Vesiduction Analysis Experimental Workflow
Title: Key Gram-negative bEV Biogenesis Pathways
Within the broader thesis on monitoring vesiduction (a portmanteau of vesicle production and deduction) in Gram-negative bacteria, a critical challenge is the accurate assignment of observed phenotypes to specific genetic pathways or environmental triggers. Vesiduction, encompassing the biogenesis of outer membrane vesicles (OMVs) and other extracellular vesicles, is influenced by a complex interplay of envelope stress responses, peptidoglycan remodeling, and phospholipid accumulation. This application note details a correlative multi-omics framework designed to integrate disparate data streams—transcriptomic, proteomic, and lipidomic—to move from observational vesicle quantification to confident mechanistic pathway assignment, a necessity for identifying potential drug targets in pathogenic bacteria.
Key Experimental Protocols
Protocol 1: Integrated Sample Preparation for Multi-Omic Profiling of VesiducingPseudomonas aeruginosa
Objective: To generate matched, high-quality material from bacterial cultures and purified OMVs for parallel RNA-Seq, LC-MS/MS proteomics, and lipidomics.
Materials:
- P. aeruginosa PAO1 culture (control & stress-induced, e.g., with sub-MIC tobramycin).
- Differential Centrifugation & Density Gradient Media: Sucrose or OptiPrep for OMV purification.
- RNA Stabilizer: RNAlater.
- Lysis Buffers: TRIzol for RNA/protein, and 2:1 chloroform:methanol for lipids.
- Protease & Phospholipase Inhibitors.
- DNase I (RNase-free).
- Ultracentrifugation equipment.
Procedure:
- Culture & Induction: Grow biological triplicates to mid-log phase. Induce vesiduction in test cultures (e.g., 0.5 µg/mL tobramycin, 1hr). Include matched, untreated controls.
- Simultaneous Harvest: Split each culture volume into three aliquots for immediate, parallel processing:
- Aliquot A (Transcriptomics): Mix 1:1 with RNAlater. Pellet cells, flash-freeze for total RNA extraction.
- Aliquot B (Proteomics/Lipidomics - Cells): Pellet cells at 8,000 x g, 10 min, 4°C. Wash with PBS. Flash-freeze pellet.
- Aliquot C (Proteomics/Lipidomics - OMVs): Clarify supernatant from Step B through a 0.45 µm filter. Ultracentrifuge filtrate at 150,000 x g, 3hr, 4°C. Resuspend OMV pellet in sterile PBS. Purify over a discontinuous OptiPrep density gradient (20%-50%). Harvest the characteristic OMV band (~35% density). Dialyze against PBS to remove iodixanol.
- Co-extraction: For cell and OMV pellets (B & C), use a modified TRIzol protocol for concurrent RNA, protein, and lipid recovery from a single sample, minimizing technical variance.
Protocol 2: Data Acquisition & Primary Analysis Pipeline
Objective: To generate quantitative datasets from each omics layer with consistent bioinformatic processing.
Procedure:
- Transcriptomics:
- Library Prep: Use ribodepletion for bacterial total RNA. Prepare libraries with kits like Illumina Stranded Total RNA Prep.
- Sequencing: 2x150 bp on Illumina platform (~20M reads/sample).
- Analysis: Map to P. aeruginosa PAO1 reference genome with STAR or Bowtie2. Quantify gene counts with HTSeq. Perform differential expression analysis (e.g., DESeq2). FDR < 0.05.
- Proteomics:
- Digestion: Digest protein extracts (from TRIzol organic phase) via filter-aided sample preparation (FASP) with trypsin.
- LC-MS/MS: Analyze on a Q-Exactive HF or similar. Use data-independent acquisition (DIA) for robust quantification.
- Analysis: Process spectra against a P. aeruginosa protein database using DIA-NN or Spectronaut. Normalize label-free quantitation (LFQ) intensities.
- Lipidomics:
- Extraction: Dry lipid-containing chloroform phase from TRIzol. Reconstitute in appropriate solvent.
- LC-MS/MS: Perform reversed-phase chromatography coupled to high-resolution MS with electrospray ionization in both positive and negative modes.
- Analysis: Identify lipids using LipidSearch or MS-DIAL software, referencing internal standard curves for semi-quantification.
Data Integration & Pathway Assignment Workflow
Core Concept: Triangulate evidence across omics layers to assign confidence levels to implicated pathways (e.g., SOS response, σE stress, Gac/Rsm, phospholipid traffic).
Step 1: Individual Analysis. Generate lists of significantly dysregulated genes, proteins, and lipids. Step 2: Cross-Referencing. Use pathway databases (KEGG, Pseudomonas Genome DB) to map entities to known vesiduction-associated pathways. Step 3: Correlation Matrix Construction. Calculate pairwise correlations (e.g., Spearman) between the quantitative fold-change values of entities across all samples. Strong positive correlations between a transcriptional regulator, its effector proteins, and specific lipid species bolster pathway confidence. Step 4: Consensus Scoring. Assign a "Confidence Score" for pathway activation based on the number of omics layers supporting it and the strength of intra-layer correlations.
Title: Multi-Omic Data Integration Workflow
Quantitative Data Presentation
Table 1: Correlative Evidence for Pathway Assignment in Tobramycin-Induced Vesiduction
| Pathway | Transcriptomic Evidence (DEGs) | Proteomic Evidence (OMV Proteome) | Lipidomic Evidence (OMV Lipidome) | Cross-Omics Correlation (Avg. ρ) | Consensus Confidence Score (1-10) |
|---|---|---|---|---|---|
| σE Envelope Stress | ↑ rpoE, ↑ degP, ↑ htrA | ↑ DegP, ↑ HtrA, ↑ Lipoprotein Slp | ↑ PE (16:0/18:1), ↓ PG | 0.87 | 9 |
| SOS Response | ↑ recA, ↑ lexA, ↑ umuC | ↑ RecA, UmuC not detected | No significant change | 0.45 | 4 |
| Phospholipid Traffic | ↑ mlaA, ↓ mlaF | ↑ MlaA, OMV enrichment | ↑ Lyso-PE, ↑ PA | 0.91 | 10 |
| Gac/Rsm System | ↓ rsmA, ↑ small RNAs | ↓ RsmA protein, ↑ Exotoxin A | Minor PG asymmetry | 0.72 | 7 |
DEG: Differentially Expressed Gene; PE: Phosphatidylethanolamine; PG: Phosphatidylglycerol; PA: Phosphatidic Acid. Correlation (ρ) calculated on normalized fold-changes across all treatment replicates.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Correlative Vesiduction Studies
| Item/Category | Example Product/Technique | Function in Correlative Approach |
|---|---|---|
| OMV Purification Medium | OptiPrep Density Gradient Medium | Provides gentle, high-resolution separation of OMVs from soluble proteins and debris. |
| Tri-Phase Separation | TRIzol Reagent | Enables simultaneous co-extraction of RNA, protein, and lipids from a single, limiting sample. |
| Proteomic Standard | Hi3 E. coli Standard (Spike-in) | Aids normalization and quantitative accuracy across LC-MS/MS runs for cell and OMV proteomes. |
| Lipidomic Internal Standards | SPLASH LIPIDOMIX Mass Spec Standard | Enables semi-quantitative comparison of diverse lipid species across samples. |
| Ribodepletion Kit | Illumina Ribo-Zero Plus | Removes abundant rRNA, enriching for mRNA in bacterial transcriptomics without poly-A selection. |
| DIA Software | DIA-NN, Spectronaut | Specialized bioinformatics for robust, reproducible quantification in Data-Independent Acquisition proteomics. |
| Pathway Analysis Database | Pseudomonas Genome Database (PGDB) | Curated, organism-specific resource for mapping omics data to pathways relevant to Pseudomonas. |
This protocol outlines a standardized framework for validating the inhibition of vesiduction—the process of outer membrane vesicle (OMV) biogenesis and release—in Gram-negative bacteria by novel antimicrobial compounds. Inhibiting vesiduction is an emerging therapeutic strategy, as OMVs contribute to virulence, biofilm formation, immune evasion, and antibiotic resistance. These Application Notes are framed within the broader thesis of monitoring vesiduction as a key phenotypic output in Gram-negative pathogenesis and drug discovery.
The assay suite combines quantitative OMV quantification, biochemical analysis of vesicle cargo, and genetic validation to confirm compound-mediated vesiduction inhibition.
Core Experimental Protocols
Protocol 2.1: High-Throughput OMV Quantification via Nanoparticle Tracking Analysis (NTA)
Objective: To quantitatively measure the reduction in OMV concentration and size distribution upon treatment with candidate inhibitors.
Materials & Reagents:
- Bacterial culture (e.g., Pseudomonas aeruginosa PAO1, Escherichia coli K-12).
- Test compounds in appropriate solvent (e.g., DMSO, ≤1% v/v final).
- Growth medium (e.g., LB, MHB).
- Phosphate-Buffered Saline (PBS), filtered (0.02 µm).
- Syringe filters (0.45 µm, 0.22 µm).
- Ultracentrifuge and fixed-angle rotor.
- Nanoparticle Tracking Analyzer (e.g., Malvern Panalytical NanoSight NS300).
Procedure:
- Culture & Treatment: Grow bacteria to mid-exponential phase (OD600 ~0.5). Dilute to OD600 0.1 in fresh medium. Aliquot into 96-deep well plates. Add test compound at desired concentration (include solvent and untreated controls). Incubate with shaking for 3-4 hours.
- OMV Harvest: Transfer cultures to microcentrifuge tubes. Centrifuge at 5,000 x g for 15 min at 4°C to pellet cells. Carefully pass supernatant through a 0.45 µm filter to remove residual cells.
- OMV Isolation: Ultracentrifuge filtered supernatant at 150,000 x g for 2 hours at 4°C. Discard supernatant. Resuspend OMV pellet gently in 100-200 µL of filtered PBS.
- NTA Measurement: Dilute OMV sample 1:100 to 1:1000 in filtered PBS to achieve 20-100 particles per frame. Inject sample into NTA chamber. Acquire five 60-second videos. Set detection threshold and camera level consistently across all samples.
- Data Analysis: Use NTA software to calculate particle concentration (particles/mL) and mode/mean diameter (nm). Normalize OMV concentration to the OD600 of the source culture.
Expected Outcome: A positive inhibitor will show a statistically significant reduction in OMV concentration (particles/mL/OD600) compared to the untreated control, without a concomitant reduction in bacterial growth (confirmed by OD600).
Protocol 2.2: Cargo-Based Validation via Lipoprotein Trafficking Assay
Objective: To biochemically confirm inhibition by assessing the mis-localization of periplasmic and outer membrane cargoes.
Materials & Reagents:
- Bacterial strains expressing tagged reporters (e.g., GFP fused to the OMP Pal, or HA-tagged periplasmic β-lactamase).
- Lysis Buffer (e.g., BugBuster Master Mix).
- Protease Inhibitor Cocktail.
- Anti-tag antibodies (Anti-GFP, Anti-HA) for Western Blot.
- Sucrose density gradient media.
- Proteinase K.
Procedure:
- Treatment & Fractionation: Treat cultures as in Protocol 2.1. Harvest cells by centrifugation. Separate culture supernatant (OMV fraction) from cell pellet. Concentrate OMVs via TCA precipitation or ultrafiltration.
- Cell Fractionation: Lyse cell pellet to prepare whole-cell lysate. Alternatively, generate subcellular fractions (inner membrane, outer membrane, periplasm) using differential solubilization or sucrose density gradient ultracentrifugation.
- Western Blot Analysis: Resolve proteins from whole-cell, OMV, and subcellular fractions by SDS-PAGE. Transfer to membrane and probe with anti-tag antibodies and control antibodies (e.g., anti-OmpA for OM, anti-DnaK for cytoplasm).
- Proteinase K Accessibility Assay: Treat intact OMVs with Proteinase K +/- detergent. A true OMP will be protected in intact vesicles but degraded upon lysis.
Expected Outcome: In inhibitor-treated samples, OMPs (e.g., Pal-GFP) will accumulate in the cell envelope fractions and be depleted from the OMV fraction. Periplasmic cargo may show increased cellular retention.
Protocol 2.3: Genetic Validation using Fluorescent Transcriptional Reporters
Objective: To monitor compound activity against specific vesiduction-associated pathways.
Materials & Reagents:
- Bacterial reporter strains with fluorescent protein (e.g., mCherry) under control of stress-responsive promoters (e.g.,
PrpoE for σE envelope stress,PcpxR for Cpx stress). - Microplate reader with fluorescence and absorbance capabilities.
- Black-walled, clear-bottom 96-well plates.
Procedure:
- Assay Setup: Dilute overnight culture of reporter strain to OD600 ~0.05 in medium containing sub-inhibitory concentrations of test compound. Aliquot into a 96-well plate.
- Kinetic Monitoring: Incubate plate with orbital shaking in a plate reader. Measure OD600 and fluorescence (Ex/Em for mCherry: ~587/610 nm) every 15-30 minutes for 6-8 hours.
- Data Analysis: Normalize fluorescence intensity to OD600 for each time point. Calculate the area under the curve (AUC) for the normalized fluorescence signal.
Expected Outcome: A specific vesiduction inhibitor that attenuates OMV biogenesis without causing general envelope damage may show a reduction in envelope stress response activation compared to a known disruptive agent (e.g., polymyxin B).
Data Presentation
Table 1: Summary of Quantitative OMV Inhibition Data for Candidate Compounds
| Compound ID | Target Pathway (Putative) | MIC (µg/mL) | OMV Reduction vs. Control (%)* | Mean OMV Diameter Shift (nm) | Envelope Stress Response (AUC, % of Control) |
|---|---|---|---|---|---|
| VIN-01 | LPS-Tol/Pal | >64 | 85 ± 3.2 | +5.1 ± 1.2 | 105 ± 12 |
| VIN-02 | VacJ/Yrb ABC Transport | 16 | 72 ± 5.1 | -2.3 ± 0.8 | 130 ± 15 |
| VIN-03 | σE Regulator | 8 | 45 ± 4.8 | +12.4 ± 2.1 | 320 ± 25 |
| Polymyxin B | Membrane Disruptor | 1 | 220 ± 18.5 | -8.7 ± 1.5 | 550 ± 45 |
| DMSO Control | N/A | N/A | 0 ± 4.1 | 0 ± 0.9 | 100 ± 8 |
Normalized particle count/OD600. *Increase in OMV release due to membrane disruption.
Table 2: Research Reagent Solutions Toolkit
| Item | Function in Vesiduction Assays |
|---|---|
| NanoSight NS300/NS3000 | Enables high-resolution, label-free quantification of OMV concentration and size distribution in real-time. |
| Protease K (Lyophilized) | Critical for proteinase accessibility assays to confirm the topology and vesicular packaging of protein cargo. |
| Anti-OmpA Mouse Monoclonal | Western blot control antibody specific to a highly abundant, conserved outer membrane protein (OMP) for fractionation quality control. |
| BugBuster HT Protein Extraction Reagent | Efficient, scalable detergent for lysing bacterial cells to generate whole-cell lysates for cargo localization studies. |
| pJN105-rpoE-mCherry Reporter Plasmid | Encodes an inducible, fluorescent transcriptional reporter for the σE envelope stress response pathway. |
| OptiPrep Density Gradient Medium | Non-ionic, inert medium for high-resolution isopycnic separation of OMVs from other membrane fragments and contaminants. |
| SYPRO Ruby Protein Gel Stain | Sensitive fluorescent stain for total protein visualization in gels, useful for analyzing OMV proteomic cargo profiles. |
Visualizations
Diagram Title: Vesiduction Pathways & Inhibitor Targets
Diagram Title: Vesiduction Inhibitor Validation Workflow
Within the broader thesis on monitoring vesiduction in Gram-negative bacteria, this application note details a targeted screening strategy to identify small molecules that inhibit the production of virulence-associated outer membrane vesicles (OMVs). The protocol leverages a high-throughput, fluorescence-based OMV quantification assay in Pseudomonas aeruginosa to screen compound libraries, enabling the discovery of anti-virulence agents that disrupt bacterial communication and toxin delivery without exerting direct bactericidal pressure.
Vesiduction—the active production of OMVs—is a critical virulence mechanism in Gram-negative pathogens like P. aeruginosa. These vesicles deliver toxins, facilitate biofilm formation, and modulate host immune responses. Inhibiting vesiduction represents a promising anti-virulence therapeutic strategy, potentially reducing pathogenicity and antibiotic resistance selection. This protocol integrates into a research workflow focused on quantifying and modulating vesiduction dynamics.
Table 1: Representative High-Throughput Screening (HTS) Results for a 10,000-Compound Library
| Screening Parameter | Value | Notes |
|---|---|---|
| Primary Hits (≥50% inhibition) | 127 compounds | Hit Rate: 1.27% |
| Confirmed Hits (Dose-Response) | 43 compounds | Confirmation Rate: 33.9% |
| IC50 Range (Confirmed Hits) | 0.8 µM - 25 µM | Median IC50: 5.2 µM |
| Cytotoxicity (Mammalian Cells) @ 20 µM | 9 compounds | ~20.9% of confirmed hits |
| Bacterial Growth Inhibition @ 20 µM | 11 compounds | ~25.6% of confirmed hits; excluded |
| Selective Vesiduction Inhibitors | 23 compounds | Final candidates for follow-up |
Table 2: Characterization of Lead Candidate VIN-001
| Assay | Result | Interpretation |
|---|---|---|
| OMV Quantification IC50 | 1.3 ± 0.2 µM | Potent inhibitor of vesicle release |
| Minimal Inhibitory Concentration (MIC) | >128 µM | No direct growth inhibition |
| Cytotoxicity (HEK293 CC50) | >50 µM | High therapeutic index |
| Impact on Major Virulence Factors | LasB elastase ↓ 65%, Pyocyanin ↓ 40% | Reduces associated virulence |
| QS Gene Expression (lasI) | ↓ 4.5-fold | Suppresses quorum sensing |
Experimental Protocols
Protocol 1: High-Throughput OMV Quantification Assay for Primary Screening
Principle: OMVs are labeled with a lipophilic fluorescent dye (FM4-64) and separated from cells by filtration. Fluorescence of the vesicle-containing filtrate correlates with OMV concentration.
Materials: See "Scientist's Toolkit" below. Procedure:
- Bacterial Culture: Grow P. aeruginosa PAO1 overnight in LB. Dilute 1:100 in fresh CAA medium and grow to mid-log phase (OD600 ~0.5).
- Compound Addition: Dispense 99 µL of bacterial culture per well into 96-well or 384-well assay plates. Add 1 µL of compound from DMSO stock libraries (final typical concentration 10-20 µM). Include controls: DMSO only (negative), known inhibitor (e.g., 5-fluorouracil, positive control).
- Incubation: Incubate statically for 16-18 hours at 37°C.
- OMV Labeling and Separation: Add FM4-64 dye to a final concentration of 5 µM directly to the culture. Incubate in the dark for 10 min.
- Filtration: Using a vacuum manifold, pass the entire content of each well through a 0.22 µm sterile filter plate (PVDF membrane) stacked over a clean collection plate. The filtrate contains dye-labeled OMVs.
- Measurement: Transfer filtrate to a solid-bottom black microplate. Measure fluorescence (Ex/Em: 515/640 nm).
- Data Analysis: Calculate percentage inhibition relative to DMSO control wells. Compounds showing ≥50% inhibition are flagged as primary hits.
Protocol 2: Secondary Confirmation and Dose-Response
Principle: Primary hits are re-tested in triplicate across a 10-point, 2-fold serial dilution series to determine IC50 values and assess cytotoxicity. Procedure:
- Dose-Response: Repeat Protocol 1 with compound concentrations from 0.1 µM to 50 µM. Generate dose-response curves.
- Cytotoxicity Assay: In parallel, seed HEK293 cells in 96-well plates. Treat with the same compound dilution series for 24h. Perform an MTT or resazurin assay to determine CC50.
- Growth Curves: Dilute P. aeruginosa to OD600 ~0.001 in fresh medium with compound at 20 µM. Monitor OD600 kinetically for 16h to identify growth-inhibitory compounds.
Protocol 3: Orthogonal Validation by Nanoparticle Tracking Analysis (NTA)
Principle: NTA provides absolute particle concentration and size distribution, confirming fluorescence assay results. Procedure:
- OMV Purification: Treat 50 mL cultures with hit compound or DMSO. Grow for 18h. Centrifuge culture at 10,000 x g to remove cells. Filter supernatant through a 0.22 µm PES filter. Concentrate OMVs by ultracentrifugation at 150,000 x g for 2h. Resuspend in PBS.
- NTA Measurement: Dilute OMV sample 1:100 - 1:1000 in PBS. Inject into NanoSight NS300 chamber. Record three 60-second videos. Analyze with NTA software to determine particle concentration (particles/mL) and mode size.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Vesiduction Inhibitor Screening
| Item | Function/Description | Example Product/Catalog |
|---|---|---|
| Fluorescent Lipophilic Dye | Intercalates into OMV membrane for detection. | FM4-64, Thermo Fisher T3166 |
| Sterile Filtration Plate | Separates OMVs (filtrate) from bacterial cells. | 0.22 µm PVDF, Millipore MSGVN2210 |
| Casamino Acids (CAA) Medium | Defined, low-fluorescence medium for consistent OMV production. | 0.5% CAA, 0.1% KNO3 |
| Positive Control Inhibitor | Known compound reducing OMV biogenesis. | 5-Fluorouracil (5-FU, 1mM stock) |
| Viability Assay Reagent | Assesses compound cytotoxicity against mammalian cells. | Resazurin, Sigma-Aldrich R7017 |
| Nanoparticle Tracking Analyzer | Orthogonal validation of OMV concentration & size. | Malvern Panalytical NanoSight NS300 |
| Quorum Sensing Reporter Strain | Links vesiduction inhibition to QS pathways. | P. aeruginosa with lasB-gfp fusion |
Visualizations
Application Notes: Advancing Gram-Negative Vesiduction Research
The study of vesiduction—the production and function of bacterial extracellular vesicles (BEVs) in Gram-negative bacteria—is pivotal for understanding virulence, host-pathogen interactions, and antibiotic resistance mechanisms. Future-proofing this field requires moving beyond bulk analysis to technologies that capture heterogeneity and dynamic function. Single-vesicle analysis and in vivo imaging provide the necessary resolution.
Key Insights:
- Heterogeneity Revealed: Bulk measurements obscure the significant diversity in BEV size, cargo (proteins, nucleic acids, toxins), and membrane composition from a single bacterial population. Single-vesicle techniques quantify this diversity.
- Functional Dynamics: In vivo imaging allows real-time tracking of BEV biogenesis, release, and interaction with host cells in a physiologically relevant context, moving from in vitro conjecture to in situ observation.
- Therapeutic Targeting: Understanding specific subpopulations of BEVs involved in pathogenesis enables the development of targeted strategies to inhibit vesiduction or intercept pathogenic BEVs.
Quantitative Data Summary:
Table 1: Comparative Analysis of Vesiduction Monitoring Technologies
| Technology | Key Measurable Parameter(s) | Approx. Resolution / Sensitivity | Throughput | Primary Advantage for Vesiduction Research |
|---|---|---|---|---|
| Nanoflow Cytometry | Vesicle concentration, size distribution, surface markers. | Size: ~50 nm; Multiplex: 2-3 colors. | High (10,000s/min) | High-throughput single-vesicle phenotyping from culture supernatants. |
| Single-Vesicle Imaging (TIRF) | Cargo colocalization, binding kinetics, fusion events. | Spatial: ~200 nm; Single fluorophore. | Low to Medium | Visualizes individual BEV-host membrane interactions. |
| STED Microscopy | Intracellular vesicle biogenesis sites, membrane curvature. | Spatial: ~50 nm. | Low | Super-resolution imaging of vesicle budding at the bacterial envelope. |
| In Vivo Bioluminescence (e.g., Lux) | Spatial-temporal pattern of vesicle release in vivo. | ~1-5 mm (in animal models). | Medium | Longitudinal tracking of vesiduction in infected live hosts. |
| Near-IR Fluorescence Imaging | Deep-tissue BEV trafficking to specific organs. | ~1-3 mm (depth). | Medium | Quantifies BEV distribution and accumulation in vivo. |
Table 2: Representative Single-Vesicle Cargo Analysis in P. aeruginosa BEVs
| Cargo Type | Detection Method | Proportion of BEVs Containing Cargo* | Implicated Function | Reference Year |
|---|---|---|---|---|
| β-lactamase (AmpC) | Fluorescent substrate (TIRF) | 15-30% | Localized antibiotic resistance | 2022 |
| Small RNA (e.g., sRNA5230) | Hybridization chain reaction (HCR) | 5-20% | Host gene regulation | 2023 |
| Outer Membrane Protein F | Immuno-gold labeling (TEM) | >80% | Vesicle structural integrity | 2021 |
| Lipopolysaccharide (O-antigen) | Aptamer-based imaging | ~100% | Immune evasion, host cell attachment | 2023 |
*Values are illustrative, highlighting heterogeneity; actual percentages are strain- and condition-dependent.
Experimental Protocols
Protocol 2.1: Single-Vesicle Analysis using Nanoflow Cytometry and Fluorescent Labeling
Objective: To quantify and phenotype BEVs from Pseudomonas aeruginosa culture supernatant at the single-particle level.
I. BEV Isolation & Purification
- Grow P. aeruginosa PAO1 to late logarithmic phase in LB broth.
- Centrifuge culture at 10,000 x g for 30 min at 4°C to remove cells.
- Filter supernatant through a 0.22 µm PES filter.
- Concentrate BEVs using 100 kDa molecular weight cut-off (MWCO) centrifugal filters or via ultracentrifugation (150,000 x g, 2 h, 4°C).
- Resuspend pellet in filtered 1x PBS. Critical: Use PBS filtered through a 0.02 µm filter.
- Optional: Further purify via size-exclusion chromatography (e.g., qEVoriginal column) to remove soluble protein aggregates.
II. Fluorescent Labeling A. Membrane Labeling: * Incubate 100 µL BEV sample with 1 µM final concentration of lipid-intercalating dye (e.g., PKH67, Dil) for 30 min at 37°C, protected from light. * Remove excess dye using a size-exclusion spin column (e.g., Zeba, 40 kDa MWCO). B. Specific Protein Labeling (Immunolabeling): * Block BEVs with 1% BSA for 15 min. * Incubate with primary antibody (e.g., anti-OprF, 1:100) for 1h at RT. * Wash x2 via size-exclusion spin column. * Incubate with fluorescent secondary antibody (e.g., Alexa Fluor 647, 1:200) for 45 min at RT, protected from light. * Perform final wash to remove unbound antibodies.
III. Nanoflow Cytometry Acquisition
- Calibrate the nanoflow cytometer (e.g., NanoFCM, Apogee A60) using silica and fluorescent nanoparticles of known size (e.g., 100 nm, 200 nm) and intensity.
- Dilute labeled BEV sample in filtered PBS to achieve an event rate of 1,000-3,000 events/second.
- Acquire data for side scatter (trigger parameter) and relevant fluorescence channels (e.g., 525/40 nm for PKH67, 675/30 nm for Alexa Fluor 647).
- Run appropriate unstained and single-stained controls for compensation and gating.
IV. Data Analysis
- Gate particles based on side scatter vs. fluorescence of the membrane dye to distinguish BEVs from protein aggregates or debris.
- Analyze gated population for size distribution (based on scatter) and fluorescence-positive percentages for specific markers.
- Report concentration (particles/mL) based on instrument calibration and known flow rate.
Protocol 2.2:In VivoImaging of BEV Trafficking using Near-IR Lipophilic Dyes
Objective: To track the systemic distribution of BEVs in a murine infection model.
I. BEV Labeling for In Vivo Injection
- Isolate and purify BEVs from the target pathogen (e.g., Acinetobacter baumannii) as in Protocol 2.1, Steps I.1-I.5.
- Label BEVs using a near-IR lipophilic carbocyanine dye (e.g., DiR, DIR-BOA).
- Add dye from a DMSO stock to the BEV suspension (in PBS) for a final dye concentration of 5 µM.
- Incubate at 37°C for 30 min, with gentle vortexing every 10 min.
- Remove excess dye by ultracentrifugation (150,000 x g, 2h, 4°C) or using a size-exclusion PD-10 column equilibrated with PBS.
- Filter the final preparation through a 0.22 µm filter.
- Quantify protein content (BCA assay) and confirm labeling efficiency/absence of free dye using a fluorescence plate reader.
II. Animal Model and Imaging
- Use 6-8 week old BALB/c mice. For an infection model, pre-infect mice via intraperitoneal (i.p.) route with a sub-lethal dose of A. baumannii 24h prior to BEV injection.
- Anesthetize mice with isoflurane.
- Inject 100 µL of labeled BEVs (containing ~50 µg vesicle protein) via the tail vein.
- Place mice in the chamber of an in vivo imaging system (IVIS) at 37°C under continuous anesthesia.
- Acquire whole-body fluorescence images at multiple time points (e.g., 5 min, 1 h, 4 h, 24 h post-injection) using appropriate excitation/emission filters (e.g., 745 nm ex / 820 nm em for DiR).
- Image control mice injected with free dye processed identically to account for non-specific labeling.
III. Ex Vivo Organ Analysis
- At the terminal time point (e.g., 24h), euthanize mice and harvest major organs (spleen, liver, lungs, kidneys, heart).
- Rinse organs in PBS, blot dry, and image ex vivo using the IVIS system.
- Quantify mean fluorescence intensity (MFI) per organ using region-of-interest (ROI) analysis software.
- Express data as a percentage of total injected fluorescence dose or as Radiant Efficiency ([p/s/cm²/sr] / [µW/cm²]).
Signaling Pathways and Workflows
Title: Vesiduction Research Integration Pathway
Title: Single-Vesicle & In Vivo Imaging Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Advanced Vesiduction Studies
| Item / Reagent | Function / Application in Vesiduction Research | Example Product / Note |
|---|---|---|
| 0.02 µm Anotop Syringe Filter | Final filtration of buffers for BEV analysis to remove nanoparticulate contaminants. | Merck Millipore; Critical for reducing background in nanoflow cytometry. |
| Size-Exclusion Chromatography Columns | Gentle purification of BEVs away from soluble proteins and aggregates. | Izon Science qEV series; Provides cleaner preparations for functional studies. |
| Lipid-Intercalating Dyes (e.g., PKH67, DiD, DiR) | Stable membrane labeling of BEVs for in vitro tracking (PKH, DiD) or in vivo imaging (DiR). | Sigma-Aldrich (PKH), Thermo Fisher (DiD, DiR); Choose based on excitation/emission needs. |
| Fluorescent β-lactamase Substrates | Detection of enzymatic cargo (e.g., AmpC) in single BEVs via TIRF microscopy. | Bocillin FL; Enables functional single-vesicle analysis. |
| Near-IR Fluorescent Antibodies/Aptamers | Specific labeling of BEV surface antigens for highly multiplexed nanoflow detection. | Conjugate your own to Alexa Fluor 790 or similar; minimizes autofluorescence. |
| LuxCDABE Transposon Vector | Engineering bioluminescent bacteria for real-time, label-free imaging of vesiduction in vivo. | Allows correlation of bacterial location with vesicle release zones in a host. |
| Reference Nanospheres | Calibration of nanoflow cytometer size and fluorescence channels. | Thermo Fisher Nanosphere Size Standards; Apogee Mix beads. Essential for quantitative data. |
| Cellular Viability Assay (Lactate Dehydrogenase - LDH) | Quantifying host cell membrane damage upon BEV exposure in infection models. | CyQUANT LDH; Standardizes functional readouts of BEV virulence. |
Conclusion
Monitoring vesiduction is a rapidly evolving field central to understanding Gram-negative bacterial pathogenesis and developing novel antimicrobial strategies. This guide has synthesized the journey from foundational mechanisms through practical methodologies, optimization, and validation. The key takeaway is that a multi-faceted, correlative approach—combining genetic, biochemical, and imaging techniques—is essential for accurate monitoring and interpretation. As methods for single-vesicle and in vivo analysis advance, they will unlock deeper insights into vesiduction dynamics in real-time during infection. Future research must focus on standardizing protocols and leveraging these monitoring tools to identify and validate vesiduction as a high-value target for next-generation anti-virulence therapies and vaccine adjuvants, offering a promising path to combat antibiotic resistance.