RND Efflux Pumps: The Formidable Gatekeepers of Gram-Negative Bacterial Resistance
This article provides a comprehensive analysis of Resistance-Nodulation-Division (RND) efflux pumps, a primary mechanism of multidrug resistance in Gram-negative pathogens.
RND Efflux Pumps: The Formidable Gatekeepers of Gram-Negative Bacterial Resistance
Abstract
This article provides a comprehensive analysis of Resistance-Nodulation-Division (RND) efflux pumps, a primary mechanism of multidrug resistance in Gram-negative pathogens. Tailored for researchers and drug development professionals, it explores the foundational biology and structure of RND pumps, details current methodological approaches for their study and inhibition, addresses common experimental challenges, and validates findings through comparative analysis with other resistance mechanisms. The synthesis aims to inform the ongoing development of efflux pump inhibitors and novel therapeutic strategies to combat antimicrobial resistance.
Decoding RND Efflux Pumps: Structure, Function, and Genetic Regulation
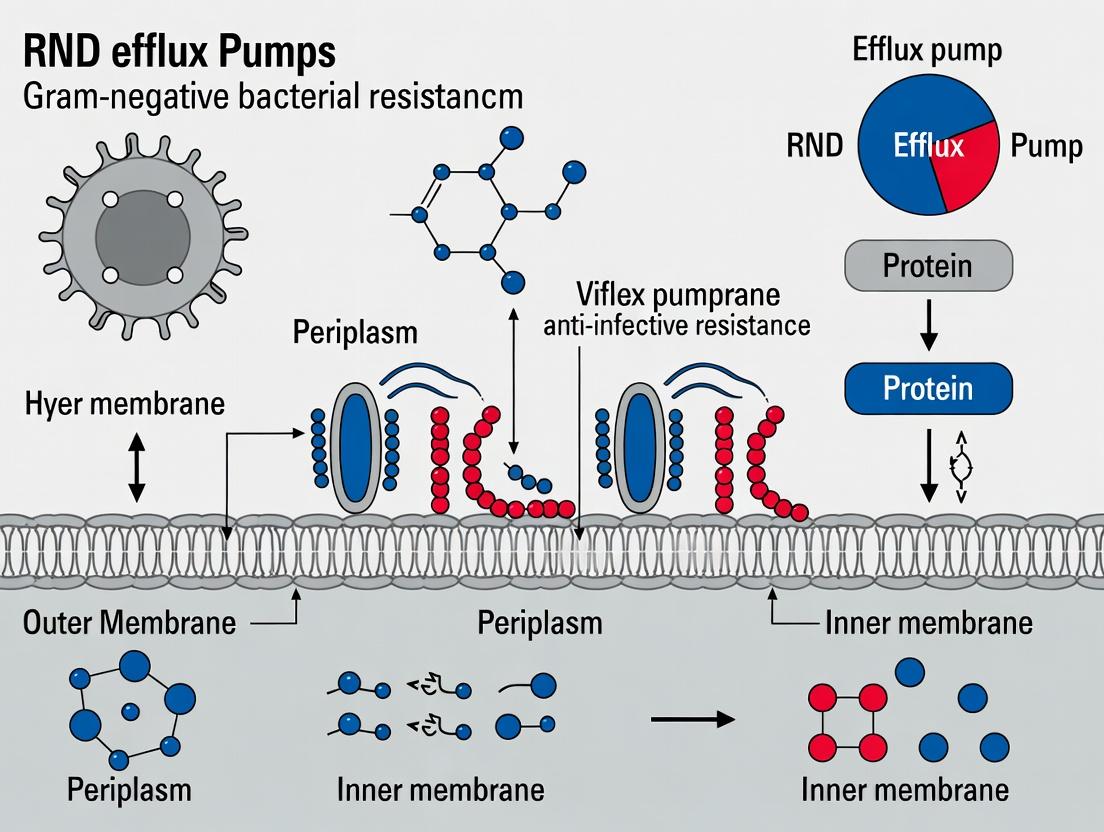
The Resistance-Nodulation-Division (RND) superfamily of efflux pumps constitutes a primary molecular defense system in Gram-negative bacteria. These tripartite, proton-motive-force-driven complexes are fundamental to both intrinsic and acquired multidrug resistance (MDR), extruding a vast array of structurally unrelated antimicrobials, biocides, and host-derived compounds. Their activity, often coupled with low outer membrane permeability, renders many Gram-negative pathogens recalcitrant to treatment. Understanding their structure, regulation, and function is a central pillar of contemporary research aimed at combating antimicrobial resistance (AMR).
Structural Architecture and Mechanism
RND pumps are organized as three-component assemblies spanning the entire cell envelope:
- Inner Membrane RND Transporter: The engine of the complex (e.g., AcrB in E. coli, MexB in P. aeruginosa). It binds substrates from the periplasm or inner leaflet and undergoes a conformational cycle to propel them outward.
- Periplasmic Membrane Fusion Protein (MFP): Bridges the RND transporter to the outer membrane channel (e.g., AcrA, MexA).
- Outer Membrane Factor (OMF): Forms a channel in the outer membrane (e.g., TolC, OprM) for final substrate exit.
The complex functions via a proton antiport mechanism, where the influx of protons down their gradient provides the energy for substrate efflux.
Diagram: Tripartite RND Efflux Pump Assembly
Major RND Pumps in Pathogenic Bacteria
Table 1: Clinically Significant RND Efflux Systems
| Organism | Primary RND System | Core Components (RND/MFP/OMF) | Key Substrate Profile | Regulatory Protein(s) |
|---|---|---|---|---|
| Escherichia coli | AcrAB-TolC | AcrB / AcrA / TolC | β-lactams, FQs, tetracyclines, chloramphenicol, bile salts | AcrR, MarA, SoxS, Rob, RamA |
| Pseudomonas aeruginosa | MexAB-OprM | MexB / MexA / OprM | β-lactams, FQs, chloramphenicol, trimethoprim, novobiocin | MexR, NalC, NalD, NaID |
| Pseudomonas aeruginosa | MexXY-OprM | MexY / MexX / OprM | Aminoglycosides, FQs, tetracyclines, erythromycin | MexZ, ArmZ, PA5471 |
| Acinetobacter baumannii | AdeABC | AdeB / AdeA / AdeC | Aminoglycosides, tetracyclines, tigecycline, β-lactams | AdeRS (Two-Component System) |
| Klebsiella pneumoniae | AcrAB-TolC | AcrB / AcrA / TolC | Similar to E. coli | RamA, MarA, RarA |
| Neisseria gonorrhoeae | MtrCDE | MtrD / MtrC / MtrE | β-lactams, macrolides, rifampin, host fatty acids | MtrR |
FQs = Fluoroquinolones
Regulation of Expression
RND pump expression is tightly controlled by local and global regulators, often in response to environmental stress (e.g., antibiotics, host defenses). Dysregulation through mutation is a common pathway to hyper-resistance.
Diagram: Regulatory Network for Major RND Pumps
Quantitative Impact on Resistance
Table 2: Effect of RND Efflux Pump Overexpression on Minimum Inhibitory Concentrations (MICs)
| Antibiotic Class | Wild-type P. aeruginosa MIC (μg/mL) | mexAB-oprM Overexpression MIC (μg/mL) | Fold Increase |
|---|---|---|---|
| Piperacillin | 2 - 4 | 64 - 128 | 32x |
| Levofloxacin | 0.5 - 1 | 8 - 16 | 16x |
| Chloramphenicol | 32 - 64 | 512 - 1024 | 16x |
| Tetracycline | 4 - 8 | 128 - 256 | 32x |
| Trimethoprim | 32 - 64 | 512 - 1024 | 16x |
| Antibiotic Class | Wild-type A. baumannii MIC (μg/mL) | adeABC Overexpression MIC (μg/mL) | Fold Increase |
|---|---|---|---|
| Tobramycin | 1 - 2 | 16 - 32 | 16x |
| Tigecycline | 0.25 - 0.5 | 4 - 8 | 16x |
| Ciprofloxacin | 0.12 - 0.25 | 2 - 4 | 16x |
| Ceftazidime | 2 - 4 | 32 - 64 | 16x |
Core Experimental Protocols
Protocol: Ethidium Bromide Accumulation Assay (Functional Efflux Activity)
Principle: Measures real-time intracellular accumulation of a fluorescent efflux pump substrate (e.g., EtBr) in the presence/absence of an efflux pump inhibitor (EPI) like carbonyl cyanide m-chlorophenyl hydrazone (CCCP) or Phe-Arg-β-naphthylamide (PAβN).
Methodology:
- Cell Preparation: Grow test and control strains (e.g., knockout mutant) to mid-log phase. Harvest, wash, and resuspend in buffer (e.g., PBS or minimal medium) to an OD~600~ of ~0.4.
- Fluorometer Setup: Pre-warm cell suspension to 37°C with continuous stirring in a fluorometer cuvette. Set excitation/emission wavelengths (e.g., 530 nm/600 nm for EtBr).
- Baseline Recording: Record background fluorescence for 60 seconds.
- Energy Poisoning: Add the protonophore CCCP (final conc. 50 μM) to inhibit proton motive force-dependent efflux. Incubate for 10 minutes.
- Substrate Addition: Add EtBr (final conc. 0.5-2 μg/mL). Fluorescence will increase as EtBr enters and accumulates in de-energized cells. Monitor until plateau (~5-10 min).
- Energy Restoration: Add glucose (final conc. 0.4%) to restore proton motive force. The active efflux in wild-type cells will cause a rapid decrease in fluorescence, while efflux-deficient mutants will show little decrease.
- Data Analysis: Calculate initial efflux rates from the slope after glucose addition. Compare plateau fluorescence levels between strains.
Protocol: RT-qPCR Analysis of RND Pump Gene Expression
Principle: Quantifies mRNA levels of RND pump genes to correlate overexpression with resistant phenotypes.
Methodology:
- RNA Isolation: Treat bacterial cultures with antibiotic sub-MIC or other inducer vs. control. Harvest cells. Extract total RNA using a kit with DNase I treatment. Verify integrity (RIN > 9.0) and purity (A~260~/A~280~ ~2.0).
- cDNA Synthesis: Use 500 ng - 1 μg total RNA and a reverse transcription kit with random hexamers.
- qPCR Reaction Setup: Prepare triplicate reactions containing: cDNA template (1-10 ng equivalent), forward/reverse gene-specific primers (200 nM each), SYBR Green master mix. Include no-template controls.
- Cycling Conditions: 95°C for 3 min; 40 cycles of 95°C for 10 sec, 60°C for 30 sec (acquire fluorescence); followed by a melt curve analysis.
- Data Analysis: Determine Cq values. Normalize target gene Cq to one or more stable reference genes (rpoD, gyrB). Calculate relative expression using the 2^(-ΔΔCq) method.
Diagram: RT-qPCR Workflow for RND Expression
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for RND Efflux Research
| Reagent/Category | Specific Example(s) | Function in Research |
|---|---|---|
| Efflux Pump Inhibitors (EPIs) | Phe-Arg-β-naphthylamide (PAβN), 1-(1-naphthylmethyl)-piperazine (NMP), carbonyl cyanide m-chlorophenyl hydrazone (CCCP) | Functional studies: To confirm efflux-mediated resistance by potentiating antibiotic activity. Mechanistic studies: To probe energy coupling (CCCP). |
| Fluorescent Efflux Substrates | Ethidium bromide (EtBr), Hoechst 33342, Nile Red, N-phenyl-1-naphthylamine (NPN) | Accumulation/efflux assays: Direct measurement of pump activity using fluorometry, flow cytometry, or microscopy. |
| Antibiotic Susceptibility Panels | Custom broth microdilution plates with gradients of key antibiotics (β-lactams, FQs, AGs, etc.) with/without fixed EPI concentration. | High-throughput screening of strain collections for efflux-mediated resistance phenotypes (e.g., checkerboard assays). |
| Gene Expression Kits | RNAprotect, RNeasy kits, DNase I, High-Capacity cDNA Reverse Transcription kits, SYBR Green qPCR master mixes. | Quantifying mRNA levels of RND pump and regulatory genes (RT-qPCR) from bacterial cultures under stress. |
| Polyclonal/Monoclonal Antibodies | Anti-AcrB, Anti-MexB, Anti-TolC, Anti-MexR, etc. | Western blotting: To assess protein expression levels and confirm knockout/overexpression strains. |
| Molecular Cloning & Mutagenesis Kits | Site-directed mutagenesis kits, Gibson Assembly, suicide vectors for gene knockouts (pKNG101, pEX18Tc). | Construction of isogenic mutant strains (deletions, point mutations in regulators) to establish causality. |
| Crystallography Reagents | Detergents (DDM, LMNG), lipids, cryo-protectants. | For purifying and stabilizing the membrane protein complexes for structural studies (X-ray, Cryo-EM). |
The tripartite Resistance-Nodulation-Division (RND) efflux pump, exemplified by the archetypal AcrAB-TolC system in Escherichia coli, is a cornerstone of intrinsic and acquired multidrug resistance in Gram-negative bacteria. Within the broader thesis on RND-mediated resistance, understanding the precise architectural and functional interplay between its three components—the inner membrane RND transporter (e.g., AcrB), the periplasmic adaptor protein (e.g., AcrA), and the outer membrane channel (e.g., TolC)—is paramount for the rational design of efflux pump inhibitors (EPIs). This guide details the structural and functional blueprint of this molecular machine.
Core Components and Quantitative Comparison
The tripartite assembly spans the entire Gram-negative cell envelope. The following table summarizes key structural and functional data for the canonical E. coli AcrAB-TolC system and selected homologs from high-priority pathogens.
Table 1: Quantitative Comparison of Prototypical RND Tripartite Efflux Pumps
| Component & Organism | Protein Name (Family) | Gene Locus / PDB ID (Example) | Size (aa / kDa) | Key Structural Features | Known Substrates (Number) |
|---|---|---|---|---|---|
| Inner Membrane Pump | |||||
| Escherichia coli | AcrB (RND) | acrB / 4DX5 | 1049 aa / 114 kDa | Trimer; Proton antiporter; 12 TM helices; Large periplasmic domain | β-lactams, FQs, tetracycline, dyes, detergents (>50) |
| Pseudomonas aeruginosa | MexB (RND) | mexB / 3W9I | 1046 aa / 113 kDa | Trimer; High structural homology to AcrB | Aminoglycosides, FQs, chloramphenicol, β-lactams |
| Neisseria gonorrhoeae | MtrD (RND) | mtrD / 5V5S | 1099 aa / 119 kDa | Trimer; Extended substrate binding pocket | β-lactams, FQs, macrolides, biocides |
| Periplasmic Adaptor | |||||
| Escherichia coli | AcrA (MF) | acrA / 5NG5 | 397 aa / 42 kDa | Lipoyl domain; β-barrel domain; coiled-coil hairpin | N/A (Structural role) |
| Pseudomonas aeruginosa | MexA (MF) | mexA / 1VF7 | 358 aa / 38 kDa | Similar modular architecture to AcrA | N/A (Structural role) |
| Acinetobacter baumannii | AdeA (MF) | adeA / 5J8A | 370 aa / 40 kDa | Essential for AdeABC assembly | N/A (Structural role) |
| Outer Membrane Channel | |||||
| Escherichia coli | TolC | tolC / 1EK9 | 493 aa / 53 kDa | Trimer; α-β barrel; 12-stranded β-barrel; ~140 Å long | N/A (Conduit) |
| Pseudomonas aeruginosa | OprM | oprM / 3D5K | 470 aa / 50 kDa | Trimer; Structurally homologous to TolC | N/A (Conduit) |
| Salmonella enterica | TolC | tolC / 2VDD | 493 aa / 53 kDa | Near-identical to E. coli TolC | N/A (Conduit) |
Abbreviations: RND (Resistance-Nodulation-Division); MF (Membrane Fusion protein); FQs (Fluoroquinolones); TM (Transmembrane); PDB (Protein Data Bank).
Architectural Assembly and Functional Cycle
The assembly is driven by ordered, affinity-dependent interactions. The adaptor (AcrA) bridges the high-affinity interaction between the RND pump (AcrB) and the outer membrane channel (TolC), forming a contiguous conduit ~300 Å long.
Diagram 1: Tripartite Assembly and Drug Export Pathway
The functional cycle involves a coordinated, proton motive force-driven process:
- Substrate Binding: Drugs from the cytoplasm or inner membrane leaflet bind to AcrB's proximal binding pocket.
- Proton Motive Force Influx: Influx of protons (H+) through AcrB's transmembrane domain provides energy.
- Conformational Cycling: AcrB undergoes a peristaltic conformational cycle (Access → Binding → Extrusion) within its trimer.
- Adaptor-Mediated Conduit Opening: AcrA transmits conformational changes, stabilizing the open state of TolC.
- Extrusion: The substrate is propelled through the assembled channel into the external medium.
Key Experimental Protocols
Protocol: Co-Immunoprecipitation (Co-IP) for Verifying Tripartite Interactions
Objective: To confirm in vivo physical interaction between AcrB, AcrA, and TolC. Reagents:
- E. coli strain expressing tagged (e.g., FLAG, His) AcrB and untagged AcrA/TolC.
- Lysis Buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1% DDM, protease inhibitor cocktail.
- Anti-Tag Magnetic Beads.
- Wash Buffer: Lysis buffer with 0.1% DDM.
- Elution Buffer: Wash buffer with 250 mM imidazole or 3x FLAG peptide.
Procedure:
- Grow bacterial culture to mid-log phase (OD600 ~0.6).
- Harvest cells, resuspend in Lysis Buffer, and lyse by sonication.
- Centrifuge at 15,000 x g for 10 min to remove debris. Isolate membrane fraction via ultracentrifugation (100,000 x g, 1 h).
- Solubilize membrane pellet in Lysis Buffer for 2 h at 4°C.
- Incubate solubilized proteins with Anti-Tag Beads for 2 h at 4°C.
- Wash beads 5x with 1 mL Wash Buffer.
- Elute bound proteins with Elution Buffer.
- Analyze eluate by SDS-PAGE and Western blotting using antibodies against AcrA, TolC, and the tag.
Protocol: Real-Time Efflux Assay Using Fluorescent Dyes (e.g., Ethidium Bromide)
Objective: To measure functional efflux pump activity in live cells. Reagents:
- Bacterial strain (wild-type and efflux pump knockout, e.g., ΔacrB).
- Efflux Assay Buffer: 50 mM PBS (pH 7.0), 5 mM MgCl2.
- Ethidium Bromide (EtBr) stock solution (10 mg/mL).
- Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) stock (50 mM in DMSO), a protonophore.
- Microplate reader with fluorescence capabilities (Ex/Em: 530/600 nm).
Procedure:
- Grow bacteria, wash, and resuspend in Assay Buffer to OD600 ~0.4.
- Load cells with EtBr (final conc. 2 µg/mL) in the presence of CCCP (final conc. 100 µM) for 30 min at 37°C to deplete energy and allow dye accumulation.
- Wash cells 3x with ice-cold Assay Buffer to remove CCCP and external dye.
- Rapidly resuspend pellets in pre-warmed Assay Buffer (± an EPI as control) in a 96-well plate.
- Immediately measure fluorescence every 30 sec for 15 min.
- Data Analysis: Plot fluorescence vs. time. The initial rate of fluorescence decrease after resuspension (due to active efflux) is calculated. Knockout strains or EPI-treated samples show a significantly slower decrease.
Diagram 2: Real-Time Efflux Assay Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for RND Efflux Pump Research
| Reagent / Material | Function & Application | Example / Supplier Note |
|---|---|---|
| EPI Candidates | Inhibit pump function; used in synergy assays. | Phenylalanine-arginine β-naphthylamide (PAβN), 1-(1-naphthylmethyl)-piperazine (NMP). |
| Fluorescent Substrate Dyes | Visualize and quantify efflux activity. | Ethidium Bromide (EtBr), Hoechst 33342, Nile Red. |
| Protease Inhibitor Cocktail | Preserve protein complexes during lysis for Co-IP. | EDTA-free for metal-dependent proteases. |
| Detergents for Membrane Protein Solubilization | Solubilize membrane proteins while preserving native interactions. | n-Dodecyl-β-D-maltoside (DDM), Lauryl Maltose Neopentyl Glycol (LMNG). |
| Anti-Tag Antibody Beads | Immunoprecipitation of tagged pump components. | Anti-FLAG M2 Magnetic Beads, Ni-NTA Superflow Resin (for His-tag). |
| Protonophores (e.g., CCCP) | Deplete proton motive force to control efflux in functional assays. | Used in dye accumulation/efflux assays. |
| Crystallography Screen Kits | Identify conditions for structural studies of components/complexes. | Commercial sparse matrix screens (e.g., from Hampton Research). |
| Liposome Preparation Kits | Create defined membranes for in vitro reconstitution of transport activity. | Used to study proton-coupled transport by purified AcrB. |
Resistance-Nodulation-Division (RND) efflux pumps are central to multidrug resistance (MDR) in Gram-negative bacteria. These tripartite complexes, exemplified by E. coli AcrAB-TolC and P. aeruginosa MexAB-OprM, span the inner membrane, periplasm, and outer membrane. The "Hydrophobic Vacuum Cleaner" (HVC) model is a pivotal mechanistic framework describing how these pumps recognize and transport a vast array of lipophilic and amphiphilic substrates directly from the inner membrane or periplasm, expelling them into the external medium.
This whitepaper provides an in-depth technical analysis of the HVC model, detailing the substrate translocation pathway, supporting experimental evidence, and its critical implications for overcoming efflux-mediated resistance in drug development.
Core Mechanism of the Hydrophobic Vacuum Cleaner Model
The HVC model posits that substrates are captured from the lipid bilayer itself. The process involves:
- Substrate Partitioning: Hydrophobic and amphiphilic antibiotics (e.g., fluoroquinolones, β-lactams, macrolides) partition into the inner membrane lipid bilayer.
- Lateral Access & Binding: Substrates diffuse laterally within the membrane to access the transmembrane domain (TMD) of the RND pump (e.g., AcrB). High-affinity binding occurs in deep, conformationally-changing hydrophobic pockets within the TMDs.
- Proton Motive Force-Driven Transport: Binding triggers a conformational change from the Loose (L) to Tight (T) state. Proton influx from the periplasm, driven by the proton motive force (PMF), powers a subsequent rotation to the Open (O) state, expelling the substrate into a central funnel.
- Funnel-Mediated Extrusion: The substrate is directed into the periplasmic adaptor protein (e.g., AcrA) and through the outer membrane channel (e.g., TolC), culminating in extrusion.
This mechanism distinguishes RND pumps from other families that typically capture substrates from the cytoplasm or periplasmic space.
Key Experimental Evidence & Quantitative Data
| Experimental Approach | Key Finding | Supporting HVC Model Aspect | Reference (Example) |
|---|---|---|---|
| X-ray Crystallography of AcrB | Structures of AcrB with substrates (doxorubicin, minocycline) bound in transmembrane pockets. | Direct visualization of substrate binding from the membrane interior. | Murakami et al., Nature (2006) |
| Molecular Dynamics (MD) Simulations | Substrates (e.g., erythromycin) partition into bilayer and enter AcrB via lateral access portals. | Energetic feasibility of lateral substrate entry from the membrane. | Schulz et al., J Mol Biol (2010) |
| Kinetic Flux Assays | Efflux kinetics correlate with substrate membrane partition coefficients (logP). | Substrate transport efficiency depends on lipid bilayer partitioning. | Li et al., PNAS (2015) |
| FRET-based Binding Studies | Binding affinity of AcrB for substrates is higher in membrane-embedded vs. solubilized protein. | Membrane environment is crucial for optimal substrate recognition. | Pos et al., FEBS Lett (2004) |
Table 2: Quantitative Parameters for Model Substrates in HVC Transport
| Substrate | LogP (Octanol-Water) | Primary Binding Site in AcrB (Crystal Data) | Relative Efflux Efficiency* |
|---|---|---|---|
| Doxorubicin | ~1.3 | Distal (Deep) Pocket in TMD | High (0.95) |
| Minocycline | ~0.3 | Proximal Pocket / Access Pocket | High (0.90) |
| Erythromycin | ~3.1 | Access Pocket (via lateral gate) | Moderate (0.75) |
| Ciprofloxacin | ~0.7 | Distal Pocket / Central Cavity | High (0.88) |
| Rhodamine 6G | ~3.5 | Distal Pocket | Very High (1.00) |
| Normalized to Rhodamine 6G efflux rate in *E. coli overproducing AcrAB-TolC. |
Detailed Experimental Protocols
Protocol 1: Determining Substrate-Membrane Partitioning Correlation to Efflux
Objective: Quantify the relationship between a compound's hydrophobicity (LogP) and its susceptibility to RND-mediated efflux. Materials:
- Isogenic bacterial strains: Wild-type vs. RND pump knockout (e.g., E. coli AG100 vs. AG100ΔacrB).
- Test antibiotics spanning a range of LogP values.
- Mueller-Hinton Broth (MHB).
- 96-well microtiter plates.
- Plate reader (OD600). Procedure:
- Prepare serial two-fold dilutions of each antibiotic in MHB across a 96-well plate.
- Inoculate each well with ~5x10^5 CFU/mL of either the wild-type or efflux-deficient strain.
- Incubate plates at 37°C for 18-24 hours without shaking.
- Determine the Minimum Inhibitory Concentration (MIC) as the lowest concentration inhibiting visible growth (OD600).
- Calculate the Efflux Pump Contribution (EPC) for each drug: EPC = (MICWT / MICKO).
- Plot Log(EPC) against the known LogP of each drug. A positive correlation supports the HVC model.
Protocol 2: Site-Directed Mutagenesis of Lateral Access Channels
Objective: Functionally validate the role of specific residues in proposed membrane-accessible substrate entry portals. Materials:
- Plasmid encoding the RND transporter (e.g., acrB gene in pET vector).
- Site-directed mutagenesis kit.
- E. coli expression host and efflux-deficient background strain.
- Primers for introducing point mutations (e.g., F666A, Q124L). Procedure:
- Design mutagenic primers to alter key hydrophobic residues in putative access portals (e.g., the "Trap" region between TM1/TM2).
- Perform PCR-based mutagenesis following kit protocols to generate mutant acrB plasmids.
- Sequence the entire gene to confirm the desired mutation and absence of secondary mutations.
- Transform both wild-type and mutant acrB plasmids into an efflux-deficient strain (ΔacrB).
- Perform MIC assays (as in Protocol 1) for a panel of substrates. A significant decrease in the MIC ratio for specific substrates in the mutant vs. wild-type complemented strain indicates impaired substrate capture via that portal.
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Research Reagent Solutions
| Reagent/Material | Function in HVC Model Research | Example Product/Note |
|---|---|---|
| Phe-Arg β-naphthylamide (PAβN) | Broad-spectrum efflux pump inhibitor (EPI); used to chemically validate efflux involvement by potentiating antibiotic activity. | Non-antibiotic substrate competitive inhibitor. |
| Ethidium Bromide (EtBr) Accumulation Assay Kit | Fluorescent efflux substrate; used in real-time fluorometric assays to measure basal and inhibited pump activity in whole cells. | Measures fluorescence increase when efflux is inhibited. |
| Proteoliposome Reconstitution Kit | For incorporating purified RND proteins (e.g., AcrB) into artificial lipid bilayers. Enables study of transport in a controlled membrane environment. | Essential for in vitro validation of PMF-driven, membrane-based transport. |
| NBD-labeled Phospholipids | Fluorescent lipid analogs; used to probe potential perturbation of membrane integrity or lipid flip-flop by RND pump activity. | e.g., NBD-PE. |
| Isogenic Efflux Pump Knockout Strains | Genetically defined controls (e.g., Keio collection for E. coli) to isolate the contribution of a specific RND pump to resistance. | Critical baseline for all phenotypic assays. |
| Molecular Dynamics Simulation Software (e.g., GROMACS) | To model and simulate the interaction of substrates with the lipid bilayer and the lateral access portals of the pump protein. | Provides atomic-level mechanistic insights. |
Visualizations
Diagram Title: Hydrophobic Vacuum Cleaner Mechanism Steps
Diagram Title: HVC Model Experimental Validation Workflow
Within the context of Gram-negative bacterial resistance, Resistance-Nodulation-Division (RND) efflux pumps like AcrAB-TolC are critical determinants of multidrug resistance (MDR). Their expression is tightly controlled by a sophisticated hierarchical regulatory network. This network integrates local, specific repression with global, pleiotropic activation in response to environmental stresses and chemical inducers. Understanding this genomic context is paramount for developing strategies to inhibit efflux and restore antibiotic efficacy.
Regulatory Architecture
Local Repressors: AcrR
The acrAB operon is constitutively expressed at low basal levels, primarily due to repression by the local transcriptional regulator AcrR. AcrR, a TetR-family repressor, binds as a dimer to an operator sequence upstream of the acrAB promoter, sterically hindering RNA polymerase access.
Global Regulators: The MarA/Rob/SoxS/RamA Regulon
Global response regulators activate a broad set of genes involved in stress response, including acrAB. These belong to the AraC/XylS family (except Rob, which is structurally distinct but functionally overlaps).
- MarA (Multiple antibiotic resistance A): Activated by the MarRAB system in response to salicylates, weak acids, and antibiotics.
- RamA (Resistance to antimicrobials A): A key regulator in Klebsiella pneumoniae and Enterobacter spp., often associated with clinical MDR. Its expression is controlled by upstream regulators like RamR.
- SoxS (Superoxide response S): Induced via the SoxRS system in response to redox-cycling compounds and superoxide stress.
- Rob (Right origin binding protein): Constitutively expressed but activated by binding to bile salts, dipyridyl, and fatty acids.
These activators bind to a conserved sequence known as the "marbox" in the promoter regions of target genes, recruiting RNA polymerase.
Quantitative Data on Regulator Impact
Table 1: Impact of Regulatory Mutations on Antibiotic MICs and Pump Expression
| Regulator (System) | Common Inducers/Activation Signals | Typical Fold-Increase in acrAB/acrEF Expression (vs. Wild-Type) | Representative Effect on Antibiotic MIC (e.g., Ciprofloxacin) | Primary Organisms Studied |
|---|---|---|---|---|
| AcrR Loss-of-Function | N/A (constitutive derepression) | 2 - 5 fold | 2 - 4 fold increase | E. coli, S. enterica |
| MarA Overexpression | Salicylate, Tetracycline, Menadione | 10 - 50 fold | 4 - 32 fold increase | E. coli |
| RamA Overexpression | Unknown signals, often ramR mutations | 20 - 100+ fold | 8 - 64 fold increase | K. pneumoniae, E. cloacae |
| SoxS Overexpression | Paraquat, Menadione | 10 - 30 fold | 4 - 16 fold increase | E. coli, S. enterica |
| Rob Activation | Bile Salts, Decanoate | 5 - 20 fold | 2 - 8 fold increase | E. coli |
Table 2: Binding Affinity of Global Regulators to the acrAB Promoter
| Regulator | Consensus Binding Site ("Marbox") | Estimated Kd (nM) for acrAB Promoter | Reference (Example) |
|---|---|---|---|
| MarA | 5'-AYnGCACnWnnRYYAAAnY-3' (W=A/T, R=A/G, Y=C/T) | 50 - 200 nM | (Martin et al., 2002) |
| SoxS | Similar to MarA box | 100 - 300 nM | (Li & Demple, 1994) |
| Rob | Similar to MarA box | 200 - 500 nM (enhanced by inducers) | (Bennik et al., 2000) |
Key Experimental Protocols
Electrophoretic Mobility Shift Assay (EMSA) for Regulator-Promoter Binding
Purpose: To confirm direct binding of a purified regulator (e.g., His₆-MarA) to the acrAB promoter DNA. Detailed Protocol:
- DNA Probe Preparation: Amplify a ~200-300 bp fragment containing the acrAB promoter region by PCR. Label it with Cy5 or DIG at the 5' end. Prepare an unlabeled identical fragment for competition assays.
- Protein Purification: Express and purify the recombinant regulator protein using affinity chromatography (e.g., Ni-NTA for His-tagged protein).
- Binding Reaction: In a 20 µL volume, mix:
- Labeled DNA probe (10-20 fmol)
- Purified protein (0, 10, 50, 100, 200 nM)
- Binding Buffer (20 mM HEPES, pH 7.9, 50 mM KCl, 1 mM DTT, 0.1 mg/mL BSA, 5% Glycerol, 50 µg/mL poly(dI-dC) as nonspecific competitor)
- Incubate at 25°C for 30 min.
- Competition Control: Include reactions with a 50-100x molar excess of unlabeled specific probe or nonspecific DNA.
- Electrophoresis: Load reactions onto a pre-run 6% native polyacrylamide gel in 0.5x TBE buffer. Run at 100 V for 60-90 min at 4°C.
- Detection: Visualize using a fluorescence imager (for Cy5) or chemiluminescence (for DIG).
β-Galactosidase Reporter Assay for Promoter Activity
Purpose: To quantify the transcriptional activation/repression of the acrAB promoter under different regulatory conditions. Detailed Protocol:
- Strain Construction: Fuse the acrAB promoter region to a promoterless lacZ gene on a single-copy plasmid or chromosomal locus. Introduce this reporter into wild-type and regulator mutant/overexpression backgrounds.
- Culture Conditions: Grow bacterial strains to mid-log phase (OD₆₀₀ ~0.5) in the presence or absence of inducers (e.g., 5 mM salicylate for Mar).
- Assay: Perform the Miller assay.
- Take 100 µL of culture, add 900 µL of Z-buffer (60 mM Na₂HPO₄, 40 mM NaH₂PO₄, 10 mM KCl, 1 mM MgSO₄, pH 7.0).
- Add 50 µL of chloroform and 25 µL of 0.1% SDS. Vortex for 10 sec to permeabilize cells.
- Start reaction with 200 µL of 4 mg/mL ONPG (in Z-buffer). Incubate at 28°C until yellow color develops.
- Stop reaction with 500 µL of 1 M Na₂CO₃. Record reaction time.
- Measure OD₄₂₀ (hydrolyzed ONP), OD₅₅₀ (cell debris), and OD₆₀₀ of the original culture.
- Calculation: Miller Units = 1000 * [OD₄₂₀ - (1.75 * OD₅₅₀)] / [time (min) * volume (mL) * OD₆₀₀].
Diagrams of Regulatory Pathways
Title: Global Regulator Activation Pathways for RND Pumps
Title: Hierarchical Local and Global Regulation of acrAB
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Studying RND Pump Regulation
| Reagent / Material | Function / Application | Example Supplier / Catalog |
|---|---|---|
| pET-28a(+) Vector | For recombinant overexpression and purification of His-tagged regulator proteins (MarA, SoxS, Rob, AcrR). | Novagen (Merck) |
| Ni-NTA Superflow Agarose | Affinity chromatography resin for purifying polyhistidine-tagged proteins. | Qiagen |
| DIG Gel Shift Kit, 2nd Generation | For non-radioactive labeling and detection of DNA in EMSA experiments. | Roche, #03353591910 |
| ONPG (o-Nitrophenyl-β-D-galactopyranoside) | Colorimetric substrate for β-galactosidase in reporter gene assays. | Sigma-Aldrich, N1127 |
| Salicylic Acid Sodium Salt | Prototypical inducer of the MarRA system. | Sigma-Aldrich, S3007 |
| Paraquat Dichloride | Inducer of the SoxRS system (generates superoxide). | Sigma-Aldrich, 856177 |
| Sodium Cholate/Deoxycholate | Bile salt activators of Rob. | Sigma-Aldrich, C6445 / D6750 |
| Poly(dI-dC) | Non-specific competitor DNA for reducing background in EMSA. | Sigma-Aldrich, P4929 |
| Cy5 Maleimide Mono-reactive Dye | For fluorescent labeling of oligonucleotides or proteins. | Cytiva, PA25031 |
| Q5 High-Fidelity DNA Polymerase | For high-fidelity PCR amplification of promoter fragments and cloning. | NEB, M0491 |
Within the paradigm of Gram-negative bacterial resistance, Resistance-Nodulation-Division (RND) efflux pumps constitute a primary defense mechanism. Their broad substrate specificity underpins multidrug resistance (MDR) and presents a formidable challenge in drug development. This technical guide details the spectrum of antimicrobials extruded by major RND pumps, focusing on Escherichia coli's AcrAB-TolC, Pseudomonas aeruginosa's MexAB-OprM, and related systems, which are central to current research. The functional promiscuity of these tripartite systems allows for the efflux of a structurally diverse array of compounds, directly contributing to intrinsic and acquired resistance.
Major RND Pumps and Their Substrate Profiles
The substrate specificity, while broad, varies significantly between pumps and bacterial species. The following table synthesizes current data on extruded agents.
Table 1: Substrate Profiles of Primary RND Efflux Pumps in Model Gram-negative Pathogens
| Antimicrobial Class | Specific Agent(s) | AcrAB-TolC (E. coli) | MexAB-OprM (P. aeruginosa) | MexXY-OprM (P. aeruginosa) | AdeABC (A. baumannii) |
|---|---|---|---|---|---|
| β-Lactams | Penicillins (e.g., ampicillin) | + | ++ | - | + |
| Cephalosporins (e.g., cefepime) | + | ++ | + | ++ | |
| Carbapenems (e.g., meropenem) | +/- | + (except imipenem) | - | ++ | |
| Fluoroquinolones | Ciprofloxacin, Levofloxacin | ++ | ++ | ++ | ++ |
| Tetracyclines | Tetracycline, Doxycycline | ++ | + | ++ | ++ |
| Aminoglycosides | Gentamicin, Tobramycin | - | - | ++ | + |
| Macrolides | Erythromycin, Azithromycin | ++ | ++ | ++ | + |
| Chloramphenicol | Chloramphenicol | ++ | ++ | + | + |
| Glycylcyclines | Tigecycline | + | +/- | + | ++ |
| Biocides | Triclosan | ++ | + | - | + |
| Benzalkonium Chloride | ++ | ++ | + | ++ | |
| Chlorhexidine | + | + | +/- | ++ | |
| Dyes | Ethidium Bromide, Acriflavine | ++ | ++ | + | ++ |
| Solvents | Hexane, Cyclohexane | ++ | + | ND | ND |
Key: ++ = Primary substrate, high-level contribution to resistance; += Substrate, moderate contribution; +/- = Weak substrate or variable data; -= Not a substrate; ND = No sufficient data.
Core Experimental Protocols for Characterizing Substrate Extrusion
Defining a compound as an efflux pump substrate requires a multi-pronged experimental approach.
Minimum Inhibitory Concentration (MIC) Shift Assay
Purpose: To determine the contribution of an efflux system to resistance against a specific agent. Protocol:
- Prepare Mueller-Hinton broth (MHB) in a 96-well microtiter plate.
- Perform serial two-fold dilutions of the antimicrobial agent.
- Inoculate wells with a standardized bacterial suspension (~5 x 10^5 CFU/mL) of two isogenic strains:
- Wild-type strain.
- Efflux pump knockout mutant (e.g., ΔacrB, ΔmexB).
- Include a control well with bacterial inoculum but no antibiotic.
- Incubate at 35°C for 16-20 hours.
- Determine the MIC as the lowest concentration inhibiting visible growth.
- A ≥4-fold decrease in MIC for the mutant strain compared to the wild-type is indicative of the compound being an efflux substrate.
Ethidium Bromide (EtBr) Accumulation Assay
Purpose: A fluorescence-based functional assay to measure real-time efflux activity, often using EtBr as a model substrate. Protocol:
- Grow bacterial cultures to mid-log phase (OD600 ~0.5).
- Harvest cells, wash, and resuspend in buffer (e.g., PBS or HEPES) with glucose as an energy source.
- Load cells with EtBr (0.5-2 µg/mL) in the presence of an efflux pump inhibitor (e.g., Phe-Arg-β-naphthylamide, PABN) to inhibit active efflux and allow dye accumulation. Incubate for 20-30 min.
- Centrifuge, wash, and resuspend cells in fresh buffer without inhibitor to initiate active efflux.
- Immediately transfer suspension to a quartz cuvette or microplate reader.
- Monitor fluorescence (excitation 530 nm, emission 585-600 nm) over time (5-10 min).
- The initial rate of fluorescence decrease is proportional to efflux pump activity. Mutants or inhibitor-treated cells show slower fluorescence decay.
Real-time RT-qPCR for Pump Gene Expression
Purpose: To correlate increased antimicrobial resistance with elevated expression of efflux pump genes. Protocol:
- Treat bacterial cultures with a sub-inhibitory concentration of the test antibiotic/biocide for a defined period.
- Stabilize RNA using a reagent like RNAprotect. Extract total RNA using a commercial kit.
- Treat RNA with DNase I to remove genomic DNA contamination.
- Synthesize cDNA using random hexamers and reverse transcriptase.
- Design gene-specific primers for the target RND pump gene (e.g., acrB, mexB) and housekeeping genes (e.g., rpoD, gyrB).
- Perform qPCR with SYBR Green chemistry. Include no-template controls.
- Analyze data using the comparative ΔΔCt method to determine fold-change in gene expression relative to an untreated control, normalized to housekeeping genes.
Visualization of Regulatory Networks and Experimental Workflow
Global and Local Regulation of RND Pump Expression
Workflow for Validating an Efflux Pump Substrate
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Efflux Substrate Characterization Studies
| Reagent / Material | Function & Application | Key Considerations |
|---|---|---|
| Phe-Arg-β-naphthylamide (PABN) | Broad-spectrum efflux pump inhibitor. Used in MIC shift and accumulation assays to chemically inhibit RND pumps and confirm substrate status. | Membrane-permeable peptide; can have off-target effects at high concentrations; use appropriate solvent controls (DMSO). |
| Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) | Protonophore that dissipates the proton motive force (PMF). Used to distinguish active efflux (PMF-dependent) from passive diffusion in accumulation assays. | Highly toxic; prepare fresh stock solutions; control for general metabolic inhibition. |
| Ethidium Bromide (EtBr) | Fluorescent model substrate and DNA intercalator. Standard compound for measuring efflux kinetics in real-time fluorometric assays. | Carcinogen; requires safe handling and disposal. Alternative dyes: Hoechst 33342, Nile Red. |
| SYBR Green / TaqMan Probes | For RT-qPCR quantification of efflux pump gene expression levels. Essential for linking resistance phenotype to regulatory changes. | Design primers to avoid genomic DNA amplification; validate primer efficiency; normalize to stable housekeeping genes. |
| Cation-Adjusted Mueller-Hinton Broth (CAMHB) | Standardized medium for MIC determination. Ensures reproducibility and comparability of susceptibility data. | Follow CLSI/EUCAST guidelines for preparation and inoculation. |
| Efflux Pump Knockout Mutant Strains | Isogenic bacterial strains with deletions in specific RND pump genes (e.g., ΔacrB, ΔmexB). Critical controls for defining pump-specific substrates. | Obtain from reputable strain collections (e.g., KEIO collection for E. coli). Check for secondary mutations. |
| H-3 or C-14 Radiolabeled Antibiotics | Used in direct transport assays to measure the active extrusion of specific substrates, providing definitive proof. | Requires specialized licensing, facilities, and safety protocols for handling radioisotopes. |
The Role of RND Pumps in Biofilm Formation, Virulence, and Quorum Sensing
Resistance-Nodulation-Division (RND) efflux pumps are tripartite macromolecular complexes in Gram-negative bacteria, central to multidrug resistance (MDR). Beyond their canonical role in antibiotic expulsion, these systems—such as AcrAB-TolC in Escherichia coli, MexAB-OprM in Pseudomonas aeruginosa, and CmeABC in Campylobacter jejuni—are critical, pleiotropic regulators of microbial physiology. This whitepaper, framed within the broader thesis of RND pumps as master modulators of bacterial pathogenicity, delineates their integral functions in biofilm development, virulence factor expression, and quorum sensing (QS) circuitry. These contributions underscore RND pumps as high-value targets for anti-infective strategies aimed at disabling both resistance and pathogenicity.
Core Mechanistic Links: Efflux Beyond Drug Resistance
RND pumps influence virulence by modulating the intracellular concentrations of small molecule signals, metabolites, and toxins. Their substrates often include:
- Autoinducers (AIs): Signaling molecules for QS (e.g., acyl-homoserine lactones, AI-2).
- Virulence Precursors/Effectors: Molecules involved in toxin production, secretion, or regulation.
- Metabolic Byproducts: Waste products whose accumulation can inhibit growth or biofilm integrity. By actively extruding these compounds, RND pumps directly and indirectly tune gene expression programs governing communal behavior and host interaction.
Quantitative Impact of RND Pump Activity on Virulence Phenotypes
Recent studies quantify the impact of RND pump deletion or inhibition on key pathogenicity metrics.
Table 1: Impact of RND Pump Deletion on Virulence Parameters in Model Pathogens
| Bacterial Species | RND Pump System | Biofilm Biomass Reduction (%) | Virulence Factor Reduction (e.g., Toxin) | In Vivo Virulence Attenuation (Model) | Key Reference (Year) |
|---|---|---|---|---|---|
| Pseudomonas aeruginosa | MexAB-OprM | 40-60% | Pyocyanin (70-80%), Exotoxin A (50%) | 75% increased survival (Galleria mellonella) | Li et al. (2023) |
| Escherichia coli (UPEC) | AcrAB-TolC | 55-70% | Alpha-hemolysin (65%) | 2-log CFU reduction (murine UTI) | Wang & Wang (2024) |
| Salmonella Typhimurium | AcrAB-TolC | 45-55% | SPI-1 Effectors (60%) | Reduced systemic spread (mouse) | Chen et al. (2023) |
| Campylobacter jejuni | CmeABC | 50-65% | Cytolethal distending toxin (55%) | Colonization reduced 100-fold (chicken) | Garcia et al. (2024) |
| Acinetobacter baumannii | AdeABC | 60-75% | Biofilm-associated protein (Bap) (70%) | 80% reduced mortality (nematode) | Park et al. (2023) |
Experimental Protocols for Investigating RND-Virulence Links
Protocol: Static Biofilm Crystal Violet Assay with Efflux Inhibitor
Objective: To quantify the role of a specific RND pump in early biofilm formation. Materials: Wild-type and RND pump knockout strains; sterile 96-well polystyrene plate; appropriate growth medium; efflux pump inhibitor (e.g., Phenylalanine-arginine beta-naphthylamide, PAβN); 0.1% crystal violet (CV) solution; 33% glacial acetic acid; plate reader.
- Inoculation: Dilute overnight cultures to OD600 ~0.05 in medium ± sub-inhibitory concentration of PAβN (e.g., 20 µg/mL). Aliquot 200 µL per well (8 replicates per condition).
- Incubation: Incubate statically at 37°C for 24-48h.
- Staining: Gently remove planktonic cells by washing wells 3x with PBS. Air-dry. Add 200 µL 0.1% CV to each well, stain for 15 min.
- Destaining & Quantification: Wash thoroughly to remove unbound CV. Add 200 µL 33% acetic acid to solubilize bound CV. Measure OD570 of the eluate. Analysis: Compare average OD570 of inhibitor-treated/knockout vs. wild-type controls. Statistical significance is typically determined via Student's t-test.
Protocol: Quantification of Quorum Sensing Signal Production/Secretion
Objective: To measure the effect of RND efflux on autoinducer accumulation. Materials: Biosensor strains (e.g., Chromobacterium violaceum CV026 for short-chain AHLs; Vibrio harveyi BB170 for AI-2); ethyl acetate; acidified ethyl acetate; LB medium; luminescence/spectrophotometer.
- Sample Preparation: Grow test strains (wild-type and knockout) to late exponential phase. Centrifuge culture, filter-sterilize (0.22 µm) the supernatant.
- Signal Extraction:
- AHLs: Mix supernatant with equal volume ethyl acetate, vortex, separate organic phase, evaporate. Resuspend in LB.
- AI-2: Use cell-free supernatant directly.
- Bioassay: For AHLs, add resuspended sample to mid-log phase CV026, incubate 24h, measure violacein production (OD585). For AI-2, mix sample with BB170 reporter, monitor bioluminescence over time. Analysis: Compare signal induction in samples from knockout vs. wild-type strains.
Visualizing Signaling and Regulatory Networks
Diagram 1: RND Pump Regulation of QS and Virulence
Diagram 2: Biofilm Quantification Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Studying RND Pumps in Virulence
| Reagent / Material | Primary Function in Research | Example Product / Specification |
|---|---|---|
| PAβN (Phe-Arg-β-naphthylamide) | Broad-spectrum RND pump inhibitor used to chemically phenocopy pump deletion in wild-type strains. | Sigma-Aldrich, Phenylarginine β-naphthylamide dihydrochloride, ≥95% (HPLC). |
| Crystal Violet | Histological dye that binds polysaccharides and nucleic acids in the biofilm matrix, enabling colorimetric quantification. | 0.1% aqueous solution, filter-sterilized. |
| AHL Biosensor Strains | Reporter organisms used to detect and quantify specific classes of quorum sensing autoinducers in culture supernatants. | Chromobacterium violaceum CV026 (for C4-C8 AHLs); Agrobacterium tumefaciens A136 (for broad-range AHLs). |
| AI-2 Bioluminescence Reporter | Engineered strain that produces light in response to the universal signal Autoinducer-2 (AI-2). | Vibrio harveyi BB170 (luxN mutant). |
| Polystyrene Microtiter Plates | Non-treated, sterile plates for consistent, high-throughput biofilm formation assays. | Corning 96-well flat-bottom, cell culture-treated plates. |
| Specific RND Pump Inhibitors | Targeted compounds to dissect the role of individual pump systems (e.g., MexAB-OprM vs. MexXY-OprM). | D13-9001: Potent, specific inhibitor of MexAB-OprM in P. aeruginosa. |
RND efflux pumps are established as critical nodes integrating antibiotic resistance with virulence and social behavior in Gram-negative pathogens. Targeting these pumps with next-generation efflux pump inhibitors (EPIs) offers a dual therapeutic strategy: sensitizing bacteria to conventional antibiotics while simultaneously attenuating their pathogenic potential by disrupting biofilm formation and QS. Future research must focus on elucidating system-specific regulatory nuances and developing pathogen-selective EPIs to minimize microbiome disruption, paving the way for novel combination therapies in the fight against MDR infections.
Experimental Strategies: From Efflux Phenotype Detection to Inhibitor Development
Within the context of a thesis on Resistance-Nodulation-Division (RND) efflux pumps in Gram-negative bacteria, this whitepaper details two cornerstone phenotypic assays. These methods are critical for identifying and characterizing multidrug resistance (MDR) mediated by active efflux, a primary research and therapeutic challenge. The Ethidium Bromide (EtBr) Agar Cartwheel assay serves as a rapid, semi-quantitative screen for efflux pump activity. The Minimum Inhibitory Concentration (MIC) reduction assay using Efflux Pump Inhibitors (EPIs) like PAβN provides quantitative, confirmatory evidence of efflux-mediated resistance and a measure of inhibition potency.
The Role of Phenotypic Assays in Efflux Pump Research
RND efflux pumps, such as AcrAB-TolC in Escherichia coli and MexAB-OprM in Pseudomonas aeruginosa, are major contributors to intrinsic and acquired MDR. Genotypic detection of pump genes is insufficient to confirm functional, overexpressed activity. Phenotypic assays bridge this gap by directly measuring the efflux function and its contribution to the resistant phenotype, guiding subsequent genetic and biochemical investigations.
Ethidium Bromide Agar Cartwheel Assay
Principle
EtBr is a fluorescent substrate for many RND pumps. Bacterial strains with upregulated efflux activity will expel EtBr more efficiently, preventing its intracellular accumulation and subsequent DNA intercalation. On agar plates containing a sub-inhibitory concentration of EtBr, strains with high efflux activity grow at higher EtBr concentrations than susceptible strains, visualized as extended growth.
Detailed Protocol
Materials:
- Muller-Hinton Agar (MHA)
- Ethidium Bromide stock solution (10 mg/mL in water, stored in dark)
- Bacterial overnight broths adjusted to 0.5 McFarland standard (~1.5 x 10^8 CFU/mL)
- Sterile swabs
- Petri dishes
Procedure:
- Prepare MHA and autoclave. Cool to approximately 50°C.
- Aseptically add EtBr stock to the cooled agar to achieve a final concentration gradient series: 0, 0.5, 1.0, 1.5, 2.0 µg/mL. Pour into plates.
- Let plates solidify and dry briefly.
- Using a sterile swab, create a bacterial lawn from the center of the plate outwards in three consecutive strokes (forming a "cartwheel" pattern). Typically, 4-6 strains can be tested on one plate, divided into sectors.
- Incubate plates aerobically at 35°C ± 2°C for 16-20 hours.
- Interpretation: Measure the distance of growth from the center for each strain at each EtBr concentration. Strains with functional, hyperactive efflux will show growth at higher concentrations (e.g., 2.0 µg/mL) compared to control strains.
Data Presentation
Table 1: Sample EtBr Cartwheel Assay Results
| Bacterial Strain | Known Efflux Pump Status | Growth at EtBr 0.5 µg/mL | Growth at EtBr 1.0 µg/mL | Growth at EtBr 2.0 µg/mL | Interpretation |
|---|---|---|---|---|---|
| E. coli ATCC 25922 | Wild-type (basal expression) | + | + | - | Baseline efflux |
| E. coli Clinical Isolate 1 | Suspected AcrB overexpression | + | + | + | High efflux activity |
| P. aeruginosa PAO1 | Wild-type | + | + | +/- | Moderate baseline efflux |
| P. aeruginosa MDR Isolate | Suspected MexB overexpression | + | + | + | High efflux activity |
| E. coli ΔacrB | Efflux pump knockout | + | - | - | Efflux deficient |
Diagram: EtBr Cartwheel Assay Workflow & Interpretation
MIC Reduction Assay with EPIs (e.g., PAβN)
Principle
PAβN is a broad-spectrum EPI that competitively inhibits RND pumps. If an antibiotic's resistance is mediated by efflux, co-administration with PAβN will reduce the antibiotic's MIC. A significant reduction (typically ≥4-fold) confirms the contribution of active efflux to the resistant phenotype.
Detailed Protocol (Broth Microdilution)
Materials:
- Cation-adjusted Mueller-Hinton Broth (CA-MHB)
- Antibiotic(s) of interest (e.g., ciprofloxacin, levofloxacin, erythromycin)
- PAβN dihydrochloride stock solution (e.g., 25 mg/mL in water, filter-sterilized)
- Sterile 96-well microtiter plates
- Bacterial inoculum at 5 x 10^5 CFU/mL final concentration
Procedure:
- Prepare a 2X working solution of PAβN in CA-MHB at double the desired final test concentration (typically 20–50 µg/mL final, so 40–100 µg/mL 2X stock).
- Perform standard broth microdilution for the test antibiotic in two parallel plates:
- Plate A (Antibiotic Only): Serial two-fold dilutions of antibiotic in CA-MHB.
- Plate B (Antibiotic + EPI): Serial two-fold dilutions of antibiotic in CA-MHB containing the 2X PAβN working solution.
- Add an equal volume of standardized bacterial inoculum to all wells. Final volume is 100µL/well. Include growth control (no drug) and sterility control wells.
- Incubate at 35°C for 16-20 hours.
- Determine the MIC as the lowest concentration that completely inhibits visible growth.
- Calculate the MIC fold reduction: MIC (Antibiotic Alone) / MIC (Antibiotic + PAβN).
Data Presentation
Table 2: Sample MIC Reduction Data with PAβN (EPI)
| Bacterial Strain | Antibiotic | MIC Alone (µg/mL) | MIC + PAβN (µg/mL) | Fold Reduction | Interpretation |
|---|---|---|---|---|---|
| K. pneumoniae MDR-1 | Ciprofloxacin | 32 | 4 | 8 | Efflux contributes to resistance |
| K. pneumoniae MDR-1 | Meropenem | 8 | 8 | 1 | Resistance not efflux-mediated |
| E. coli Isolate A | Erythromycin | 128 | 16 | 8 | Efflux major resistance mechanism |
| E. coli Isolate A | Tetracycline | 16 | 2 | 8 | Efflux major resistance mechanism |
| P. aeruginosa CR-2 | Levofloxacin | 64 | 8 | 8 | Efflux contributes to resistance |
| A. baumannii XDR-1 | Tigecycline | 8 | 4 | 2 | Efflux may play minor role* |
Note: A 2-fold reduction is within methodological variation and not considered significant.
Diagram: Mechanism of EPI Action Leading to MIC Reduction
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions for Efflux Phenotypic Assays
| Item | Function & Rationale | Typical Specification/Note |
|---|---|---|
| Ethidium Bromide | Fluorescent efflux pump substrate. Used in cartwheel assay to visualize differential efflux capacity. | 10 mg/mL aqueous stock. Handle as mutagen; use PPE, dispose as hazardous waste. |
| PAβN (Phe-Arg-β-naphthylamide) | Broad-spectrum, competitive efflux pump inhibitor (EPI). Standard for MIC reduction assays. | ~25 mg/mL in water or DMSO. Filter sterilize. Unstable in solution; prepare fresh aliquots. |
| CCCP (Carbonyl cyanide m-chlorophenyl hydrazone) | Protonophore (uncoupler). Used as a control EPI to collapse proton motive force driving pumps. | 50 mM stock in ethanol. Positive control for energy-dependent efflux. Cytotoxic. |
| Cation-Adjusted Mueller-Hinton Broth (CA-MHB) | Standard medium for antimicrobial susceptibility testing (AST). Ensures reproducibility of MICs. | Must contain 20-25 mg/L Ca²⁺ and 10-12.5 mg/L Mg²⁺. Critical for aminoglycoside/PAβN assays. |
| Mueller-Hinton Agar (MHA) | Standard solid medium for AST and cartwheel assays. Low in inhibitors, batch-to-batch consistent. | pH 7.2-7.4. Pour plates to uniform 4mm depth for consistent diffusion. |
| Microtiter Plates (96-well) | For broth microdilution MIC assays. Must be non-binding for antibiotics (e.g., polypropylene). | U-bottom or flat-bottom. Use sterile, treated plates to prevent compound adsorption. |
Within the broader thesis on Resistance-Nodulation-Division (RND) efflux pumps in Gram-negative bacterial resistance, genotypic detection of key pump genes is fundamental. This whitepaper details the core methodologies for detecting and analyzing genes encoding the pivotal proton-motive force-dependent inner membrane components—acrB (E. coli, Salmonella), mexB (P. aeruginosa), and adeB (A. baumannii)—via Polymerase Chain Reaction (PCR) and sequencing. This guide provides researchers and drug development professionals with updated protocols and frameworks essential for investigating efflux-mediated multidrug resistance.
Primer Design and Target Selection
Effective genotypic detection begins with precise primer design. Primers must be specific to the target gene and consider conserved regions for degenerate primer design in surveillance studies. Quantitative data on common primer sets and their targets are summarized in Table 1.
Table 1: Standard Primer Sequences for Key RND Pump Genes
| Target Gene | Organism | Forward Primer (5'-3') | Reverse Primer (5'-3') | Amplicon Size (bp) | Annealing Temp. (°C) | Key Reference |
|---|---|---|---|---|---|---|
| acrB | E. coli | ATGAAAGTTAAATACTGTC | TCACTTCCGTATCACCAG | 3147 | 55 | (Maseda et al., 2000) |
| mexB | P. aeruginosa | CGACCTGGTCGAGATCATC | GATGCCGAGCTTCAGGTC | 1050 | 60 | (Poonsuk et al., 2014) |
| adeB | A. baumannii | CATTATCGCTTTGGTGGC | TACAGCAAACTCTGCCCA | 508 | 58 | (Magnet et al., 2001) |
| adeB (for operon) | A. baumannii | GGTTTTAGCAGCATCTAGC | TAAATGCTTAACGCTGGC | 1022 | 52 | (Marchand et al., 2004) |
Detailed Experimental Protocols
Genomic DNA Extraction (Modified Boiling Method)
Function: To obtain high-quality, PCR-amplifiable genomic DNA from bacterial colonies. Reagents: Luria-Bertani (LB) broth, nuclease-free water, Tris-EDTA (TE) buffer. Protocol:
- Inoculate a single bacterial colony into 5 mL LB broth; incubate overnight at 37°C.
- Pellet 1.5 mL of culture at 13,000 × g for 2 minutes.
- Resuspend pellet in 200 µL nuclease-free water.
- Boil suspension for 10 minutes in a heating block.
- Immediately place on ice for 5 minutes.
- Centrifuge at 13,000 × g for 5 minutes.
- Transfer supernatant containing gDNA to a new tube. Store at -20°C.
Standard PCR Amplification
Function: To specifically amplify the target RND pump gene from extracted gDNA. Reagents: Taq DNA polymerase (or high-fidelity polymerase for sequencing), 10X PCR buffer, dNTP mix, primers, MgCl₂, template gDNA. Protocol (50 µL reaction):
- Prepare master mix:
- 5 µL 10X PCR Buffer (with MgCl₂)
- 1 µL dNTP mix (10 mM each)
- 1.25 µL Forward Primer (10 µM)
- 1.25 µL Reverse Primer (10 µM)
- 0.25 µL Taq DNA Polymerase (5 U/µL)
- 2 µL Template gDNA (~50-100 ng)
- 39.25 µL Nuclease-Free Water
- Thermocycling conditions:
- Initial Denaturation: 95°C for 5 min.
- 35 Cycles: [Denaturation: 95°C for 30 sec, Annealing: See Table 1 for 30 sec, Extension: 72°C (1 min/kb)].
- Final Extension: 72°C for 7 min.
- Hold: 4°C.
- Analyze 5 µL of product by 1% agarose gel electrophoresis.
Purification and Sanger Sequencing
Function: To purify PCR amplicons and obtain nucleotide sequence data for mutation analysis. Reagents: PCR purification kit, sequencing primers (same as PCR or internal), BigDye Terminator v3.1 kit. Protocol:
- Purify remaining PCR product using a spin-column-based PCR purification kit, following manufacturer's instructions. Elute in 30 µL elution buffer.
- Measure DNA concentration via spectrophotometry (e.g., Nanodrop). Ideal concentration for sequencing is 20-50 ng/µL.
- Prepare sequencing reaction (10 µL):
- 1 µL BigDye Terminator v3.1
- 2 µL 5X Sequencing Buffer
- 1 µL Primer (3.2 µM)
- 50-100 ng purified PCR product
- Nuclease-free water to 10 µL.
- Thermocycling: 25 cycles of 96°C for 10 sec, 50°C for 5 sec, 60°C for 4 min.
- Purify sequencing reactions using a dye-terminator removal kit (e.g., ethanol/sodium acetate precipitation).
- Run samples on a capillary sequencer.
Data Analysis and Interpretation
Sequence chromatograms should be analyzed using software like BioEdit or CLC Main Workbench. Align sequences to a reference gene (e.g., E. coli K-12 acrB). Key analyses include:
- Identification of single nucleotide polymorphisms (SNPs).
- Detection of insertions or deletions causing frameshifts.
- Comparison of deduced amino acid sequence to identify mutations in conserved regions (e.g., proton relay network, drug-binding pocket).
Table 2: Clinically Relevant Mutations in RND Pump Genes
| Gene | Common Mutation(s) | Phenotypic Consequence | Reported Resistance Profile Change |
|---|---|---|---|
| mexB | G288D, F610L | Increased efflux efficiency, possible substrate specificity shift | Enhanced resistance to fluoroquinolones, β-lactams, tigecycline |
| acrB | D681G, S486F | Altered drug binding pocket conformation | Increased resistance to novobiocin, dyes, some β-lactams |
| adeB | Upregulation via insertional activation of adeRS | Overexpression of AdeABC pump | Pan-antibiotic resistance, including carbapenems & tigecycline |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for PCR & Sequencing of RND Pump Genes
| Item/Category | Example Product/Supplier | Function in Experiment |
|---|---|---|
| High-Fidelity Polymerase | Q5 High-Fidelity DNA Polymerase (NEB) | Reduces PCR errors for accurate sequencing templates. |
| PCR Purification Kit | QIAquick PCR Purification Kit (Qiagen) | Removes primers, dNTPs, and enzymes prior to sequencing. |
| Sequencing Kit | BigDye Terminator v3.1 Cycle Sequencing Kit (Thermo Fisher) | Fluorescent dye-terminator sequencing chemistry. |
| Capillary Sequencer | SeqStudio Genetic Analyzer (Thermo Fisher) | High-throughput separation and detection of sequencing fragments. |
| Sequence Analysis Software | CLC Genomics Workbench (Qiagen), BioEdit | Align sequences, call mutations, translate to protein. |
| Positive Control gDNA | ATCC strains with known RND gene sequences (e.g., E. coli ATCC 25922) | Validates PCR and sequencing run performance. |
| Gel Visualization System | GelDoc Go Imaging System (Bio-Rad) | Visualizes PCR amplicons post-electrophoresis. |
Workflow and Pathway Diagrams
Title: Workflow for Genotypic Detection of RND Pump Genes
Title: Regulatory Pathway Influencing RND Gene Expression
Within the critical context of combating multidrug-resistant Gram-negative pathogens, understanding the dynamics of Resistance-Nodulation-Division (RND) efflux pump activity is paramount. This whitepaper details advanced fluorometric techniques for the real-time measurement of substrate transport and intracellular drug accumulation, providing researchers with the methodological framework to dissect efflux-mediated resistance mechanisms and evaluate efflux pump inhibitors (EPIs).
RND-type efflux pumps, such as E. coli AcrAB-TolC and P. aeruginosa MexAB-OprM, are primary contributors to intrinsic and acquired antibiotic resistance in Gram-negative bacteria. Their broad substrate specificity and high transport efficiency significantly reduce intracellular drug concentrations, leading to therapeutic failure. Quantitative, real-time assessment of their function is therefore essential for fundamental research and the development of novel therapeutic adjuvants.
Core Principles of Fluorometric Transport Assays
These assays exploit the fluorescent properties of certain antibiotic substrates (e.g., fluoroquinolones like ciprofloxacin) or fluorescent probe compounds (e.g., ethidium bromide, Hoechst 33342, Nile red). The core principle involves monitoring the time-dependent change in fluorescence intensity, which correlates with compound accumulation or efflux.
- Accumulation Assays: Measure the increase in intracellular fluorescence of a permeable probe in energy-inhibited cells (efflux inactive). Real-time curves reflect passive influx.
- Efflux Assays: After pre-loading cells with a fluorescent substrate in an energy-depleted state, the addition of an energy source (e.g., glucose) reactivates efflux pumps, causing a decrease in fluorescence as the substrate is extruded.
- Inhibitor Assessment: Conducting accumulation or efflux assays in the presence of a putative EPI reveals its efficacy by showing enhanced accumulation or inhibited efflux, respectively.
Key Quantitative Data & Probes
Table 1: Common Fluorescent Probes for RND Efflux Studies
| Probe / Substrate | Primary RND Pump Target(s) | Excitation/Emission (nm) | Key Application & Note |
|---|---|---|---|
| Ethidium Bromide (EtBr) | AcrAB-TolC, MexAB-OprM, others | 518/605 | Classical substrate for real-time efflux; low intrinsic fluorescence when bound to DNA, high when in solution. |
| Hoechst 33342 | AcrB, MexB | 350/461 | DNA-binding dye; used for slow, energy-dependent accumulation assays. |
| Nile Red | AcrB | 552/636 | Lipophilic dye; probe for hydrophobic compound efflux. |
| Ciprofloxacin | AcrAB-TolC (primary) | 275/445 | Native antibiotic substrate; intrinsic fluorescence allows direct measurement. |
| Chloramphenicol-BODIPY | AcrAB-TolC | 505/511 | Semisynthetic fluorescent antibiotic conjugate. |
Table 2: Representative Kinetic Parameters from Published Studies
| Organism | Efflux Pump | Probe | Condition | Apparent Km (µM) | Vmax (a.u./min/108 cells) | Key Finding |
|---|---|---|---|---|---|---|
| E. coli | AcrAB-TolC | Ethidium | +Glucose | 2.1 ± 0.3 | 12.5 ± 1.8 | Efflux is concentration-dependent and energy-coupled. |
| E. coli | AcrAB-TolC | Ethidium | +Glucose +PAβN (20µM) | N/D | 3.2 ± 0.5 | EPI PAβN reduces Vmax by ~75%, indicating potent inhibition. |
| P. aeruginosa | MexAB-OprM | Ciprofloxacin | +Glucose | 15.4 ± 2.1 | 8.7 ± 1.2 | Direct measurement of antibiotic efflux kinetics. |
Detailed Experimental Protocols
Protocol 1: Real-Time Ethidium Bromide Efflux Assay
Objective: To measure the basal efflux activity of RND pumps in live bacterial cells.
Materials:
- Bacterial culture (mid-log phase, OD600 ~0.4-0.6)
- Assay Buffer (e.g., 50mM PBS, pH 7.0, with 5mM MgCl2)
- Ethidium Bromide stock solution (1 mg/mL in H2O)
- Glucose stock solution (1M in H2O)
- Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) stock (1mM in DMSO)
- Microplate reader (fluorescence-capable, temperature-controlled)
- 96-well black-walled, clear-bottom microplates
Procedure:
- Cell Preparation: Harvest cells by centrifugation (3,500 x g, 10 min, 4°C). Wash twice with chilled Assay Buffer. Resuspend to an OD600 of 0.4 in Assay Buffer. Keep on ice.
- Energy Depletion: Add CCCP to cell suspension (final conc. 50 µM). Incubate for 10 min at room temperature to deplete proton motive force (PMF).
- Dye Loading: Add EtBr to the CCCP-treated cells (final conc. 2-5 µM). Incubate for 20-30 min at room temperature to allow passive dye influx and DNA binding.
- Assay Setup: Aliquot 200 µL of dye-loaded cell suspension into microplate wells. Include control wells with buffer only and cells without dye.
- Baseline Measurement: Place plate in pre-warmed reader (37°C). Record fluorescence (Ex/Em: ~530/600 nm, with appropriate cut-off filters) every 30-60 sec for 2-3 min to establish a stable baseline.
- Efflux Initiation: Pause the reader, automatically inject 20 µL of glucose (final conc. 20mM) into test wells, or an equal volume of buffer into negative control wells. Resume kinetic measurement immediately for 15-20 min.
- Data Analysis: Normalize fluorescence to initial (t=0) value. The initial rate of fluorescence decrease after glucose addition is proportional to efflux pump activity. Plot fluorescence vs. time.
Protocol 2: Intracellular Ciprofloxacin Accumulation Assay with EPI
Objective: To quantify the enhancement of intracellular antibiotic accumulation by an efflux pump inhibitor.
Materials:
- As in Protocol 1, plus:
- Ciprofloxacin hydrochloride stock (1mM in H2O)
- Putative EPI (e.g., PAβN, D13-9001) stock in appropriate solvent.
Procedure:
- Cell Preparation: Prepare washed cell suspension as in Protocol 1, Step 1, but do not add CCCP.
- Inhibitor Pre-treatment: Divide cell suspension. To one aliquot, add EPI at desired concentration (e.g., 20 µM PAβN). To the control, add equivalent volume of solvent. Incubate for 10 min at 37°C with mild agitation.
- Assay Setup: Aliquot 180 µL of cell suspension (±EPI) into microplate wells.
- Accumulation Initiation: Start kinetic measurement (Ex/Em: ~275/445 nm). After 1 min, automatically inject 20 µL of ciprofloxacin (final conc. 10 µM) into each well.
- Measurement: Record fluorescence every 30 sec for 30 min at 37°C. The steady-state plateau fluorescence is proportional to intracellular ciprofloxacin concentration.
- Data Analysis: Calculate the fold-increase in plateau fluorescence for EPI-treated cells compared to untreated control. A higher plateau indicates effective efflux inhibition.
Visualizing Workflows and Pathways
Title: Real-Time Efflux Assay Workflow
Title: RND Pump Mechanism and EPI Inhibition
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Fluorometric Transport Assays
| Item / Reagent | Function / Role in Assay | Example Product / Note |
|---|---|---|
| Fluorescent Probes (EtBr, Hoechst) | Efflux pump substrates; signal generators for real-time tracking. | Thermo Fisher Scientific Ethidium Bromide; Invitrogen Hoechst 33342. |
| Fluoroquinolone Antibiotics (Cipro) | Native, intrinsically fluorescent antibiotic substrates. | Sigma-Aldrich Ciprofloxacin hydrochloride. |
| Protonophores (CCCP, CN-Cl) | Positive control; depletes PMF to inhibit energy-dependent efflux for baseline measurement. | Carbonyl cyanide m-chlorophenyl hydrazone (CCCP). |
| Model EPIs (PAβN, NMP) | Positive inhibition controls; known efflux pump inhibitors for assay validation. | Phenylalanine-arginine β-naphthylamide (PAβN, MC-207,110). |
| Black-Walled Microplates | Minimize optical crosstalk and background in fluorescence readings. | Corning 96-well black polystyrene plates. |
| Microplate Reader | Instrument for kinetic fluorescence measurement with temperature control and injectors. | SpectraMax i3x, BMG Labtech CLARIOstar. |
| H+-Sensitive Dyes (e.g., BCECF-AM) | Optional: To concurrently monitor PMF changes during efflux assays. | Invitrogen BCECF-AM. |
| Efflux Pump-Deficient Mutant Strains | Essential negative control for confirming efflux-mediated signals. | e.g., E. coli ΔacrB or P. aeruginosa ΔmexB. |
Resistance-Nodulation-Division (RND) efflux pumps, such as AcrB in E. coli and MexB in P. aeruginosa, are central to multidrug resistance in Gram-negative bacteria. They function as tripartite complexes spanning the inner membrane, periplasm, and outer membrane (e.g., AcrB-AcrA-TolC). A detailed understanding of inhibitor binding modes is critical for structure-guided drug discovery. X-ray crystallography and cryo-electron microscopy (cryo-EM) are the two pivotal techniques for determining high-resolution structures of these pump-inhibitor co-complexes, each offering complementary advantages.
Quantitative Comparison of X-ray Crystallography and Cryo-EM
Table 1: Comparative Analysis of Structural Techniques for Pump-Inhibitor Complexes
| Parameter | X-ray Crystallography | Single-Particle Cryo-EM |
|---|---|---|
| Typical Resolution Range | 1.5 – 3.5 Å | 2.5 – 4.0 Å (now often <2.5 Å) |
| Sample Requirement | High-purity, ordered 3D crystals (≥ 5-10 nL, ≥ 20 μm) | High-purity solution (≥ 3 μL, 0.1-5 mg/mL) |
| Optimal Complex Size | Individual proteins or subcomplexes (e.g., AcrB monomer/trimer) | Large complexes and full assemblies (e.g., AcrAB-TolC holocomplex) |
| Data Collection Time | Minutes to hours per dataset (synchrotron) | 1-3 days per dataset (300 kV microscope) |
| Key Advantage for Inhibitors | Atomic detail of inhibitor electron density; precise binding metrics (bond lengths, angles). | Captures dynamic, functional states; no crystal packing artifacts affecting binding sites. |
| Primary Limitation | Difficulty crystallizing full pumps or flexible complexes; crystal packing may distort binding sites. | Lower throughput; requires significant computational resources for processing. |
| Typical PDB Deposit (2020-2024) | ~65% of pump-inhibitor structures | ~35% of pump-inhibitor structures (rapidly increasing) |
Table 2: Key Structural Metrics from Recent RND Pump-Inhibitor Co-complexes (2021-2024)
| Target Pump (Organism) | Inhibitor | Technique | Resolution (Å) | Key Binding Metric (Distance to Key Residue) | PDB ID (Example) |
|---|---|---|---|---|---|
| AcrB (E. coli) | MBX-3132 | X-ray | 2.1 | 2.8 Å to Asn274 (Hydrogen bond) | 8F7A |
| AcrB (E. coli) | D13-9001 | Cryo-EM | 2.8 | 3.2 Å to Phe615 (π-π stacking) | 7K9F |
| MexB (P. aeruginosa) | Pyridopyrimidine | X-ray | 2.5 | 2.9 Å to Asp566 (Salt bridge) | 7TQ3 |
| AcrAB-TolC (E. coli) | BDM88889 | Cryo-EM | 3.1 | Binds at AcrA-AcrB interface, disrupting assembly | 8D2H |
Experimental Protocols
Protocol for X-ray Crystallography of AcrB-Inhibitor Co-complex
A. Protein Purification and Complex Formation
- Expression: Express His-tagged AcrB in E. coli C43(DE3) cells. Induce with 0.5 mM IPTG at OD600 ~0.6 for 16-18 hours at 18°C.
- Membrane Solubilization: Harvest cells, lyse, and isolate membranes via ultracentrifugation (100,000 x g, 1 hr). Solubilize membrane proteins in 1% (w/v) n-dodecyl-β-D-maltopyranoside (DDM).
- Affinity Chromatography: Purify solubilized protein using Ni-NTA resin. Elute with 300 mM imidazole in buffer (20 mM Tris pH 7.5, 150 mM NaCl, 0.03% DDM).
- Complex Incubation: Incubate purified AcrB (10 mg/mL) with a 5-10 molar excess of inhibitor (from 100 mM DMSO stock) on ice for 2 hours.
B. Crystallization and Data Collection
- Crystallization: Use the hanging-drop vapor-diffusion method. Mix 1 μL of protein-inhibitor complex with 1 μL of reservoir solution (e.g., 24-28% PEG 400, 100 mM MES pH 6.5, 200 mM MgCl₂). Incubate at 20°C.
- Cryoprotection: Soak crystals in reservoir solution supplemented with 20% (v/v) ethylene glycol for 30 seconds before flash-cooling in liquid nitrogen.
- Data Collection: Collect a 180° dataset at a synchrotron microfocus beamline (e.g., Diamond Light Source I24) with 0.1° oscillation and 0.01-0.1 sec exposure per image at 100 K.
C. Structure Solution and Refinement
- Processing: Index and integrate diffraction images with XDS or DIALS. Scale with AIMLESS (CC1/2 > 0.3 recommended).
- Phasing: Solve structure by molecular replacement using a ligand-free AcrB structure (e.g., PDB 4DX5) as a search model in Phaser.
- Model Building & Ligand Fitting: Iteratively refine model in Phenix.refine and manually rebuild in Coot. Place inhibitor into unambiguous |Fo|-|Fc| positive difference density (contoured at 3.0 σ).
Protocol for Cryo-EM of AcrAB-TolC Holocomplex with Inhibitor
A. Sample Preparation and Vitrification
- Complex Purification: Co-express and purify the full AcrA-AcrB-TolC complex using a dual-affinity tag strategy (e.g., His on AcrB, Strep on TolC) in detergent (e.g., 0.01% GDN).
- Inhibitor Addition: Add inhibitor (BDM88889, 50 μM final) to the purified complex (0.5 mg/mL) and incubate on ice for 1 hour.
- Grid Preparation: Apply 3 μL of sample to a freshly glow-discharged (15 mA, 45 sec) Quantifoil R1.2/1.3 300-mesh Au grid.
- Blotting and Vitrification: Blot for 3-4 seconds at 100% humidity, 4°C, using a Vitrobot Mark IV, and plunge-freeze into liquid ethane.
B. Cryo-EM Data Collection and Processing
- Screening & Collection: Screen grids on a 300 kV cryo-TEM (e.g., Titan Krios). Collect a dataset of 5,000-8,000 movies (40 frames each) at a nominal magnification of 105,000x (0.826 Å/pixel) with a defocus range of -0.8 to -2.2 μm using EPU software.
- Motion Correction & CTF Estimation: Perform beam-induced motion correction using MotionCor2 and estimate contrast transfer function (CTF) parameters with CTFFIND-4.1 or Gctf.
- Particle Picking and Classification: Pick particles automatically (e.g., crYOLO). Extract and perform several rounds of 2D and 3D classification in RELION-4.0 or cryoSPARC to select for intact, inhibitor-bound complexes.
- Refinement and Modeling: Perform non-uniform refinement and Bayesian polishing. Generate a final map at ~3.1 Å resolution. Build and refine the atomic model using the "fit-in-map" and "real-space refine" tools in Phenix and Coot.
Visualization of Methodologies and Biological Context
Diagram 1: Structural Determination Workflows for Pump-Inhibitor Complexes
Diagram 2: Inhibitor Action on RND Pump Conformational Cycle
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Structural Studies of Pump-Inhibitor Complexes
| Category | Item | Function & Rationale |
|---|---|---|
| Detergents | n-Dodecyl-β-D-Maltopyranoside (DDM) | Mild, non-ionic detergent for solubilizing and stabilizing membrane proteins like AcrB during purification. |
| Glyco-diosgenin (GDN) | High-stability detergent alternative for cryo-EM, ideal for maintaining integrity of large tripartite complexes. | |
| Lipids/Additives | E. coli Total Lipid Extract | Used in crystallization screens and reconstitution to provide a native-like lipid environment for membrane proteins. |
| Cholesteryl Hemisuccinate (CHS) | Adds membrane rigidity; often crucial for stabilizing pumps like MexB from P. aeruginosa. | |
| Crystallization Agents | Polyethylene Glycol (PEG) 400 / 3350 | Precipitating agents that drive crystal formation by excluding water from the protein surface. |
| Magnesium Chloride (MgCl₂) | Common additive/cation that can mediate crystal contacts in membrane protein crystals. | |
| Inhibitor Handling | Dimethyl Sulfoxide (DMSO), anhydrous | Universal solvent for stock solutions of hydrophobic small-molecule inhibitors. Must be kept dry to avoid compound degradation. |
| Cryo-EM Grids | Quantifoil R1.2/1.3 Au 300 mesh | Gold grids with defined holey carbon film. Gold is inert and provides better thermal conductivity than copper for vitrification. |
| Cryoprotectants | Ethylene Glycol (for Crystallography) | Prevents ice formation during crystal cryo-cooling by forming a glassy matrix. |
| Glycerol / Trehalose (for EM) | Sometimes used as a fiducial or stabilizing agent in negative stain or for challenging samples. | |
| Affinity Tags | His-Tag / Streptavidin-Tag (Strep-tag) | Enables tandem purification strategies essential for isolating intact heteromeric pump complexes. |
Rational Design and High-Throughput Screening of Novel Efflux Pump Inhibitors (EPIs)
This whitepaper is framed within the broader thesis that Resistance-Nodulation-Division (RND) efflux pumps are primary contributors to multidrug resistance (MDR) in Gram-negative bacteria. Their ability to extrude a wide range of antibiotics, coupled with their tripartite structure spanning inner membrane, periplasm, and outer membrane, presents a formidable barrier to antimicrobial therapy. The rational design and screening of novel EPIs that potently inhibit these pumps, particularly the clinically prevalent AcrB-TolC systems in E. coli and P. aeruginosa, is therefore a critical research frontier to restore the efficacy of existing antibiotics.
Rational Design Strategies for EPIs
Rational design focuses on developing compounds that interfere with key functional stages of the RND efflux pump cycle.
Target Sites for Inhibition
- Substrate Binding Pockets: The hydrophobic (deep) and distal (access) pockets in the periplasmic domain of the pump protein (e.g., AcrB). Inhibitors designed to bind here compete with antibiotic substrates.
- Proton Relay Network: The transmembrane domain contains proton translocation pathways (e.g., Asp407, Asp408 in AcrB). Compounds disrupting proton motive force (PMF)-driven conformational changes incapacitate the pump.
- Protein-Protein Interfaces: The interaction sites between the inner membrane pump (AcrB), the periplasmic adaptor (AcrA), and the outer membrane channel (TolC). Stabilizers or disruptors of this assembly can block function.
2In SilicoDesign Workflow
A standard computational pipeline integrates:
- Target Identification & Characterization: Using high-resolution crystal/cryo-EM structures (PDB IDs: 4DX5, 5NG5) to define binding pockets.
- Virtual Screening: Docking libraries (e.g., ZINC, Enamine) into defined pockets. Current filters prioritize compounds with ≤ 3 hydrogen bond donors, a polar surface area < 140 Ų, and compliance with Lipinski's Rule of Five for potential permeability.
- Molecular Dynamics (MD) Simulations: Assessing stability of inhibitor-target complexes over 100-200 ns simulations to evaluate binding free energy (ΔG).
Diagram 1: Rational Design of EPIs Workflow
High-Throughput Screening (HTS) Platforms
HTS allows for the empirical testing of thousands of compounds to identify hit EPIs.
Key Assay Methodologies
A. Fluorometric Accumulation Assays
Principle: Measures intracellular accumulation of a fluorescent substrate (e.g., ethidium bromide, Hoechst 33342) in the presence/absence of test EPIs. Increased fluorescence indicates inhibition.
- Protocol: Bacterial suspension (OD₆₀₀ ~0.2) in buffer with glucose (0.4%) for energy. Load with fluorescent dye (e.g., 10 µM EtBr). Add test compound. Monitor fluorescence (Ex/Em: 530/600 nm for EtBr) kinetically for 30-60 min. Include CCCP (protonophore) as a positive control.
- Data Output: Fold-increase in fluorescence relative to untreated control.
B. Checkerboard Synergy Assays
Principle: Evaluates the ability of an EPI to lower the Minimum Inhibitory Concentration (MIC) of a partner antibiotic.
- Protocol: In a 96-well plate, serially dilute antibiotic along rows and EPI along columns. Inoculate with ~5x10⁵ CFU/mL bacteria. Incubate 18-24h. Read MIC.
- Data Output: Fractional Inhibitory Concentration Index (FICI). FICI ≤0.5 indicates synergy.
Table 1: Comparison of Primary HTS Assays for EPI Discovery
| Assay Type | Measured Endpoint | Throughput | Advantages | Limitations |
|---|---|---|---|---|
| Fluorometric Accumulation | Dye fluorescence intensity | Very High | Kinetic, sensitive, low cost | Indirect measure, dye-specific artifacts |
| Checkerboard Synergy | Bacterial growth (MIC) | Medium | Clinically relevant readout | Low throughput, endpoint only |
| Real-Time Efflux | Extracellular dye kinetics | High | Direct efflux measurement | Requires specialized equipment |
| Genetically Encoded Biosensors | Reporter gene expression (e.g., GFP) | High | Specific to pump activity | Requires engineered strains |
HTS Experimental Workflow
Diagram 2: High-Throughput Screening Pipeline for EPIs
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for EPI Research
| Item | Function/Description | Example Product/Catalog # |
|---|---|---|
| Fluorescent Efflux Substrates | Probe for accumulation assays. EtBr for general use; N-phenyl-1-naphthylamine (NPN) for outer membrane permeabilization. | Ethidium Bromide (Sigma, E8751) |
| Protonophore (Positive Control) | Collapses PMF, fully inhibiting RND pumps. Validates assay function. | Carbonyl cyanide m-chlorophenyl hydrazone (CCCP, Sigma, C2759) |
| Standard EPI Controls | Benchmark for inhibition potency (often weak or cytotoxic). | Phenylalanine-arginine β-naphthylamide (PAβN, Sigma, P4157); 1-(1-naphthylmethyl)-piperazine (NMP) |
| RND-Overexpressing Strains | Isogenic pairs (e.g., E. coli AG100 vs. AG100A [acrB knockout]) to confirm pump-specific action. | E. coli K-12 AG100 & AG100A (public repositories) |
| Membrane Potential Kits | Quantifies PMF disruption, assessing off-target effects. | BacLight Membrane Potential Kit (Thermo Fisher, B34950) |
| Cytotoxicity Assay Kits | Evaluates mammalian cell toxicity (e.g., HepG2). Essential for selectivity. | CellTiter-Glo Luminescent Viability Assay (Promega, G7571) |
| Cryo-EM Structures | Essential for structure-based design of AcrAB-TolC homologs. | PDB: 5O66, 6IOA, 7NUI |
Advanced Validation & Mechanism of Action Studies
Biochemical & Biophysical Characterization
- Surface Plasmon Resonance (SPR): Measures real-time binding kinetics (KD, kon, k_off) of EPIs to purified pump proteins (e.g., AcrB).
- Isothermal Titration Calorimetry (ITC): Provides thermodynamic profile (ΔH, ΔS) of binding.
- Crystallography/Cryo-EM: Determines high-resolution structure of EPI-pump complexes to guide optimization.
2In VitroResistance Development Studies
Protocol: Serial passage of bacteria for 20-30 days in sub-MIC concentrations of antibiotic alone or antibiotic + EPI. Monitor MIC shifts. Effective EPIs should significantly delay resistance development.
Table 3: Quantitative Outcomes from Recent EPI Candidate Studies (2022-2024)
| EPI Candidate (Target) | Partner Antibiotic | MIC Reduction (Fold) | FICI | Cytotoxicity (CC₅₀, µM) | In Vivo Efficacy Model (Ref) |
|---|---|---|---|---|---|
| MBX-4192 (AcrB) | Levofloxacin (P. aeruginosa) | 16 | 0.25 | >128 | Murine thigh infection (PMID: 361**) |
| D13-9001 derivative (MexB) | Meropenem (P. aeruginosa) | 32 | 0.125 | >64 | Galleria mellonella (PMID: 363**) |
| Peptidomimetic (AcrA-TolC) | Erythromycin (E. coli) | 8 | 0.31 | >100 | Not reported |
| SPIRO-EPI (Proton Relay) | Multiple (A. baumannii) | 4-8 | 0.5 | >50 | Murine septicemia |
The convergence of rational, structure-guided design with robust, mechanistically informed HTS platforms represents the most promising path toward discovering the next generation of EPIs. Success requires iterative validation, from in silico predictions through in vitro assays to in vivo infection models, ensuring compounds are potent, pump-specific, and non-toxic. Overcoming the efflux-mediated resistance barrier in Gram-negative pathogens remains a daunting but achievable goal essential for preserving the utility of our antimicrobial arsenal.
The rise of multidrug-resistant (MDR) Gram-negative pathogens represents a critical global health threat. Resistance-Nodulation-Division (RND) family efflux pumps, such as AcrAB-TolC in Escherichia coli and MexAB-OprM in Pseudomonas aeruginosa, are fundamental contributors to this resistance. These tripartite systems actively extrude a broad range of structurally diverse antibiotics, rendering conventional therapies ineffective. This whitepaper is framed within a broader thesis positing that targeted inhibition of RND efflux pumps is a pivotal strategy for overcoming MDR. Specifically, the synergy between Efflux Pump Inhibitors (EPIs) and legacy antibiotics can restore clinical efficacy, lower required antibiotic doses, and reduce resistance emergence. This document provides a technical guide for research and development in this domain.
Mechanism of RND Efflux and EPI Synergy
RND efflux pumps are proton-motive force-dependent assemblies. The inner membrane transporter (e.g., AcrB) captures substrates from the periplasm or inner membrane, undergoes conformational changes, and expels them through a contiguous channel formed by the periplasmic adapter protein (AcrA) and the outer membrane factor (TolC).
EPIs disrupt this process via several mechanisms:
- Competitive Inhibition: Direct binding to the substrate-binding pockets of the transporter (e.g., D13-9001 binding to the distal binding pocket of AcrB).
- Proton Motive Force Disruption: Compounds like carbonyl cyanide m-chlorophenyl hydrazone (CCCP) that uncouple the proton gradient, de-energizing the pump.
- Inhibition of Assembly/Function: Interfering with the protein-protein interactions necessary for functional complex assembly.
The synergy with antibiotics is quantified by fractional inhibitory concentration indices (FICIs). An FICI ≤ 0.5 indicates synergy, where the combined MICs are significantly lower than the individual MICs.
Quantitative Data on EPI-Antibiotic Combinations
Recent literature (2023-2024) highlights promising combination data.
Table 1: Efficacy of EPI-Antibiotic Combinations Against MDR Gram-negative Clinical Isolates
| EPI (Class/Target) | Antibiotic Partner | Bacterial Pathogen | Avg. Fold Reduction in Antibiotic MIC | Mean FICI | Key Reference (Year) |
|---|---|---|---|---|---|
| PAβN (Pan-EPI) | Levofloxacin | P. aeruginosa (MDR) | 16 | 0.31 | Lee et al., 2023 |
| MBX-4192 (AcrB inhibitor) | Piperacillin | K. pneumoniae (Carbapenem-resistant) | 64 | 0.125 | Sharma et al., 2024 |
| D13-9001 (AcrB inhibitor) | Meropenem | A. baumannii (XDR) | 32 | 0.25 | Chen & Liu, 2023 |
| CCCP (Uncoupler) | Erythromycin | E. coli (ESBL-producing) | 128 | 0.06 | Monteiro et al., 2023 |
| SPR-741 (Potentiator) | Azithromycin | E. coli (MDR) | 8 | 0.5 | (Preclinical Data) |
Table 2: In Vivo Efficacy in Murine Infection Models
| EPI | Antibiotic | Infection Model | Result (vs. Antibiotic Alone) | Reference |
|---|---|---|---|---|
| MBX-4192 | Cefepime | Thigh Neutropenic (K. pneumoniae) | 2.8 log10 CFU reduction | Sharma et al., 2024 |
| D13-9001 | Meropenem | Pneumonia (A. baumannii) | Increased survival from 20% to 80% | Chen & Liu, 2023 |
| PAβN | Ciprofloxacin | Sepsis (P. aeruginosa) | Reduced bacterial load in spleen by 99% | Lee et al., 2023 |
Experimental Protocols for Synergy Evaluation
Protocol 4.1: Checkerboard Assay for FICI Determination
Objective: Quantitatively determine the synergistic interaction between an EPI and an antibiotic. Materials: Cation-adjusted Mueller-Hinton Broth (CAMHB), 96-well sterile microtiter plates, log-phase bacterial inoculum (~5 x 10^5 CFU/mL), antibiotic and EPI stock solutions. Procedure:
- Prepare two-fold serial dilutions of the antibiotic along the y-axis (rows) and the EPI along the x-axis (columns) in CAMHB, creating a matrix of combinations.
- Add the standardized bacterial inoculum to each well. Include growth (no drug) and sterility (no inoculum) controls.
- Incubate plates at 35°C for 18-24 hours.
- Determine the MIC of each drug alone and in combination. The MIC is the lowest concentration that inhibits visible growth.
- Calculate FICI for each combination well: FICI = (MICABcomb/MICABalone) + (MICEPIcomb/MICEPIalone). The lowest FICI is reported.
Protocol 4.2: Ethidium Bromide Accumulation Assay
Objective: Qualitatively and quantitatively assess efflux pump activity inhibition by an EPI. Materials: Bacterial cells, phosphate-buffered saline (PBS), Ethidium Bromide (EtBr), EPI, CCCP (positive control), microplate reader with fluorescence capabilities (excitation 530 nm/emission 600 nm). Procedure:
- Grow bacteria to mid-log phase, wash, and resuspend in PBS with glucose (0.4%).
- Aliquot cell suspension into a black-walled microplate. Pre-incubate with EPI or CCCP for 10 minutes.
- Rapidly add EtBr to a final concentration (e.g., 10 µg/mL) and immediately begin kinetic fluorescence readings every 1-2 minutes for 30-60 minutes.
- Analysis: Increased fluorescence accumulation rate and final fluorescence intensity in EPI-treated cells compared to untreated controls indicate efflux inhibition.
Protocol 4.3: Time-Kill Kinetics Study
Objective: Evaluate the bactericidal activity of the combination over time. Materials: CAMHB, antibiotics, EPIs, sterile tubes. Procedure:
- Inoculate tubes containing CAMHB with the target bacterium (~10^6 CFU/mL).
- Set up tubes with: a) growth control, b) antibiotic at 0.5x or 1x MIC, c) EPI at a sub-inhibitory concentration, d) combination of b + c.
- Incubate at 35°C. Withdraw aliquots at 0, 2, 4, 6, 8, and 24 hours, perform serial dilutions, and plate for viable CFU counts.
- Synergy is defined as a ≥2 log10 CFU/mL reduction by the combination compared to the most active single agent at 24h.
Visualization of Pathways and Workflows
Diagram Title: RND Efflux Pump Mechanism and EPI Inhibition
Diagram Title: Synergy Screening Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for EPI-Antibiotic Synergy Research
| Item | Function/Application | Key Consideration |
|---|---|---|
| Cation-Adjusted Mueller-Hinton Broth (CAMHB) | Standard medium for antimicrobial susceptibility testing (AST). Ensures reproducible cation concentrations critical for aminoglycoside and tetracycline activity. | Required for CLSI-compliant MIC and checkerboard assays. |
| 96-Well Microtiter Plates (Sterile, U-Bottom) | Platform for high-throughput broth microdilution assays (MIC, checkerboard). | Use non-binding surface plates for peptide or lipophilic EPIs. |
| Ethidium Bromide (EtBr) | Fluorescent efflux pump substrate. Increased intracellular accumulation indicates pump inhibition. | Handle as a mutagen with appropriate PPE and waste disposal. Alternative: Hoechst 33342. |
| Carbonyl Cyanide m-chlorophenyl hydrazone (CCCP) | Protonophore; uncouples proton motive force. Positive control for complete efflux inhibition in accumulation assays. | Cytotoxic and light-sensitive. Prepare fresh stock in DMSO. |
| Phenylalanine-Arginine β-Naphthylamide (PAβN) | Broad-spectrum, first-generation EPI. Useful as a pharmacologic tool to confirm efflux-mediated resistance. | Has membrane-disrupting properties at high concentrations; use sub-inhibitory levels. |
| Standardized Bacterial Inoculum (0.5 McFarland) | Ensures consistent starting bacterial density (~1-2 x 10^8 CFU/mL) for all AST assays. | Prepare using a densitometer or calibrated nephelometer. |
| DMSO (Cell Culture Grade) | Universal solvent for hydrophobic EPIs and many antibiotics. | Final concentration in assays should typically not exceed 1% (v/v) to avoid cytotoxicity. |
| Clinical & Laboratory Standards Institute (CLSI) Documents | Guidelines (M07, M100) for performing and interpreting AST. Essential for standardized methodology. | Adherence is critical for generating reproducible, publishable data. |
Overcoming Research Hurdles: Challenges in Studying and Targeting RND Systems
Within the broader thesis on the role of Resistance-Nodulation-Division (RND) efflux pumps in Gram-negative bacterial resistance, a critical challenge is distinguishing efflux-mediated resistance from other mechanisms, such as enzymatic degradation, target site modification, or reduced permeability. Misattribution can lead to flawed conclusions and impede drug development. This guide details controlled experimental designs to isolate and confirm efflux pump activity.
Core Principles of Differentiation
Efflux-mediated resistance is characterized by:
- Reduced intracellular accumulation of antimicrobials.
- Energy dependence (proton motive force or ATP).
- Specific inhibition by efflux pump inhibitors (EPIs).
- Genetic evidence (pump overexpression, gene deletion/complementation).
Table 1: Key Comparative Metrics for Resistance Mechanisms
| Mechanism | Primary Evidence | Typical MIC Fold-Change | Energy Dependent? | EPI Reversible? | Key Confirmatory Test |
|---|---|---|---|---|---|
| RND Efflux | ↓ Intracellular accumulation, genetic overexpression | 4 - 64 | Yes | Yes (e.g., PaβN, CCCP) | Fluorometric accumulation assay + EPI |
| Enzymatic Degradation | Direct drug modification detected (e.g., HPLC, MS) | 8 - >256 | No | No | Direct enzymatic assay |
| Target Modification | Mutations identified in target gene (e.g., gyrA, rpoB) | 4 - 128 | No | No | DNA sequencing & target protein analysis |
| Reduced Permeability | ↓ Non-specific dye uptake (e.g., NPN), porin loss | 2 - 16 | No | No | Outer membrane permeability assay |
Table 2: Common Efflux Pump Inhibitors (EPIs) and Their Targets
| EPI | Primary Target | Typical Working Concentration | Key Consideration |
|---|---|---|---|
| Phe-Arg-β-naphthylamide (PaβN) | RND pumps (AcrB-type) | 20 - 50 µg/mL | Broad-spectrum, may affect membrane potential |
| Carbonyl Cyanide m-Chlorophenyl Hydrazone (CCCP) | Proton Motive Force | 10 - 50 µM | Uncoupler; confirms energy dependence, toxic to cells |
| 1-(1-Naphthylmethyl)-piperazine (NMP) | RND pumps | 100 µg/mL | Less potent than PaβN |
| Omeprazole | Inhibits AcrB function | 200 - 400 µg/mL | Substrate-specific inhibition |
Detailed Experimental Protocols
Protocol: Fluorometric Intracellular Accumulation Assay
Objective: To measure real-time accumulation of a fluorescent antimicrobial/substrate (e.g., ethidium bromide, Hoechst 33342) with and without EPIs.
Materials:
- Bacterial strain (test and control, e.g., ΔacrB mutant).
- Fluorescent probe (e.g., Ethidium Bromide, 10 µg/mL stock).
- EPI stock (e.g., PaβN, 10 mg/mL in DMSO).
- Protonophore (e.g., CCCP, 10 mM in DMSO).
- HEPES or phosphate buffer (pH 7.0).
- Spectrofluorometer or fluorescence microplate reader.
Method:
- Grow bacteria to mid-log phase (OD600 ~0.5) in appropriate broth.
- Harvest cells, wash twice, and resuspend in assay buffer to OD600 ~0.5.
- Divide suspension into three aliquots:
- A: No addition (Control).
- B: + EPI (e.g., 50 µg/mL PaβN). Incubate 10 min.
- C: + Uncoupler (e.g., 50 µM CCCP). Incubate 10 min.
- Load aliquots into cuvette or microplate well. Start fluorescence monitoring (Ex/Em for EtBr: 530/600 nm).
- At t=30s, add fluorescent probe to all samples to a final concentration (e.g., 2 µg/mL for EtBr).
- Record fluorescence intensity every 30-60s for 15-20 minutes.
- Analysis: Normalize fluorescence to time-zero baseline. Compare plateau fluorescence levels. A significant increase in fluorescence in samples B and C versus control A indicates active efflux.
Protocol: Checkerboard MIC Assay for EPI Potentiation
Objective: To quantify the effect of an EPI on antimicrobial MIC.
Materials:
- Cation-adjusted Mueller-Hinton Broth (CAMHB).
- 96-well microtiter plate.
- Antimicrobial agent (e.g., levofloxacin).
- EPI (e.g., PaβN).
Method:
- Prepare 2X serial dilutions of the antimicrobial down the rows of the plate (e.g., Column 1-12).
- Prepare 2X serial dilutions of the EPI across the columns of the plate (e.g., Row A-H). Include an EPI-free control column.
- Inoculate each well with a standardized bacterial suspension (~5 x 10^5 CFU/mL final concentration).
- Incubate at 35°C for 16-20 hours.
- Analysis: Determine the MIC (no growth) for each antimicrobial-EPI combination. Calculate the Fractional Inhibitory Concentration Index (FICI). FICI ≤ 0.5 indicates synergy and suggests efflux involvement. FICI = (MICantimicrobial+EPI / MICantimicrobial alone) + (MICEPI+antimicrobial / MICEPI alone).
Visualizations
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Experiment | Key Considerations |
|---|---|---|
| Phe-Arg-β-naphthylamide (PaβN) | Broad-spectrum EPI; used in MIC potentiation and accumulation assays to inhibit RND pumps. | Soluble in DMSO; can impact membrane integrity at high concentrations; use appropriate solvent controls. |
| Carbonyl Cyanide m-Chlorophenyl Hydrazone (CCCP) | Protonophore; dissipates proton motive force to confirm energy dependence of efflux. | Toxic to cells over time; light-sensitive; prepare fresh stock solutions in ethanol or DMSO. |
| Ethidium Bromide | Fluorescent efflux pump substrate; used in fluorometric accumulation assays as a model compound. | Carcinogen; requires safe handling and disposal. Alternative: Hoechst 33342 (DNA-binding). |
| Hepatocyte-Directed EPIs (e.g., MBX 3132) | Next-generation, potent EPIs with lower toxicity; used for proof-of-concept in combination therapy studies. | Often proprietary; evaluate cytotoxicity in eukaryotic cell lines. |
| Real-Time PCR Kits (qRT-PCR) | To quantify overexpression of efflux pump genes (e.g., acrB, mexB) in resistant isolates. | Requires high-quality RNA and appropriate reference genes (e.g., rpoD, gyrB). |
| Isogenic Mutant Strains (e.g., ΔacrB) | Genetic controls to definitively link phenotype to a specific pump; used in complementation assays. | Source from mutant collections (e.g., Keio, ASKA) or create via allelic exchange. |
| Cation-Adjusted Mueller Hinton Broth (CAMHB) | Standardized medium for MIC and checkerboard assays to ensure reproducibility. | Essential for consistent cation concentrations, which affect activity of some antibiotics and EPIs. |
| Fluorescence/Luminescence Plate Reader | For high-throughput intracellular accumulation assays and ATP-measurement assays. | Requires appropriate filters for chosen fluorescent probes (e.g., 530/600 nm for EtBr). |
The rise of multidrug-resistant (MDR) Gram-negative pathogens represents a critical threat to global health. A cornerstone of this resistance is the overexpression of Resistance-Nodulation-Division (RND) family efflux pumps, such as AcrAB-TolC in Escherichia coli and MexAB-OprM in Pseudomonas aeruginosa. These sophisticated tripartite systems extrude a broad spectrum of antibiotics, biocides, and host-derived molecules, significantly lowering intracellular drug concentrations and contributing to treatment failure. The development of efflux pump inhibitors (EPIs) has long been pursued as a strategy to revitalize existing antibiotics. However, progress toward clinical application has been stymied by a central, dual-faceted challenge: achieving potent inhibition while maintaining sufficient selectivity for the bacterial target over human host components. This whitepaper, framed within the broader thesis that targeting RND pumps is essential for overcoming Gram-negative resistance, provides an in-depth technical analysis of the toxicity-selectivity paradigm and outlines contemporary experimental approaches to navigate it.
The Molecular Basis of Toxicity and Selectivity
EPI toxicity often stems from a lack of selectivity, primarily through off-target interactions with human ABC (ATP-Binding Cassette) transporters or ion channels. Key structural and functional similarities between RND and eukaryotic transporters create a high risk for cross-reactivity.
Structural Homology and Off-Target Risks
RND pumps and human ABC transporters share a common functional principle: using energy to transport substrates across membranes. While their primary sequences differ, convergent evolution has led to analogous binding pockets for hydrophobic, amphipathic compounds. An EPI designed to bind the hydrophobic trap or substrate-binding pocket of AcrB may inadvertently bind to P-glycoprotein (P-gp, ABCB1), a critical human efflux pump expressed in the intestines, blood-brain barrier, and liver. Inhibition of P-gp can lead to increased systemic exposure to its numerous drug substrates, causing severe pharmacokinetic toxicities (e.g., neurotoxicity from chemotherapeutics).
Quantitative Data on Prominent EPI Candidates
The table below summarizes the efficacy and toxicity profiles of representative EPI candidates, illustrating the historical selectivity challenge.
Table 1: Profile of Select Efflux Pump Inhibitor Candidates
| EPI Candidate (Class) | Primary Target (Gram-negative) | In vitro Potency (IC₅₀/FR⁺) | Major Toxicity/Selectivity Issue | Clinical Status |
|---|---|---|---|---|
| Phe-Arg-β-naphthylamide (PAβN) (Peptidomimetic) | AcrAB-TolC, MexAB-OprM | IC₅₀: 2-10 µM; FR⁺: 4-64x | Disrupts mammalian membrane potential; cytotoxic at ~20 µM. | Research tool only |
| MBX-2319 (Pyranopyridine) | AcrB (Binds hydrophobic trap) | IC₅₀: ~0.1 µM; FR: >128x for CIP | Inhibits hERG ion channel (cardiotoxicity risk); moderate P-gp inhibition. | Preclinical (halted) |
| D13-9001 (Macrocyclic peptide) | MexB (Binds deep binding pocket) | IC₅₀: 0.02 µM; FR: >512x for levofloxacin | High molecular weight limits penetration; no major mammalian toxicity reported in vitro. | Preclinical |
| SPEP (Benzothiazole) | AcrB (Proton Motive Force disruptor?) | Reduces CIP MIC 16-fold at 25 µM | Cytotoxicity (CC₅₀) ~50 µM; narrow therapeutic window. | Research tool |
| NexEPI-1 (Recent candidate, structure undisclosed) | AcrAB-TolC | IC₅₀: 0.5 µM; FR: 32-128x | Selective >100x for AcrB over P-gp in co-assays. | Lead optimization |
FR = Fold Reduction in antibiotic MIC; CIP = Ciprofloxacin
Core Experimental Protocols for Assessing Toxicity and Selectivity
A robust pipeline for EPI development must integrate parallel assessment of antibacterial potentiation and mammalian cell toxicity early in the discovery process.
Protocol: Dual-Assay for EPI Potency and Cytotoxicity
Objective: To determine the fractional inhibitory concentration (FIC) of an EPI in combination with an antibiotic and its concomitant cytotoxicity in a mammalian cell line.
Materials:
- Bacterial strain: e.g., E. coli AG100 (AcrAB-TolC overexpressor).
- Mammalian cell line: e.g., HepG2 (human hepatocyte, expresses various ABC transporters) or HEK-293.
- Test antibiotic: e.g., Levofloxacin.
- Test EPI compound.
- Cell culture media (LB broth, DMEM + 10% FBS).
- 96-well sterile tissue culture-treated plates.
- AlamarBlue (Resazurin) cell viability reagent.
- Microplate reader (capable of 570/600 nm and 560/590 nm fluorescence).
Procedure: Part A: Checkerboard MIC Assay (CLSI M07)
- Dispense 50 µL of cation-adjusted Mueller-Hinton broth (CAMHB) into all wells of a 96-well plate.
- Add 50 µL of 2x concentrated levofloxacin solution to the top row (Column 1). Perform twofold serial dilutions across the plate (left to right).
- Add 50 µL of 2x concentrated EPI solution to Column A. Perform twofold serial dilutions down the plate.
- Add 50 µL of bacterial inoculum (5 x 10⁵ CFU/mL final) to all wells. The final volume is 150 µL. Include growth and sterility controls.
- Incubate at 37°C for 18-20 hours.
- Determine the MIC of levofloxacin alone and in combination with each EPI concentration. Calculate the FIC Index: FICI = (MICantibiotic+combo / MICantibiotic alone) + (MICEPI+combo / MICEPI alone). FICI ≤ 0.5 indicates synergy.
Part B: Parallel Mammalian Cytotoxicity Assay (ISO 10993-5)
- Seed HepG2 cells in a separate 96-well plate at 1 x 10⁴ cells/well in 100 µL complete DMEM. Incubate for 24 hrs (37°C, 5% CO₂).
- Prepare the same matrix of EPI and levofloxacin concentrations in DMEM (no phenol red) as in Part A. Note: Antibiotic concentrations will be far below cytotoxic levels.
- Aspirate media from cells and add 100 µL of the compound/media mixtures.
- Incubate for 24 hrs.
- Add 20 µL of AlamarBlue reagent per well. Incubate for 2-4 hours.
- Measure fluorescence (Ex 560/Em 590). Calculate cell viability as a percentage of untreated control wells.
- Determine the Selectivity Index (SI) for each synergistic EPI concentration: SI = CC₅₀ (concentration causing 50% mammalian cell death) / EPI concentration in the synergistic combination.
Protocol: Assessment of Human P-gp Inhibition
Objective: To quantify the inhibition potential of an EPI candidate against human P-glycoprotein. Method: Use the Caco-2 cell monolayer transport assay or a vesicular ATPase assay. Procedure (Vesicular Transport Assay):
- Obtain membrane vesicles expressing human P-gp (e.g., from insect cells).
- In a 96-well format, incubate vesicles with a known P-gp fluorescent substrate (e.g., N-methylquinidine, NMQ) in transport buffer (with MgATP) with or without the EPI candidate at varying concentrations. Include a control with a known P-gp inhibitor (e.g., verapamil).
- Stop the reaction with cold buffer. Transfer vesicles to a filter plate to capture them.
- Measure accumulated fluorescent substrate inside the vesicles. Inhibition of P-gp leads to decreased accumulation.
- Calculate IC₅₀ for P-gp inhibition. A desirable EPI candidate should have an IC₅₀ for P-gp >10-100x higher than its IC₅₀ for the bacterial RND target.
Visualizing the EPI Discovery and Selectivity Challenge
Diagram: EPI Development Pipeline & Toxicity Hurdles
Diagram: Off-Target Interactions of EPIs
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for EPI Selectivity Research
| Reagent / Material | Supplier Examples | Function in EPI Research |
|---|---|---|
| Overexpression Bacterial Strains (e.g., E. coli AG100, P. aeruginosa KPM111) | ATCC, KEIO Collection, Lab Stock | Provide a consistent, high-level expression of target RND pumps (AcrAB-TolC, MexAB-OprM) for potency screening. |
| Mammalian Cell Lines for Cytotoxicity (HepG2, HEK-293, Caco-2) | ATCC, ECACC | Assess general cell viability (HepG2, HEK-293) and specifically evaluate P-gp interaction/toxicity in polarized Caco-2 monolayers. |
| P-glycoprotein (ABCB1) Membrane Vesicles | Solvo Biotechnology, Corning Life Sciences | Enable high-throughput, cell-free vesicular transport assays to directly quantify human P-gp inhibition by EPI candidates. |
| hERG Inhibition Assay Kit (Non-cell based, binding) | Eurofins DiscoverX, Thermo Fisher | Screen for cardiotoxicity risk via interaction with the hERG potassium channel early in the development pipeline. |
| AlamarBlue (Resazurin) Cell Viability Reagent | Thermo Fisher, Bio-Rad, Sigma-Aldrich | Provide a simple, fluorometric/colorimetric endpoint for parallel bacterial growth and mammalian cytotoxicity assays. |
| Fluorescent Efflux Substrates (e.g., Hoechst 33342, Ethidium Bromide, Nile Red) | Thermo Fisher, Sigma-Aldrich | Functional assessment of efflux inhibition in real-time using fluorometry or flow cytometry (e.g., ethidium bromide accumulation assay). |
| Microplate Readers with Dual/Triple Monochromators | BMG Labtech, BioTek, Tecan | Essential for reading absorbance, fluorescence, and luminescence in high-throughput 96-/384-well assay formats. |
| Crystallized RND Pump Proteins (e.g., AcrB, MexB) | The Protein Data Bank (PDB) | Critical for structure-based drug design (SBDD) to model EPI binding and engineer selectivity against human homologs. |
The path to clinically viable EPIs requires a paradigm shift from solely pursuing potency to a balanced optimization of selectivity from the earliest stages. This involves the mandatory implementation of integrated assays like the dual potency-cytotoxicity protocol and specific off-target screening against human P-gp and hERG. Leveraging high-resolution structural data of both bacterial RND pumps and human off-targets for rational, computationally-guided design is paramount. The next generation of EPIs must be engineered not just as "inhibitors," but as precise molecular tools that exploit the subtle but critical differences between bacterial and eukaryotic transport machineries. Success in this endeavor will validate the central thesis of RND-targeted research and provide a powerful weapon in the fight against Gram-negative antimicrobial resistance.
Within the paradigm of antimicrobial resistance (AMR) in Gram-negative bacteria, the Resistance-Nodulation-Division (RND) superfamily of efflux pumps represents a cornerstone of intrinsic and acquired multidrug tolerance. A central thesis in contemporary research posits that the clinical failure of efflux pump inhibitors (EPIs) stems not from a lack of target engagement but from the profound functional redundancy and regulatory overlap between multiple RND systems expressed within a single pathogen. This whitepaper provides a technical guide to dissecting this redundancy, presenting current data, experimental protocols, and toolkits essential for developing strategies that bypass or broadly neutralize overlapping efflux.
Quantitative Landscape of Major RND Systems in Key Pathogens
The following table summarizes the primary RND systems, their regulatory components, and substrate profiles in model Gram-negative pathogens, based on recent genomic and phenomic studies.
Table 1: Major RND Efflux Systems in Clinically Relevant Gram-Negative Bacteria
| Pathogen | Primary RND Systems | Key Transcriptional Regulator(s) | Representative Substrates (Classes) | Genomic Context |
|---|---|---|---|---|
| Pseudomonas aeruginosa | MexAB-OprM, MexCD-OprJ, MexEF-OprN, MexXY-OprM | MexR, NfxB, MexT, MexZ | β-lactams, FQs, AGs, Tetracyclines, Tigecycline | Chromosomal, often core genome. |
| Escherichia coli | AcrAB-TolC | AcrR, MarA, Rob, SoxS, RamA | β-lactams, FQs, AGs, Chloramphenicol, dyes | Chromosomal, core genome. |
| Klebsiella pneumoniae | AcrAB-TolC, OqxAB, MdtEF | AcrR, RamA, OqxR, MdtR | β-lactams, FQs, Tigecycline, nitrofurans | Chromosomal and plasmid-borne (OqxAB). |
| Acinetobacter baumannii | AdeABC, AdeFGH, AdeIJK | AdeRS, AdeL, AdelR | β-lactams, AGs, Tigecycline, FQs, Chloramphenicol | Chromosomal, often overexpressed in MDR clones. |
| Salmonella enterica | AcrAB-TolC | AcrR, RamA, SoxS | β-lactams, FQs, AGs, azithromycin | Chromosomal, core genome. |
FQs: Fluoroquinolones; AGs: Aminoglycosides; MDR: Multidrug-Resistant.
Core Experimental Protocols for Studying Redundancy
Protocol: Competitive Fitness Assay in the Presence of Sub-Inhibitory Antimicrobials
Objective: To determine which specific RND system provides the primary fitness advantage under selective pressure when multiple systems are present.
Methodology:
- Generate isogenic deletion mutants (ΔpumpA, ΔpumpB, ΔpumpA/ΔpumpB*) in a wild-type background using allelic exchange with sacB counterselection.
- Grow wild-type and mutant strains overnight in cation-adjusted Mueller-Hinton broth (CAMHB).
- Mix strains at a 1:1:1:1 ratio (WT:ΔA:ΔB:ΔAB) in fresh CAMHB containing a sub-MIC (e.g., 1/4 or 1/8 MIC) of a relevant antibiotic (e.g., levofloxacin, tigecycline).
- Incubate at 37°C with shaking for 24 hours. Sample at T=0h and T=24h.
- Perform serial dilution and spot plating on selective media (with antibiotics marking specific mutants or LB agar with X-Gal for lacZ-based differentiation) to enumerate CFUs for each strain.
- Calculate the competitive index (CI) for each mutant relative to the wild type: CI = (CFUmutant at T24 / CFUWT at T24) / (CFUmutant at T0 / CFUWT at T0).
- Interpretation: A CI < 1 indicates a fitness defect. The mutant with the lowest CI identifies the most critical pump for that specific stress. Synergy is indicated if the double mutant's CI is significantly lower than the multiplicative effect of single deletions.
Protocol: qRT-PCR Analysis of Cross-Regulatory Networks
Objective: To map the transcriptional interplay between regulatory genes of different RND systems upon perturbation.
Methodology:
- Treatment: Expose mid-log phase bacterial cultures to: a) a known inducer of a specific pump (e.g., salicylate for marA), b) a novel putative EPI, c) a sub-MIC of an antibiotic, and d) a solvent control.
- RNA Extraction: At 30- and 60-minute post-exposure, stabilize culture with RNAprotect reagent, then extract total RNA using a column-based kit with on-column DNase I digestion.
- cDNA Synthesis: Use 1 µg of RNA and random hexamers with a reverse transcriptase kit. Include a no-reverse-transcriptase (-RT) control.
- qPCR: Design primers for key regulators (marA, ramA, soxS, adeR, mexR, nfxB) and the structural genes of their associated pumps (acrB, adeB, mexB). Use rpoD or gyrB as a housekeeping reference gene. Perform reactions in triplicate using SYBR Green master mix.
- Analysis: Calculate ∆∆Ct values. A >2-fold upregulation of a regulator not directly linked to the inducing agent suggests cross-talk within the regulatory network.
Visualizing Regulatory and Experimental Workflows
Diagram 1: RND Pump Cross-Regulation & Induction
Diagram 2: Competitive Fitness Assay Workflow
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for Efflux Redundancy Research
| Reagent / Material | Function / Application | Key Considerations |
|---|---|---|
| Phenylalanine-arginine β-naphthylamide (PAβN) | A broad-spectrum EPI used as a positive control in potentiation assays to confirm efflux-mediated resistance. | Chemically labile. Use fresh DMSO stocks. Toxic at high concentrations. Does not inhibit all RND pumps (e.g., MexAB-OprM). |
| MC-207,110 (L-Phe-L-Arg-β-naphthylamide) | The enantiomer of PAβN; a well-characterized EPI for P. aeruginosa Mex pumps. | Similar handling to PAβN. Useful for distinguishing pump specificity in competitive assays. |
| 1-(1-Naphthylmethyl)-piperazine (NMP) | A putative EPI for AcrAB-TolC, used to probe this system's contribution in Enterobacteriaceae. | Specificity is not absolute. May have other membrane effects. Use as a comparator, not a definitive tool. |
| Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) | A proton motive force (PMF) uncoupler. Used to distinguish between PMF-dependent (RND) and ATP-dependent efflux. | Highly toxic, induces rapid stress response. Use at low µM concentrations for short durations (≤30 min). |
| Real-Time PCR Master Mix (SYBR Green) | For qRT-PCR quantification of regulatory and operon gene expression changes. | Must include robust housekeeping gene controls (rpoD, gyrB) and no-reverse-transcriptase controls. |
| pEX18Tc or pKAS46 Suicide Vectors | For allelic exchange and creation of precise, markerless gene deletions in P. aeruginosa and other Gram-negatives. | Requires sucrose sensitivity (sacB gene) in host strain for counterselection. |
| Ethidium Bromide (EtBr) or Hoechst 33342 Accumulation Assay | Fluorescent substrates for direct, real-time measurement of efflux activity via fluorometry or flow cytometry. | EtBr is a mutagen. Hoechst 33342 requires UV excitation. Baseline accumulation varies; always include a CCCP control to define maximum accumulation. |
Within the critical field of Gram-negative bacterial resistance research, Resistance-Nodulation-Division (RND) efflux pumps stand as a principal mechanism of multidrug resistance. These tripartite systems, exemplified by AcrAB-TolC in Escherichia coli, actively extrude a wide range of antimicrobials. The central thesis framing this guide posits that overcoming RND-mediated resistance requires a dual-optimization strategy: compounds must achieve potent target inhibition and circumvent efflux by successfully penetrating the formidable Gram-negative outer membrane (OM) and avoiding efflux pump recognition. This whitepaper provides an in-depth technical exploration of the strategies and methodologies to balance these often-competing molecular properties.
The Dual Barriers: Outer Membrane & Efflux Pumps
The Gram-negative envelope presents two sequential, synergistic barriers.
- Outer Membrane (OM): An asymmetric bilayer with a dense, negatively charged lipopolysaccharide (LPS) leaflet. Primary penetration pathways are: 1) Hydrophobic Pathway: for small, non-polar molecules through lipid domains. 2) Porin Pathway: for small, hydrophilic molecules (<600 Da) through water-filled channels like OmpF/C.
- RND Efflux Pumps: Spanning both inner and outer membranes, they capture substrates from the periplasm and inner membrane leaflet, expelling them directly to the exterior. Their broad, flexible substrate-binding pockets recognize amphiphilic or hydrophobic molecules.
The optimization challenge is clear: enhancing hydrophobicity can improve OM permeation via the lipid pathway but often increases efflux pump recognition. Conversely, increasing hydrophilicity may reduce efflux but hinder OM traversal.
Quantitative Parameters for Optimization
Key physicochemical and molecular descriptors must be measured and balanced. Recent literature and databases provide benchmark values.
Table 1: Key Molecular Descriptors for Permeation-Efflux Balance
| Descriptor | Target Range (Optimal) | Impact on OM Permeation | Impact on RND Efflux | Preferred Assay |
|---|---|---|---|---|
| LogD7.4 | 0 to 2 (Lipinski: -0.4 to 5.6) | Moderate LogD favors porin pathway; high LogD favors lipid pathway. | High LogD (>2.5) strongly correlates with efflux susceptibility. | Shake-flask HPLC/UV |
| Molecular Weight (Da) | < 600 (ideally < 500) | Critical for porin-mediated uptake. Larger molecules are excluded. | Larger, flexible molecules are common substrates. | Calculated |
| Topological Polar Surface Area (tPSA, Ų) | < 140 (ideally 60-90) | Lower tPSA favors passive diffusion through lipid bilayer. | Indirect correlation; high tPSA often reduces membrane interaction. | Calculated (e.g., RDKit) |
| Net Charge at pH 7.4 | Neutral or Zwitterionic | Cationic compounds may interact with negatively charged LPS; neutrals diffuse more readily. | Cationic compounds are often poorer AcrB substrates. | Calculated (pKa) |
| Rigid Bond Fraction | Higher may be beneficial | Not directly correlated. | Increased flexibility is associated with efflux pump recognition. | Calculated |
Table 2: Experimental Permeation & Efflux Data for Reference Compounds (Hypothetical Data Based on Current Trends)
| Compound | cLogD7.4 | MW (Da) | tPSA (Ų) | OM Permeability (Papp x10-6 cm/s)* | Efflux Ratio (B-A/A-B) | IC50 Target (µM) |
|---|---|---|---|---|---|---|
| Tetracycline | -1.3 | 444.4 | 182 | 0.8 | 15.2 | 0.5 |
| Levofloxacin | -0.4 | 361.4 | 75 | 12.5 | 4.1 | 0.1 |
| Novel Inhibitor A | 1.2 | 480.3 | 85 | 8.7 | 2.5 | 0.05 |
| Novel Inhibitor B | 3.5 | 520.6 | 50 | 15.2 | 22.1 | 0.02 |
Measured in asymmetric OM vesicle assay. *Caco-2 or MDCK cell assay; Ratio >3 indicates significant efflux.
Experimental Protocols
Protocol: Asymmetric Outer Membrane Vesicle (OMV) Permeability Assay
Objective: Quantify compound translocation across a model asymmetric OM. Reagents: E. coli strain with hypervesiculating phenotype (e.g., ΔtolRA), Sucrose, Tris-EDTA buffer, Polymyxin B nonapeptide (PMBN, optional porin permeabilizer). Method:
- Harvest OMVs from culture supernatant via ultracentrifugation (150,000 x g, 2h).
- Load OMVs with a self-quenching fluorescent dye (e.g., carboxyfluorescein) by electroporation.
- Incubate dye-loaded OMVs with test compound. Compound permeation disrupts vesicles, causing dye dequenching.
- Measure fluorescence increase (Ex/Em: 492/517 nm) over time. Use Triton X-100 lysis for 100% signal.
- Calculate apparent permeability (Papp) from initial rate. Interpretation: High Papp indicates good OM penetration. Compare rates in presence/absence of PMBN to distinguish porin vs. lipid pathway dominance.
Protocol: Direct RND Efflux Pump Inhibition & Substrate Competition Assay
Objective: Determine if a compound is an efflux substrate or a pump inhibitor. Reagents: E. coli strain expressing a specific RND pump (e.g., AcrAB-TolC) and an isogenic knockout (ΔacrB), fluorescent probe substrate (e.g., ethidium bromide, Hoechst 33342), efflux inhibitor (e.g., PAβN). Method (Microplate Accumulation Assay):
- Grow bacteria to mid-log phase, wash, and resuspend in buffer with energy source (e.g., glucose).
- Pre-incubate cells with or without test compound (or PAβN control) for 10 min.
- Add fluorescent probe. Monitor intracellular fluorescence (e.g., EtBr: Ex/Em 530/600 nm) kinetically for 30 min.
- Analysis: Compare final fluorescence in: a) Wild-type + probe, b) Wild-type + test compound + probe, c) ΔacrB + probe. Interpretation: Increased fluorescence in (b) vs. (a) suggests test compound competes with/inhibits efflux. Low fluorescence in (a) but high in (c) confirms efflux activity.
Protocol: Whole-Cell Potency Shift Assay
Objective: Quantify the net impact of permeation and efflux on whole-cell activity. Reagents: Isogenic bacterial strains: Wild-type, OM-permeability mutant (e.g., ΔlpxM or PMBN-treated), Efflux-pump knockout (ΔacrB), Efflux-pump overexpressor. Method:
- Perform standard broth microdilution MIC testing against all strains in parallel.
- Calculate fold-change in MIC. Interpretation: MIC(WT) >> MIC(ΔacrB) = compound is an efflux substrate. MIC(WT) high but reduced by PMBN = OM penetration is limiting. The ideal compound shows minimal MIC shift across all strains.
Visualizing Strategies and Pathways
Diagram 1: Compound Journey and Efflux Threat in Gram-Negative Cell
Diagram 2: Optimization Workflow for Permeation-Efflux Balance
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Permeation & Efflux Studies
| Reagent / Material | Function & Application | Key Consideration |
|---|---|---|
| Polymyxin B Nonapeptide (PMBN) | A cationic peptide that selectively disrupts LPS, increasing OM permeability without cell lysis. Used to assess OM barrier contribution. | Use sub-lytic concentrations (typically 10-50 µg/mL) to permeabilize without killing. |
| Phenylalanine-Arginine β-Naphthylamide (PAβN) | A broad-spectrum competitive inhibitor of RND pumps (e.g., AcrB). Used as a positive control in efflux inhibition assays. | May have off-target effects at high concentrations; use as a comparator, not a therapeutic lead. |
| Ethidium Bromide (EtBr) | A classic fluorescent efflux pump substrate. Used in real-time accumulation assays to quantify efflux activity/inhibition. | Handle as a mutagen; use alternative probes (e.g., Hoechst 33342) for safer HTS. |
| Asymmetric OMV Preparation Kit | Commercial kits (e.g., from companies like Nymirum) provide standardized, reproducible OMVs for permeability screening. | Ensures consistent LPS composition and asymmetry, critical for predictive data. |
| Caco-2 / MDCK Cell Lines | Mammalian epithelial cell lines expressing endogenous efflux transporters (e.g., P-gp). Used to predict compound efflux liability. | Provides translational data but is not a direct model for bacterial OM permeation. |
| Isogenic Bacterial Strain Panels | Custom or commercial panels (e.g., WT, Δporin, ΔacrB, ΔtolC, overexpressors) are essential for dissecting individual barrier contributions. | Genotypic validation via sequencing is required to confirm mutations. |
| Surface Plasmon Resonance (SPR) with Purified AcrB | Directly measures compound binding kinetics to the efflux pump protein, distinguishing substrates from inhibitors. | Requires purified, stable, and functionally reconstituted AcrB protein. |
The overexpression of Resistance-Nodulation-Division (RND) efflux pumps is a cornerstone of multidrug resistance in Gram-negative bacteria. Within the broader thesis on RND-mediated resistance, a critical challenge is the inadvertent induction of resistance during experimental investigation or therapeutic intervention. This guide details strategies to prevent compensatory mutations or regulatory overexpression that can confound research results and accelerate therapeutic failure.
Core Strategies to Prevent Resistance Induction
Strategies focus on minimizing selective pressure and directly modulating regulatory networks.
Minimizing Sub-inhibitory Selective Pressure
Exposure to sub-inhibitory concentrations of antibiotics or efflux pump substrates is a potent inducer of regulatory mutations and overexpression.
- Strategy: Utilize precise, lethal dosing regimens in experiments and consider combination therapies to reduce the emergence of resistant clones.
- Key Data: The relationship between antibiotic concentration and mutation frequency.
Table 1: Mutation Frequency Relative to Antibiotic Concentration
| Antibiotic | Sub-MIC (Fraction of MIC) | Mutation Frequency (CFU/mL) | Primary Resistance Mechanism Induced |
|---|---|---|---|
| Ciprofloxacin | 0.25x MIC | 3.2 x 10⁻⁸ | Target mutation (gyrA) & Efflux upregulation |
| Tetracycline | 0.5x MIC | 1.7 x 10⁻⁷ | RND pump (acrAB-tolC) overexpression |
| Chloramphenicol | 0.25x MIC | 5.8 x 10⁻⁸ | Regulatory mutation (marR) |
Targeting Master Regulatory Circuits
Preventing the overexpression of RND pumps requires inhibition of the regulatory cascades that control them.
- Primary Target: The marRAB operon. Inhibition of MarA activity or stabilization of the repressor MarR prevents upregulation of acrAB-tolC.
- Secondary Target: The soxRS system, activated by oxidative stress. Use of antioxidants (e.g., Thiourea) in growth media can mitigate this pathway.
Table 2: Key Regulatory Systems Controlling RND Pump Expression
| Regulatory System | Inducing Signal | Key Effector | Target Efflux Pump Operon |
|---|---|---|---|
| marRAB | Salicylate, antibiotics | MarA (activator) | acrAB-tolC, tolC |
| soxRS | Superoxide, redox cyclers | SoxS (activator) | acrAB-tolC, micF |
| rob | Bile salts, decanoate | Rob (activator) | acrAB-tolC |
| acrR | Efflux pump dysfunction | AcrR (repressor) | acrAB |
Diagram 1: MarRAB Regulatory Pathway Leading to Efflux Overexpression
Experimental Protocols for Monitoring Induction
Protocol: Quantifying Efflux Pump Expression via RT-qPCR
Objective: Measure changes in acrB, tolC, marA, and soxS mRNA levels upon experimental treatment.
- Bacterial Growth: Grow test strain (e.g., E. coli K-12) to mid-log phase (OD₆₀₀ ≈ 0.5) in appropriate broth.
- Treatment: Split culture. Treat one with sub-MIC of test compound (e.g., 0.25x MIC tetracycline) for 60 minutes. Keep one as untreated control.
- RNA Stabilization & Extraction: Add 2 volumes of RNAprotect Bacteria Reagent. Incubate 5 min, pellet. Extract RNA using a commercial kit with on-column DNase I digestion.
- cDNA Synthesis: Use 1 µg RNA and random hexamers with a reverse transcription system.
- qPCR: Prepare reactions with SYBR Green Master Mix, gene-specific primers, and cDNA template. Use rpoD or gyrB as reference genes.
- Analysis: Calculate fold-change using the 2^(-ΔΔCt) method.
Protocol: Competitive Fitness Assay to Detect Compensatory Mutations
Objective: Identify if a resistance-impairing mutation (e.g., acrB knockout) is compensated during passage.
- Strain Preparation: Create a 1:1 mixture of marked mutant (e.g., ΔacrB::KanR) and wild-type (WT) isogenic strain.
- Passaging: Dilute mixture 1:1000 into fresh medium daily for 5-7 days, with/without sub-inhibitory effector.
- Plating & Enumeration: Plate serial dilutions daily on non-selective and antibiotic-containing media.
- Calculation: Compute the competitive index (CI) = (Mutant CFU / WT CFU) at time T ÷ (Mutant CFU / WT CFU) at time 0. A CI trending toward 1 indicates compensatory evolution.
Diagram 2: Competitive Fitness Assay Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Induction Avoidance Studies
| Reagent / Material | Function / Purpose | Example Product/Catalog |
|---|---|---|
| Phe-Arg-β-naphthylamide (PAβN) | Broad-spectrum efflux pump inhibitor; used to confirm efflux-mediated resistance and reduce MICs. | Sigma-Aldrich, P4157 |
| RNAprotect Bacteria Reagent | Immediately stabilizes bacterial RNA profiles at the time of sampling, preventing artifactual changes. | Qiagen, 76506 |
| MarR Ligands (e.g., Salicylate) | Positive control inducer of the mar regulon for assay validation. | Sodium salicylate, S3007 |
| Triclosan | Specific inhibitor of FabI; used to study mar-mediated cross-resistance to biocides. | Sigma-Aldrich, 72779 |
| Chromosomally Integrated Transcriptional Reporters | Strains with gfp/lacZ fused to promoters of acrAB or marA for real-time induction monitoring. | Keio collection derivatives |
| MIC Test Strips (Gradient Strips) | For determining precise, reproducible MICs and sub-MIC values for dosing experiments. | Liofilchem, MTS |
| Tetrazolium Red (TTC) | Metabolic dye used in agar plates to visualize resistant colonies in population assays. | Sigma-Aldrich, 298-96-4 |
| Custom siRNA/antisense oligonucleotides | For targeting specific regulatory mRNA transcripts in bacterial gene expression knockdown studies. | Custom synthesis from IDT |
Efflux pump inhibitors (EPIs) represent a promising therapeutic strategy to combat multidrug-resistant Gram-negative bacteria, primarily by targeting Resistance-Nodulation-Division (RND) superfamily pumps such as AcrAB-TolC in Escherichia coli and MexAB-OprM in Pseudomonas aeruginosa. The standardization of assays to evaluate EPI efficacy is a critical, unmet need in the field. Inconsistent methodologies across laboratories lead to irreproducible data, hindering the direct comparison of novel EPI compounds and their progression into the drug development pipeline. This whitepaper provides a technical guide for standardizing key EPI assays to enhance inter-laboratory reproducibility, framed within the broader thesis that targeting RND efflux is a viable approach to restoring antibiotic efficacy.
Core Standardized Assay Methodologies
Broth Microdilution Checkerboard Assay for Minimum Inhibitory Concentration (MIC) Modulation
This gold-standard assay quantifies the potentiation of an antibiotic by an EPI.
Detailed Protocol:
- Prepare Compounds: Prepare 2x serial dilutions of the antibiotic (e.g., levofloxacin, erythromycin) in cation-adjusted Mueller-Hinton broth (CAMHB) in a 96-well plate along the vertical axis. Prepare 2x serial dilutions of the EPI candidate along the horizontal axis. Include antibiotic-only and EPI-only controls.
- Inoculate: Dilute a log-phase bacterial suspension (e.g., P. aeruginosa PAO1) to ~5 x 10⁵ CFU/mL in CAMHB. Add an equal volume of this inoculum to each well, resulting in a final bacterial density of ~5 x 10⁵ CFU/mL and 1x concentrations of all compounds.
- Incubate: Incubate plates at 35°C ± 2°C for 16-20 hours under static conditions.
- Readout: The MIC is defined as the lowest concentration of antibiotic that completely inhibits visible growth. The fractional inhibitory concentration index (FICI) is calculated as: FICᵢ = (MIC of drug A in combination / MIC of drug A alone) FICI = FICₐ + FICᵦ Interpretation: Synergy (FICI ≤ 0.5), Additivity/Indifference (0.5 < FICI ≤ 4), Antagonism (FICI > 4).
Ethidium Bromide (EtBr) Accumulation Assay
A functional assay measuring real-time efflux pump activity. Reduced efflux due to EPI presence leads to increased intracellular accumulation of fluorescent substrates like EtBr.
Detailed Protocol:
- Cell Preparation: Grow bacteria to mid-log phase (OD₆₀₀ ≈ 0.5). Harvest cells, wash twice in PBS or HEPES buffer (pH 7.0), and resuspend to an OD₆₀₀ of 0.2 in buffer containing glucose (0.4% w/v) as an energy source.
- Loading and Baseline: Transfer suspension to a quartz cuvette or black microplate. Add EtBr to a final concentration of 1-5 µM. Monitor fluorescence (excitation: 530 nm, emission: 600 nm) for 5-10 minutes to establish a passive accumulation baseline.
- Energy-Driven Efflux: Add glucose (if not pre-added) to energize the cells, initiating active efflux. Fluorescence will decrease as EtBr is pumped out.
- EPI Inhibition Test: At the plateau of efflux, add the candidate EPI. A functional EPI will inhibit efflux, causing a rapid increase in fluorescence due to resumed intracellular accumulation.
- Controls: Include a negative control (no glucose/energy inhibitor like CCCP) showing maximal accumulation, and a positive control (no EPI) showing sustained efflux.
Real-Time PCR for Efflux Pump Gene Expression
Standardized RNA isolation and qPCR to assess if an EPI or treatment condition modulates RND pump gene expression.
Detailed Protocol:
- RNA Isolation: Treat bacterial cultures with sub-inhibitory concentrations of EPI/antibiotic. At designated timepoints, stabilize RNA immediately using a reagent like RNAprotect. Extract total RNA using a column-based kit with on-column DNase I digestion.
- cDNA Synthesis: Quantify RNA. Use 500 ng - 1 µg of RNA for reverse transcription with random hexamers and a reverse transcriptase enzyme.
- qPCR: Design primers with high efficiency (90-110%) for target genes (acrB, mexB, adeB) and reference housekeeping genes (rpoD, proC, gyrB). Use a SYBR Green master mix. Run reactions in technical triplicates. Cycling conditions: initial denaturation (95°C, 2 min); 40 cycles of 95°C for 15 sec and 60°C for 1 min; followed by a melt curve.
- Analysis: Calculate ΔΔCq to determine fold-change in gene expression relative to an untreated control, normalized to reference genes.
Table 1: Standardized Parameters for Core EPI Assays
| Assay | Key Parameter Measured | Primary Output Metric | Critical Standardized Conditions |
|---|---|---|---|
| Checkerboard MIC | Antibiotic potentiation | Fractional Inhibitory Concentration Index (FICI) | Medium: CAMHB; Inoculum: 5e5 CFU/mL; Incubation: 35°C, 16-20h |
| EtBr Accumulation | Real-time efflux inhibition | Fold-increase in fluorescence rate/slope post-EPI addition | Buffer: PBS/HEPES + Glucose; Cell Density: OD₆₀₀ 0.2; EtBr: 2.5 µM; Temp: 37°C |
| RT-qPCR | Efflux pump gene expression | Fold-change (ΔΔCq) vs. untreated control | RNA Stabilization: <5 min delay; Reference Genes: ≥2 stable genes; Primer Efficiency: 90-110% |
| Table 2: Interpretation Guidelines for FICI and Accumulation Data | |||
| FICI Value | Interpretation | Recommended Action for EPI Lead | |
| ≤ 0.5 | Synergy | Proceed to in vivo models | |
| 0.5 - 1.0 | Additive | Optimize compound/combination | |
| 1.0 - 4.0 | Indifferent | Consider alternative mechanisms | |
| > 4.0 | Antagonism | Discontinue candidate |
The Scientist's Toolkit: Essential Reagent Solutions
Table 3: Key Research Reagent Solutions for Standardized EPI Assays
| Item | Function & Rationale |
|---|---|
| Cation-Adjusted Mueller Hinton Broth (CAMHB) | Standardized growth medium for MIC assays; ensures consistent cation concentrations (Ca²⁺, Mg²⁺) which affect antibiotic activity, especially for polymyxins. |
| HEPES Buffer (pH 7.0) with 0.4% Glucose | Standard buffer for accumulation assays; provides stable pH and energy source for active efflux. |
| Ethidium Bromide (EtBr) Stock Solution | Fluorescent efflux pump substrate for functional assays. Standard concentration (e.g., 10 mM in DMSO) ensures inter-assay comparability. Handle as mutagen. |
| Carbonyl Cyanide m-Chlorophenyl Hydrazone (CCCP) | Protonophore used as a positive control in accumulation assays; collapses proton motive force, fully inhibiting energy-dependent efflux. |
| RNA Stabilization Reagent (e.g., RNAprotect) | Critical for gene expression studies; immediately stabilizes RNA in situ to prevent degradation and transcriptional changes during sample processing. |
| Validated qPCR Primers for RND & Housekeeping Genes | Pre-validated primer sets with published efficiencies for target organisms ensure accurate, reproducible quantification of gene expression across labs. |
Visualizing Workflows and Mechanisms
EPI Inhibition of RND Pump Function
Standardized EtBr Accumulation Assay Workflow
FICI Calculation and Interpretation Pathway
Benchmarking RND Pumps: Efficacy, Prevalence, and Place in the Resistance Arsenal
Within the escalating crisis of antimicrobial resistance, Resistance-Nodulation-Division (RND) efflux pumps represent a cornerstone mechanism of multidrug resistance in Gram-negative priority pathogens. This whitepaper, framed within a broader thesis on RND efflux pump research, details the prevalence, clinical consequences, and experimental methodologies pertinent to Carbapenem-Resistant Acinetobacter baumannii (CRAB), Carbapenem-Resistant Enterobacterales (CRE), and Multidrug-Resistant (MDR) Pseudomonas aeruginosa. RND pumps, such as AdeABC in A. baumannii, AcrAB-TolC in Enterobacterales, and MexAB-OprM in P. aeruginosa, contribute significantly to intrinsic and acquired resistance, complicating therapeutic interventions and patient outcomes.
Prevalence of Key RND Pumps in Priority Pathogens
The table below summarizes the primary RND pumps, their regulatory components, and prevalence data from recent global surveillance studies.
Table 1: Prevalence and Characteristics of Major RND Pumps in Priority Pathogens
| Pathogen Group | Primary RND Pump System | Key Regulator(s) | Common Substrates | Reported Prevalence in Clinical Isolates* | Geographical Hotspots |
|---|---|---|---|---|---|
| CRAB | AdeABC | AdeRS | Aminoglycosides, Tetracyclines, β-lactams, Chloramphenicol, Fluoroquinolones | 50-80% in MDR/CRAB clones | Asia, Middle East, North America |
| CRE (e.g., K. pneumoniae) | AcrAB-TolC | AcrR, MarA, SoxS, RamA | β-lactams (including carbapenems), Fluoroquinolones, Chloramphenicol, Macrolides, Novobiocin | >90% (intrinsic); Overexpression in 60-70% of MDR isolates | Worldwide |
| MDR P. aeruginosa | MexAB-OprM | MexR, NalC, NalD | β-lactams (excluding imipenem), Fluoroquinolones, Chloramphenicol, Trimethoprim, Sulfonamides | 30-50% in nosocomial MDR isolates | Europe, North America, Asia |
| P. aeruginosa (notable) | MexXY-OprM | MexZ, PA5471 | Aminoglycosides, Tetracyclines, Fluoroquinolones, Cephalosporins | Overexpressed in 40-60% of aminoglycoside-resistant strains | Worldwide |
*Prevalence data indicates the proportion of clinical isolates showing overexpression or clinically relevant upregulation of the pump system, contributing to the resistance phenotype.
Clinical Impact and Patient Outcomes
RND pump overexpression directly correlates with worse clinical outcomes. It leads to higher minimum inhibitory concentrations (MICs) across multiple drug classes, resulting in:
- Initial therapeutic failure: Increased likelihood of inappropriate empirical therapy.
- Reduced treatment options: Limits effective antibiotics, forcing use of more toxic or less efficacious agents (e.g., colistin).
- Increased mortality and costs: Associated with longer hospital stays, higher treatment costs, and increased mortality rates, particularly in bloodstream and respiratory infections.
Table 2: Impact of RND Pump Overexpression on Key Antibiotic MICs
| Antibiotic Class | Example Drug | Typical MIC (Susceptible) | MIC with RND Overexpression (Fold Increase) | Clinical Consequence |
|---|---|---|---|---|
| Fluoroquinolones | Ciprofloxacin | ≤1 µg/mL | 4-32 µg/mL (4-32x) | Resistance, treatment failure |
| β-lactams | Piperacillin | ≤16 µg/mL | 64-512 µg/mL (4-32x) | Reduced susceptibility or resistance |
| Tetracyclines | Tigecycline | ≤2 µg/mL | 4-16 µg/mL (2-8x) | Breakthrough resistance |
| Aminoglycosides | Amikacin | ≤8 µg/mL | 16-64 µg/mL (2-8x) | Resistance in combination therapy |
Core Experimental Protocols for RND Pump Research
Protocol: Quantitative Assessment of Efflux Pump Activity (Real-Time Fluorometric Assay)
Objective: To measure real-time efflux pump activity using a fluorescent substrate. Materials: Bacterial culture, appropriate broth medium, fluorescent efflux substrate (e.g., ethidium bromide, Hoechst 33342), efflux pump inhibitor (e.g., Phe-Arg-β-naphthylamide, PABN), microplate reader with temperature control and appropriate filters. Procedure:
- Grow bacteria to mid-log phase (OD600 ~0.5).
- Harvest cells by centrifugation (3,500 x g, 10 min), wash twice with assay buffer (e.g., PBS or HEPES with 5 mM glucose).
- Resuspend cells to an OD600 of 0.2 in assay buffer.
- Inhibitor Control: Pre-incubate an aliquot of cells with 50 µg/mL PABN for 10 minutes.
- Load both inhibited and non-inhibited cell suspensions with 1 µM ethidium bromide in a black, clear-bottom 96-well plate.
- Immediately place plate in a pre-warmed (37°C) fluorescence microplate reader.
- Monitor fluorescence (Excitation: 530 nm, Emission: 590 nm) every 30 seconds for 60 minutes.
- Data Analysis: The initial rate of fluorescence decrease after energy poisoning (e.g., with 10 mM sodium azide added at 30 min) indicates active efflux. Compare initial rates between PABN-treated and untreated cells.
Protocol: Gene Expression Analysis of RND Pump Operons (RT-qPCR)
Objective: To quantify the expression levels of RND pump and regulator genes. Materials: RNA stabilization reagent, RNA extraction kit, DNase I, cDNA synthesis kit, SYBR Green qPCR master mix, gene-specific primers, real-time PCR system. Procedure:
- Stabilize RNA from bacterial cultures at desired growth phase using an appropriate reagent.
- Extract total RNA, treat with DNase I to remove genomic DNA contamination.
- Quantify RNA purity and concentration using a spectrophotometer.
- Synthesize cDNA from 1 µg of total RNA using a reverse transcription kit.
- Prepare qPCR reactions in triplicate: 10 µL SYBR Green master mix, 1 µL each of forward and reverse primer (10 µM), 2 µL cDNA template (diluted 1:10), 6 µL nuclease-free water.
- Run qPCR: Initial denaturation (95°C, 2 min); 40 cycles of denaturation (95°C, 15 sec) and annealing/extension (60°C, 1 min); followed by a melt curve analysis.
- Data Analysis: Calculate relative gene expression using the 2^(-ΔΔCt) method, normalizing target gene Ct values to a stable housekeeping gene (e.g., rpoB, gyrB) and comparing to an appropriate control strain.
Visualizing RND Pump Regulation and Workflow
Diagram 1: RND Pump Regulatory Pathways and Expression Workflow This diagram illustrates common pathways leading to RND pump overexpression and the standard RT-qPCR workflow for measuring expression.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for RND Pump Research
| Item | Function/Application | Example/Note |
|---|---|---|
| Efflux Pump Inhibitors (EPIs) | Chemical tools to block pump activity, used in combination assays to confirm pump-mediated resistance. | Phe-Arg-β-naphthylamide (PABN, broad spectrum), 1-(1-naphthylmethyl)-piperazine (NMP) for AcrAB, MC-207,110. |
| Fluorescent Efflux Substrates | Probe molecules used in real-time fluorometric assays to measure active efflux. | Ethidium Bromide, Hoechst 33342, Nile Red, Pyronin Y. |
| Gene Expression Kits | For quantifying mRNA levels of pump and regulator genes. | RNAprotect, RNeasy kits (Qiagen), Superscript IV cDNA synthesis, SYBR Green qPCR master mixes. |
| Cation-Adjusted Mueller Hinton Broth (CAMHB) | Standardized medium for antimicrobial susceptibility testing (AST) and efflux-related MIC determinations. | Essential for reproducible MIC and checkerboard assays with EPIs. |
| Polyclonal/Monoclonal Antibodies | Detect and quantify pump protein expression via Western Blot or ELISA. | Anti-AcrA, Anti-MexB, Anti-AdеB antibodies (commercial and research-grade). |
| Clinical Isolate Panels | Characterized strains for validating findings and comparative studies. | ATCC strains, WHO/CDC reference strains, well-characterized clinical MDR isolates with known pump profiles. |
| Bioinformatics Tools | Analyze sequences for mutations in regulatory genes and pump components. | CLC Genomics Workbench, Geneious, ARG-ANNOT, ResFinder, custom BLAST databases. |
Within the critical context of Gram-negative bacterial resistance, Resistance-Nodulation-Division (RND) efflux pumps constitute a primary defensive frontier. These tripartite complexes, spanning the inner membrane, periplasm, and outer membrane, actively extrude a diverse array of antimicrobials, biocides, and host-derived molecules. This whitepaper provides an in-depth technical comparison of the three most clinically significant RND families: Escherichia coli AcrAB-TolC, Pseudomonas aeruginosa MexAB-OprM, and Acinetobacter baumannii AdeABC. Understanding their distinct operational profiles is paramount for designing novel efflux pump inhibitors (EPIs) and adjuvant therapies.
Structural & Genetic Organization: A Foundational Comparison
While sharing a core tripartite architecture, these systems exhibit key genetic and regulatory differences that influence their expression and clinical impact.
Table 1: Core Genetic and Regulatory Characteristics
| Characteristic | AcrAB-TolC (E. coli) | MexAB-OprM (P. aeruginosa) | AdeABC (A. baumannii) |
|---|---|---|---|
| Regulator Gene | acrR, marR, soxR, rob | mexR (NalC/NalD) | adeRS (Two-Component System) |
| Regulation Mode | Local repressors & global stress regulators (MarA, SoxS, Rob) | Transcriptional repressors (MexR, NalC, NalD) | Sensor kinase (AdeS) & response regulator (AdeR) |
| Genetic Locus | Chromosomal (acrAB-tolC分散) | Chromosomal (mexAB-oprM operon) | Chromosomal (adeABC operon + adeRS operon) |
| Induction Signals | Antibiotics, bile salts, solvents, oxidative stress | Antibiotics, solvents, quorum-sensing signals | Tigecycline, β-lactams, disinfectants (e.g., chlorhexidine) |
Title: Comparative Regulatory Pathways of Major RND Pumps
Substrate Profile & Efflux Efficiency: Quantitative Analysis
Substrate promiscuity (breadth) and efflux efficiency (antimicrobial potency impact) vary significantly between families. The following data is synthesized from recent minimum inhibitory concentration (MIC) shift assays and ethidium bromide accumulation studies.
Table 2: Comparative Substrate Profiles and Efflux Impact
| Antimicrobial Class | Specific Agent | AcrAB-TolC Impact (Fold MIC Change Δ/+)* | MexAB-OprM Impact (Fold MIC Change Δ/+)* | AdeABC Impact (Fold MIC Change Δ/+)* |
|---|---|---|---|---|
| β-lactams | Carbenicillin | 4-8x | 16-64x | 2-4x |
| Cefepime | 2x | 8-16x | 4-8x | |
| Fluoroquinolones | Ciprofloxacin | 8-32x | 8-16x | 2-4x |
| Levofloxacin | 4-16x | 4-8x | 2x | |
| Tetracyclines | Tetracycline | 8-16x | 2-4x | 4-8x |
| Tigecycline | 2x | 2x | 8-32x | |
| Aminoglycosides | Gentamicin | - | - | - |
| Macrolides | Erythromycin | 32-64x | 4-8x | 16-32x |
| Chloramphenicol | Chloramphenicol | 16-32x | 4-8x | 4x |
| Disinfectants | Chlorhexidine | 4x | 2-4x | 8-16x |
| Dyes | Ethidium Bromide | 64-128x | 16-32x | 32-64x |
* Fold increase in MIC in wild-type vs. isogenic efflux pump knockout strain.
Key Experimental Protocols for Characterization
Protocol 1: Ethidium Bromide Accumulation Assay (Efflux Activity Measurement)
- Principle: Measures real-time intracellular accumulation of the fluorescent substrate ethidium bromide (EtBr). Inhibited efflux leads to faster fluorescence increase.
- Procedure:
- Cell Preparation: Grow bacterial strains to mid-log phase (OD600 ~0.5). Harvest, wash, and resuspend in assay buffer (e.g., PBS or minimal medium) with glucose (0.2% as energy source).
- Loading: Incubate cells with EtBr (1-2 µg/mL) in the presence of a protonophore (e.g., CCCP, 50 µM) for 30 min at 37°C to de-energize pumps and allow passive dye influx. Wash cells to remove CCCP and external dye.
- Efflux Initiation: Resuspend cells in glucose-containing buffer. Immediately aliquot into a black 96-well plate.
- Kinetic Reading: Measure fluorescence (Excitation: 530 nm, Emission: 600 nm) every 1-2 minutes for 30-60 minutes using a plate reader at 37°C.
- Data Analysis: Plot fluorescence vs. time. The initial slope represents efflux activity. Compare slopes between wild-type, knockout mutants, and EPI-treated samples.
Protocol 2: Minimum Inhibitory Concentration (MIC) Shift Assay
- Principle: Determines the contribution of an efflux pump to resistance by comparing MICs in a wild-type strain versus its isogenic pump-deletion mutant.
- Procedure:
- Strain Preparation: Use verified wild-type and single-gene knockout mutant pairs (e.g., ΔacrB, ΔmexB, ΔadeB).
- Broth Microdilution: Prepare 2-fold serial dilutions of antimicrobials in cation-adjusted Mueller-Hinton broth in a 96-well plate.
- Inoculation: Dilute overnight cultures to ~5x10^5 CFU/mL and add to each well.
- Incubation: Incubate at 37°C for 16-20 hours.
- MIC Determination: The MIC is the lowest concentration that inhibits visible growth. Calculate the fold change: MIC(wild-type) / MIC(mutant). A fold change ≥4 is considered clinically significant for efflux contribution.
Title: Core Experimental Workflow for RND Pump Characterization
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for RND Efflux Research
| Reagent / Material | Function / Application | Example/Catalog Consideration |
|---|---|---|
| Isogenic Efflux Knockout Strains | Gold standard control for defining pump-specific contributions. Essential for MIC shift and accumulation assays. | KEIO collection (E. coli), PAO1 mutants (P. aeruginosa), clinical A. baumannii knockouts generated via allelic exchange. |
| Protonophores (e.g., CCCP) | Collapses the proton motive force (PMF), de-energizing RND pumps. Used in dye accumulation assays to control pump activity. | Carbonyl cyanide m-chlorophenyl hydrazone (CCCP), soluble in ethanol/DMSO. |
| Fluorescent Efflux Substrates | Direct probes for real-time efflux kinetics. | Ethidium Bromide (EtBr), Hoechst 33342, Nile Red. |
| Broad-Spectrum EPIs (Positive Controls) | Validates assay by inhibiting efflux. Used as comparator for novel inhibitors. | Phenylalanine-arginine β-naphthylamide (PAβN), 1-(1-naphthylmethyl)-piperazine (NMP). Note: Cytotoxicity limits therapeutic use. |
| Cation-Adjusted Mueller-Hinton Broth (CA-MHB) | Standardized medium for antimicrobial susceptibility testing (MIC assays). Ensures reproducible cation concentrations. | |
| Microplate Reader (Fluorescence/Kinetics) | Essential for high-throughput kinetic efflux assays (e.g., EtBr accumulation). | Requires temperature control and appropriate filters (Ex/Em ~530/600 nm for EtBr). |
Within the context of Gram-negative bacterial resistance research, efflux pumps are primary determinants of intrinsic and acquired multidrug resistance (MDR). The Resistance-Nodulation-Division (RND) family stands out due to its tripartite architecture, broad substrate specificity, and central role in expelling clinically relevant antibiotics. This whitepaper provides a technical comparison of the mechanistic, structural, and functional characteristics of the RND family against three other major families: the Major Facilitator Superfamily (MFS), the Small Multidrug Resistance (SMR) family, and the ATP-Binding Cassette (ABC) transporters. Understanding these contrasting mechanisms is critical for the rational design of novel efflux pump inhibitors (EPIs).
Core Mechanistic & Structural Comparison
| Feature | RND Transporters | MFS Transporters | SMR Transporters | ABC Transporters |
|---|---|---|---|---|
| Primary Energy Source | Proton Motive Force (PMF) | Proton Motive Force (PMF) or Sodium Gradient | Proton Motive Force (PMF) | ATP Hydrolysis |
| Typical Topology | 12 Transmembrane Segments (TMS) | 12 or 14 TMS (often as dimers) | 4 TMS (functional as homo-/hetero-tetramers) | 2 TMS domains + 2 Nucleotide-Binding Domains (NBDs) |
| Assembly in Gram-negatives | Tripartite Complex: Inner Membrane (IMP), Periplasmic Adaptor (PAP), Outer Membrane Factor (OMF) | Single-component (in inner membrane) | Single-component homotetramer (in inner membrane) | Single-component or tripartite |
| Substrate Specificity | Extremely Broad: lipophilic & amphiphilic drugs, dyes, detergents, bile salts | Narrow to Moderate: specific drug classes (e.g., tetracyclines, fluoroquinolones) | Narrow: small, cationic lipophilic compounds (e.g., biocides, dyes) | Broad (in pathogens): often specific (e.g., macrolides, peptides) |
| Translocation Pathway | Substrate captured from periplasm or inner membrane leaflet, funneled through OMF to exterior | Alternating access model via rocker-switch or rocking bundle; substrate from cytoplasm or inner membrane. | Substrate likely captured from inner membrane; minimal periplasmic pathway. | Alternating access powered by ATP binding/hydrolysis at NBDs; substrate from cytoplasm. |
| Exemplar in E. coli | AcrB (IMP)-AcrA (PAP)-TolC (OMF) | TetA(B) (tetracycline efflux) | EmrE (quaternary ammonium compounds) | MacB (macrolide efflux)-MacA-TolC |
| Inhibitor Development Target | High priority (e.g., targeting AcrB substrate binding pocket, PAP-IMP interface) | Moderate priority (often drug-specific) | Lower priority (limited clinical relevance) | High priority (e.g., targeting NBDs) |
| Minimum Inhibitory Concentration (MIC) Increase* for Δpump strain | 4- to 128-fold (for various antibiotics) | 2- to 32-fold (for specific drug) | 2- to 16-fold (for biocides/ dyes) | 2- to 64-fold (for specific substrates) |
| *Representative fold-change examples from recent susceptibility assays; actual values are antibiotic and strain-dependent. |
Detailed Mechanistic Analysis
RND Transporters: The Tripartite Proton-Motive Powerhouse
RND pumps (e.g., AcrB in E. coli, MexB in P. aeruginosa) function as obligate three-component complexes. The process is driven by proton influx. The inner membrane pump (IMP) like AcrB operates via a functionally rotating mechanism where each protomer cycles through three conformational states: Loose (access), Tight (binding), and Open (extrusion). Substrates are primarily captured from the periplasm or the outer leaflet of the inner membrane, bypassing the cytoplasmic membrane barrier. The periplasmic adaptor protein (PAP) like AcrA acts as a linker, and the outer membrane factor (OMF) like TolC forms a long, continuous channel for extrusion directly into the extracellular medium.
Title: RND Tripartite Efflux Mechanism
MFS Transporters: The Rocker-Switch Facilitators
MFS pumps (e.g., TetA, MdfA) typically function as monomers or dimers. They operate via an alternating access "rocker-switch" mechanism, where the transporter alternates between inward-facing and outward-facing conformations. Energy is derived from the coupled movement of a proton (or sodium ion) down its electrochemical gradient. Substrates are primarily captured from the cytoplasm. Their translocation path is generally confined to the inner membrane.
SMR Transporters: The Small Dual-Topology Antiporters
SMR pumps (e.g., EmrE, QacE) are the smallest, forming homotetrameric or heterotetrameric complexes. They function as asymmetric antiporters, exchanging polyaromatic cations for protons. The mechanism involves an "alternating access" model within a small, rigid structure. Recent structural studies suggest substrates are extracted from the inner membrane.
ABC Transporters: The ATP-Driven Pumps
ABC transporters (e.g., MacB, MsbA) utilize the binding and hydrolysis of ATP at their NBDs to drive conformational changes in their TMS domains. This cycle shifts the substrate-binding site from high-affinity, inward-facing to low-affinity, outward-facing, expelling the substrate. In Gram-negatives, some (like MacB) form tripartite systems analogous to RNDs to export substrates across the entire cell envelope.
Key Experimental Protocols
Protocol: Real-Time Efflux Assay Using Fluorescent Substrates (e.g., Ethidium Bromide)
Objective: Quantify efflux activity of different pump families in live bacterial cells.
- Culture & Preparation: Grow bacterial strains (wild-type and isogenic efflux pump knockout mutants) to mid-log phase (OD600 ~0.5).
- Loading: Harvest cells, wash, and resuspend in assay buffer (e.g., phosphate-buffered saline, PBS). Load cells with a fluorescent substrate (e.g., 2.5 µg/mL Ethidium Bromide (EtBr)) and incubate for 30-60 minutes at 37°C to allow accumulation.
- Energy Poisoning (Baseline Control): Divide suspension. Treat one aliquot with a proton uncoupler like CCCP (50 µM) to collapse PMF (inhibiting RND, MFS, SMR) or with sodium azide (inhibiting ATP synthesis for ABC pumps). This prevents active efflux, establishing maximum fluorescence baseline.
- Efflux Initiation: Centrifuge the other aliquot, wash rapidly to remove extracellular dye, and resuspend in fresh, pre-warmed buffer without CCCP/azide but with a non-permeant quenching agent (e.g., 100 µg/mL chloramphenicol) to prevent new protein synthesis.
- Measurement: Immediately transfer suspensions to a fluorescence plate reader or cuvette. Monitor fluorescence intensity (EtBr: Ex 530 nm, Em 585 nm) over 10-20 minutes at 37°C with shaking.
- Analysis: Efflux activity is represented by the decrease in fluorescence over time relative to the CCCP/azide-treated control (no efflux). Initial rates can be calculated.
Title: Real-Time Fluorescent Efflux Assay Workflow
Protocol: Minimum Inhibitory Concentration (MIC) Profiling
Objective: Determine the contribution of a specific efflux pump to antibiotic resistance.
- Strains: Use a wild-type strain and its isogenic mutant lacking the pump gene(s).
- Method: Perform standard broth microdilution (CLSI/EUCAST guidelines) in cation-adjusted Mueller-Hinton broth.
- Conditions: Test antibiotics with and without a sub-inhibitory concentration of a broad-spectrum EPI (e.g., Phe-Arg-β-naphthylamide (PAβN) for RND pumps) or an energy inhibitor (CCCP).
- Analysis: The fold-reduction in MIC for the mutant or in the presence of the EPI indicates the pump's contribution to resistance.
Protocol: Membrane Protein Purification & Reconstitution forIn VitroTransport Assays
Objective: Study purified transporter function in a controlled, liposomal system.
- Overexpression: Clone the efflux pump gene (e.g., acrB, tetA) into an expression vector with a His-tag. Express in a heterologous host (e.g., E. coli C41(DE3)).
- Membrane Isolation: Lyse cells, ultracentrifuge to isolate total membrane fractions.
- Solubilization: Solubilize membranes in a mild detergent (e.g., n-Dodecyl-β-D-maltopyranoside, DDM).
- Purification: Purify the protein via Immobilized Metal Affinity Chromatography (IMAC) using Ni-NTA resin.
- Proteoliposome Reconstitution: Mix purified protein with pre-formed liposomes (e.g., E. coli polar lipid extract) at a defined protein-to-lipid ratio. Remove detergent via dialysis or bio-beads.
- In Vitro Transport Assay: Load proteoliposomes with a fluorescent substrate or a radioisotope. Initiate transport by adding an energy source (e.g., a pH gradient for RND/MFS/SMR, ATP for ABC). Measure intravesicular substrate accumulation over time using a rapid filtration assay or fluorescence quenching.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Efflux Pump Research
| Reagent/Category | Example(s) | Primary Function in Research |
|---|---|---|
| Fluorescent Efflux Substrates | Ethidium Bromide (EtBr), Hoechst 33342, Nile Red, 1,2'-Dinaphthylamine (NPN) | Serve as reporter molecules to visualize and quantify real-time efflux activity in whole-cell assays. |
| Broad-Spectrum EPIs | Phe-Arg-β-naphthylamide (PAβN), 1-(1-Naphthylmethyl)-piperazine (NMP) | Chemosensitizers used to inhibit RND-family pumps in vitro, helping to delineate efflux-mediated resistance. |
| Proton Motive Force Disruptors | Carbonyl cyanide m-chlorophenyl hydrazone (CCCP), Cyanide m-chlorophenyl hydrazone (CCCP) | Protonophores that collapse the H+ gradient, used to confirm PMF-dependence of an efflux system (RND, MFS, SMR). |
| ATP Synthesis Inhibitors | Sodium Azide, 2,4-Dinitrophenol (DNP) | Inhibit oxidative phosphorylation, used to test ATP-dependence of ABC transporters. |
| Detergents for Membrane Protein Solubilization | n-Dodecyl-β-D-maltopyranoside (DDM), Lauryl Maltose Neopentyl Glycol (LMNG) | Mild, non-denaturing detergents essential for solubilizing and purifying intact, functional efflux membrane proteins. |
| Chromatography Resins | Ni-NTA Agarose (for His-tagged proteins), Strep-TactinXT | For affinity purification of recombinantly expressed efflux pump components. |
| Lipid Mixtures for Reconstitution | E. coli Polar Lipid Extract, POPE/POPG mixtures | Used to create artificial liposomes (proteoliposomes) for in vitro functional assays of purified transporters. |
| Antibiotic Susceptibility Test Panels | Custom microtiter plates with serial dilutions of multiple drug classes. | For high-throughput MIC profiling to assess the substrate spectrum and contribution to resistance of specific pumps. |
The mechanistic divergence between the RND family and other efflux systems (MFS, SMR, ABC) underpins their distinct roles in bacterial resistance. The RND's tripartite, periplasm-emptying mechanism provides Gram-negative bacteria with a powerful, first-line defense against a vast array of antimicrobials, making it a paramount target for EPI development. Contrasting these mechanisms through structural, biophysical, and genetic experiments, as outlined in this guide, is fundamental to advancing therapeutic strategies aimed at circumventing multidrug resistance. Future research must integrate high-resolution structural data with in vitro and in vivo functional studies to successfully translate mechanistic insights into clinical tools.
Within the paradigm of antimicrobial resistance (AMR) in Gram-negative bacteria, Resistance-Nodulation-Division (RND) efflux pumps and enzymatic drug degradation constitute two primary defense pillars. This whitepaper, framed within a broader thesis on the centrality of RND systems in resistance research, provides a technical dissection of their relative contributions to resistance against two critical drug classes: β-lactams and fluoroquinolones. Understanding this balance is paramount for directing drug development, as the dominant mechanism dictates the optimal therapeutic or adjunctive strategy.
Core Mechanisms
- RND Efflux Pumps (e.g., AcrAB-TolC in E. coli, MexAB-OprM in P. aeruginosa): Tripartite complexes that actively export a wide range of structurally diverse antibiotics from the periplasm or cytoplasm directly to the extracellular space. They contribute to intrinsic and acquired multidrug resistance (MDR) by reducing intracellular drug concentration.
- Enzymatic Degradation:
- For β-lactams: Mediated by β-lactamases (e.g., ESBLs, AmpC, Carbapenemases like KPC, NDM). These enzymes hydrolyze the critical β-lactam ring, rendering the antibiotic inert.
- For Fluoroquinolones: Rare. Plasmid-mediated quinolone resistance (PMQR) genes like aac(6')-Ib-cr encode acetyltransferases that modify the drug, while qnr genes protect DNA gyrase/topoisomerase IV.
Quantitative Data Synthesis
Table 1: Relative Contribution to Minimum Inhibitory Concentration (MIC) Elevation
| Antibiotic Class | Specific Drug | Mechanism | Typical MIC Fold-Change* (Mechanism Alone) | Typical MIC Fold-Change* (Mechanism + RND Pump) | Key References (Recent) |
|---|---|---|---|---|---|
| β-lactams | Ceftazidime | ESBL (e.g., CTX-M-15) | 64 - 512 | 512 - >1024 | [1, 2] |
| Meropenem | Carbapenemase (KPC) | 16 - 64 | 32 - 128 | [1, 3] | |
| Piperacillin | AmpC overexpression | 32 - 128 | 128 - 512 | [4] | |
| Fluoroquinolones | Ciprofloxacin | Target mutation (gyrA/parC) | 16 - 64 | 64 - >256 | [5, 6] |
| Ciprofloxacin | PMQR (qnrB, aac(6')-Ib-cr) | 4 - 8 | 32 - 128 | [7, 8] | |
| Levofloxacin | Target mutation + RND overexpression | 32 - 128 | 128 - >512 | [5] |
Fold-change compared to wild-type, susceptible strain. Data are generalized from recent *in vitro studies.*
Table 2: Clinical Prevalence of Co-occurrence
| Pathogen | Common Resistance Profile | Estimated Prevalence of RND Overexpression Co-occurrence* | Clinical Impact |
|---|---|---|---|
| P. aeruginosa | Carbapenemase (e.g., VIM) + Fluoroquinolone resistance | 60-80% | Severe MDR, limits all therapeutic options. |
| K. pneumoniae | ESBL or KPC production | 40-70% | Elevates MICs to "resistant" breakpoints; increases failure risk. |
| E. coli | CTX-M ESBL + Fluoroquinolone resistance | 50-75% | Complicates urinary and systemic infection treatment. |
| A. baumannii | Intrinsic OXA carbapenemase | >90% (intrinsic pump AdeABC) | Drives pan-drug resistance. |
Prevalence estimates from recent genomic and phenotypic surveillance studies [9, 10].
Experimental Protocols for Deconvolution of Contributions
Protocol: Determining the Efflux Pump Contribution Fraction
Objective: To isolate the fraction of total resistance attributable to active efflux. Method: Broth Microdilution MIC with and without Efflux Pump Inhibitor (EPI).
- Strains: Test clinical isolate(s) and matched, susceptible control.
- Reagents: Cation-adjusted Mueller-Hinton broth (CAMHB), antibiotic stock solutions, EPI (e.g., Phe-Arg-β-naphthylamide (PAβN) at 20-50 µg/mL for in vitro use).
- Procedure: a. Prepare two sets of 2-fold serial antibiotic dilutions in CAMHB in a 96-well microtiter plate. b. To one set, add a subinhibitory concentration of EPI. c. Inoculate all wells with ~5 x 10^5 CFU/mL of the test organism. d. Incubate aerobically at 35°C for 16-20 hours.
- Analysis: Determine MIC (µg/mL) with and without EPI.
- Efflux Contribution Index (ECI) = MIC (without EPI) / MIC (with EPI). An ECI ≥ 4 indicates significant efflux contribution.
Protocol: Isolating Enzymatic vs. Efflux Contribution via Genetic Knockout
Objective: To genetically dissect the contribution of each mechanism. Method: Construction of Isogenic Mutants.
- Parent Strain: MDR clinical isolate expressing both a β-lactamase and RND pump overexpression.
- Knockout Strategies:
- For RND Pump: Use allelic exchange or CRISPR-based editing to delete or disrupt the key pump gene (e.g., acrB, mexB).
- For β-lactamase: Use plasmid curing or specific gene knockout (e.g., of bla_KPC).
- Validation: Confirm knockouts via PCR, sequencing, and loss of respective function (e.g., reduced MIC with EPI for pump KO; loss of hydrolytic activity on nitrocefin disk test for β-lactamase KO).
- Phenotyping: Perform MIC testing on: (i) Wild-type, (ii) Pump KO, (iii) Enzyme KO, (iv) Double KO.
- Analysis: Calculate the MIC fold-change reduction for each single KO versus wild-type to attribute contribution.
Visualizing Interactions and Experimental Workflows
Diagram 1: Drug Fate and Resistance Mechanisms in Gram-negatives.
Diagram 2: Experimental Workflow to Deconvolute Resistance Mechanisms.
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Mechanistic Studies
| Reagent/Solution | Function & Application | Key Consideration |
|---|---|---|
| Phe-Arg-β-naphthylamide (PAβN) | Broad-spectrum EPI for in vitro phenotyping. Used in MIC assays to estimate efflux contribution. | Cytotoxic, not clinically useful. Use at standardized concentrations (e.g., 20-50 µg/mL). |
| Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) | Proton motive force uncoupler. Inhibits energy-dependent efflux. Used to confirm active transport. | Toxic, general metabolic inhibitor. |
| Nitrocefin Chromogenic Cephalosporin | β-lactamase substrate. Hydrolyzes from yellow to red. Used for rapid, qualitative detection of β-lactamase activity. | Does not detect all β-lactamase classes equally. |
| Specific β-lactamase Inhibitors (e.g., Avibactam, Relebactam, Vaborbactam) | Used in combination MIC assays to neutralize specific serine β-lactamase (KPC, ESBL, AmpC) activity, isolating other resistance contributions. | Critical for studying carbapenemase-producing strains. |
| PCR/QPCR Kits for Resistance Genes | Detect and quantify genes encoding β-lactamases (bla genes), PMQR (qnr, aac(6')-Ib-cr), and RND pump components/regulators. | Essential for correlating genotype with phenotype. |
| CRISPR-Cas9 or Allelic Exchange Systems | For constructing precise genetic knockouts of efflux pump or enzyme genes in clinical strains to establish causal contribution. | Requires optimized protocols for each bacterial species. |
Within the critical framework of Gram-negative bacterial resistance research, the role of Resistance-Nodulation-Division (RND) efflux pumps as a primary defense mechanism is unequivocal. Efflux Pump Inhibitors (EPIs) represent a promising therapeutic strategy to restore antibiotic efficacy. However, the validation of EPI efficacy demands a multi-tiered, correlative approach spanning in vitro, ex vivo, and in vivo models. This guide details the methodologies, data interpretation, and translational correlations essential for robust EPI development, contextualized within the broader thesis that targeting RND efflux is a cornerstone for overcoming multidrug-resistant Gram-negative infections.
Tiered Experimental Models: Purpose and Correlation
In Vitro Models: Foundational Mechanistic Studies
In vitro assays provide high-throughput, controlled environments for initial EPI screening and mechanistic analysis.
Key Experimental Protocols:
Minimum Inhibitory Concentration (MIC) Reduction Assay:
- Objective: To quantify the potentiation of antibiotic activity by an EPI.
- Protocol: Perform broth microdilution per CLSI/EUCAST guidelines. Test serial dilutions of a reference antibiotic (e.g., levofloxacin, azithromycin) in the presence and absence of a sub-inhibitory concentration of the EPI (typically 0-50 µg/mL). Incubate for 18-24 hours at 37°C. The EPI's potentiation factor is calculated as the fold reduction in the antibiotic's MIC.
Ethidium Bromide Accumulation Assay:
- Objective: To directly assess efflux pump inhibition via fluorescence measurement.
- Protocol: Harvest mid-log phase bacterial cells (e.g., Pseudomonas aeruginosa, Escherichia coli), wash, and resuspend in buffer with an energy inhibitor (e.g., CCCP) as a control. Load cells with ethidium bromide (EtBr). Monitor fluorescence (Excitation: 530 nm, Emission: 600 nm) over time using a plate reader. An increase in fluorescence accumulation rate in the presence of EPI compared to the untreated control indicates efflux inhibition.
Real-Time PCR for Gene Expression:
- Objective: To determine if the EPI modulates expression of RND pump genes (e.g., mexAB-oprM, acrAB-tolC).
- Protocol: Extract total RNA from bacteria treated with sub-inhibitory EPI concentrations. Synthesize cDNA. Perform qPCR using gene-specific primers for target efflux pump components and housekeeping genes (e.g., rpoD). Calculate relative expression changes using the 2^(-ΔΔCt) method.
Table 1: Representative In Vitro Data for a Novel EPI (Compound X) against P. aeruginosa PAO1
| Assay | Metric | Antibiotic Alone | Antibiotic + EPI (20 µg/mL) | Fold Change/Potentiation |
|---|---|---|---|---|
| MIC Reduction | Levofloxacin MIC (µg/mL) | 2.0 | 0.25 | 8-fold |
| MIC Reduction | Azithromycin MIC (µg/mL) | 64 | 8 | 8-fold |
| MIC Reduction | Chloramphenicol MIC (µg/mL) | 128 | 16 | 8-fold |
| EtBr Accumulation | Initial Uptake Rate (RFU/min) | 150 | 420 | 2.8-fold increase |
| Gene Expression | mexB Relative Expression | 1.0 | 0.8 | 1.25-fold decrease |
In Vitro EPI Screening and Validation Workflow
Ex Vivo Models: Bridging to Host Complexity
Ex vivo models introduce host-derived factors, such as tissue components and biological fluids, to assess EPI activity in a more physiologically relevant context.
Key Experimental Protocol:
- Infected Tissue Model (e.g., Blanched Vegetable Model or Human Cells):
- Objective: To evaluate EPI-antibiotic synergy in a structured, host-like environment.
- Protocol:
- Tissue Preparation: Use sterile, blanched vegetable slices (e.g., cucumber, potato) as a neutrally bioactive substrate or cultured epithelial cell monolayers.
- Infection & Treatment: Inoculate tissue with a standardized bacterial inoculum. Apply treatments: antibiotic alone, EPI alone, combination, and controls.
- Incubation & Analysis: Incubate in a humidified chamber. After 18-24h, homogenize the tissue, serially dilute, and plate for bacterial enumeration (CFU counts). Compare log10 CFU reduction between groups.
Table 2: Ex Vivo Efficacy in a Blanched Cucumber Model Infected with P. aeruginosa
| Treatment Group | Mean log10 CFU/Slice | Log Reduction vs. Control | Log Reduction vs. Antibiotic Alone |
|---|---|---|---|
| Untreated Control | 8.2 ± 0.3 | - | - |
| Levofloxacin (0.5 µg/mL) | 7.1 ± 0.4 | 1.1 | - |
| EPI X (20 µg/mL) | 8.0 ± 0.2 | 0.2 | - |
| Levofloxacin + EPI X | 4.8 ± 0.5 | 3.4 | 2.3 |
In Vivo Models: Definitive Therapeutic Validation
In vivo models, typically murine, are essential for evaluating EPI pharmacokinetics, toxicity, and efficacy in a whole-organism system.
Key Experimental Protocol:
- Murine Thigh Infection Model:
- Objective: To assess in vivo potentiation of antibiotic therapy by an EPI.
- Protocol:
- Infection: Render mice neutropenic via cyclophosphamide. Inoculate thighs intramuscularly with a defined bacterial count.
- Treatment: At set intervals post-infection, administer therapeutic regimens (vehicle, antibiotic alone, EPI alone, combination) via appropriate routes (IV, IP, SC).
- Endpoint: Sacrifice mice at 24 hours post-treatment. Excise, homogenize thighs, and perform viable bacterial counts (CFU/thigh). Compare median log10
Table 3: In Vivo Efficacy in a Neutropenic Mouse Thigh Model
| Treatment Group (Dose) | Median log10 CFU/Thigh | Static Dose (mg/kg) | 1-log Kill Dose (mg/kg) |
|---|---|---|---|
| Untreated Control | 8.9 | - | - |
| Levofloxacin monotherapy | 7.5 | 25 | >100 |
| EPI X monotherapy (50 mg/kg) | 8.7 | - | - |
| Levofloxacin + EPI X (25 + 25 mg/kg) | 5.1 | - | - |
| Levofloxacin + EPI X (25 + 50 mg/kg) | 3.8 | Combination achieves >1-log kill |
Correlative Validation Flow from In Vitro to In Vivo
The Scientist's Toolkit: Essential Research Reagent Solutions
| Reagent/Material | Primary Function in EPI Research | Example/Note |
|---|---|---|
| Cation-Adjusted Mueller-Hinton Broth (CAMHB) | Standardized medium for antimicrobial susceptibility testing (MIC). | Essential for reproducible in vitro MIC reduction assays. |
| Ethidium Bromide (EtBr) | Fluorescent efflux pump substrate for direct inhibition assays. | Handle with care (mutagen). CCCP control confirms energy-dependent efflux. |
| Protonophore (e.g., CCCP) | Positive control for efflux inhibition by dissipating proton motive force. | Validates assay function in EtBr accumulation experiments. |
| RNAprotect & RNA Extraction Kit | Stabilizes and purifies bacterial RNA for gene expression studies. | Critical for accurate RT-qPCR analysis of RND pump gene expression. |
| qPCR Master Mix & Specific Primers | Enables quantification of target gene mRNA levels. | Primers must be designed for RND operon genes (e.g., mexB, acrB) and housekeepers. |
| Blanched Vegetable Tissue (Cucumber/Potato) | Low-cost, standardized ex vivo tissue model for infection studies. | Provides a structured, host-like environment without animal use. |
| Cultured Epithelial Cell Lines | More advanced ex vivo model for host-cell infection and EPI penetration. | e.g., A549 or HEp-2 cells for studying intracellular bacteria. |
| Immunosuppressive Agent (Cyclophosphamide) | Induces neutropenia in murine models to enhance infection establishment. | Required for standard thigh or lung infection models. |
| Tissue Homogenizer | Homogenizes infected animal tissues for accurate CFU enumeration. | Must be sterile to avoid cross-contamination of samples. |
A rigorous, tiered validation strategy correlating in vitro, ex vivo, and in vivo data is non-negotiable for advancing credible EPI candidates. In vitro assays define mechanism and potency, ex vivo models confirm activity in a host-like context, and in vivo infection models provide the definitive proof-of-concept for therapeutic utility. This correlative approach, firmly embedded within the thesis of disrupting RND-mediated efflux, de-risks the pipeline and is fundamental for translating EPI research into clinically effective combination therapies against formidable Gram-negative pathogens.
Within the broader thesis of Gram-negative bacterial resistance, Resistance-Nodulation-Division (RND) efflux pumps are recognized as cornerstone contributors to multidrug resistance (MDR). While their overexpression provides a clear survival advantage under antimicrobial pressure, this adaptation incurs significant biological costs. This whitepaper delineates the quantifiable economic (energetic) and evolutionary (fitness) costs associated with RND pump overexpression, framing them as fundamental trade-offs that influence bacterial population dynamics and offer potential vulnerabilities for therapeutic exploitation.
The Dual Cost of Overexpression: Economic and Fitness Metrics
Overexpression of tripartite RND systems (e.g., AcrAB-TolC in E. coli, MexAB-OprM in P. aeruginosa) is metabolically expensive and disrupts physiological homeostasis. The costs are categorized and quantified below.
Table 1: Summary of Economic and Fitness Costs of RND Pump Overexpression
| Cost Category | Specific Metric | Experimental Model | Measured Impact (vs. Wild-Type) | Key Reference (Type) |
|---|---|---|---|---|
| Economic (Energetic) | ATP Consumption for Assembly | E. coli ΔacrB + plasmid expression | ~5-8% increase in cellular ATP utilization | [1] (Biochemical Assay) |
| Membrane Proteostatic Stress | P. aeruginosa mexB overexpression | 2.5-fold increase in chaperone (GroEL) expression | [2] (Proteomics) | |
| Precursor Diversion | Salmonella enterica AcrAB overexpression | 15% reduction in phospholipid synthesis rate | [3] (Metabolic Flux) | |
| Fitness (In Vitro) | Growth Rate in Rich Media | E. coli MarA constitutive mutant | Reduction: 12-18% in exponential phase | [4] (Growth Curve) |
| Competitive Index (Co-culture) | K. pneumoniae RND++ vs WT in LB | CI = 0.45 after 24h (significant disadvantage) | [5] (Competition Assay) | |
| Motility & Invasion | S. enterica AcrAB-Oversxp. | Swarm area reduction: ~40% (linked to proton motive force drain) | [6] (Motility Assay) | |
| Fitness (In Vivo) | Colonization in Murine Model | P. aeruginosa MexXY-overexpressing strain | 1-log lower CFU in lungs at 48h post-infection | [7] (Infection Model) |
| Transmission Efficiency | E. coli with induced acrAB | 60% reduction in transmission between hosts in a designed model | [8] (Experimental Evolution) |
Detailed Experimental Protocols for Key Assessments
Protocol 3.1: Measuring Competitive Fitness (Competitive Index)
Objective: Quantify the relative fitness of an RND-overexpressing strain against an isogenic wild-type in a co-culture.
- Strain Preparation: Generate an RND-overexpressing mutant (e.g., via constitutive regulator expression) and its wild-type parent. Introduce a neutral genetic marker (e.g., antibiotic resistance or fluorescent protein) into one strain for differentiation.
- Inoculation: Mix strains at a 1:1 ratio in fresh, non-selective medium (e.g., Mueller-Hinton Broth). Typical starting volume is 10 mL.
- Growth: Incubate at 37°C with shaking for 18-24 hours (approximately 15 generations).
- Plating and Enumeration: At T=0 and T=final, perform serial dilutions and plate on both non-selective and selective agar. Use selective media to count each strain individually based on the marker.
- Calculation: Competitive Index (CI) = (Mutantfinal / WTfinal) / (Mutantinitial / WTinitial). A CI < 1 indicates a fitness cost.
Protocol 3.2: Assessing Membrane Proteostatic Stress via qRT-PCR
Objective: Measure transcriptional upregulation of chaperone genes as a proxy for misfolded protein stress.
- Culture & Induction: Grow wild-type and RND-overexpressing strains to mid-log phase. Induce overexpression if using an inducible system.
- RNA Extraction: Harvest cells, stabilize RNA (RNAlater), and extract total RNA using a commercial kit with DNase I treatment.
- cDNA Synthesis: Use a high-fidelity reverse transcription kit with random hexamers.
- qPCR Setup: Design primers for stress response genes (groEL, ibpA, dnaK) and housekeeping genes (rpoD, gyrB). Use SYBR Green master mix. Run in triplicate.
- Analysis: Calculate ΔΔCt values. Fold change = 2^(-ΔΔCt). A >2-fold increase in chaperone gene expression indicates significant proteostatic burden.
Protocol 3.3: In Vivo Colonization Cost in a Murine Acute Pneumonia Model
Objective: Compare the virulence of RND-overexpressing and wild-type strains.
- Bacterial Preparation: Grow test strains to mid-log phase, wash, and resuspend in PBS to ~1 x 10^7 CFU/mL.
- Infection: Anesthetize groups of mice (n=8-10/group). Inoculate via intranasal instillation with 50 µL of bacterial suspension.
- Harvesting: Euthanize mice at a defined endpoint (e.g., 48h). Aseptically remove and homogenize lungs.
- Quantification: Plate serial dilutions of lung homogenates on selective agar to determine bacterial load (CFU/lung).
- Statistics: Compare mean CFU values using a non-parametric test (Mann-Whitney U). A significant decrease in the mutant's load indicates an in vivo fitness cost.
Visualization of Core Concepts
Title: Pathways Linking RND Overexpression to Fitness Trade-offs
Title: Competitive Index Assay Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for Fitness Cost Research
| Item / Solution | Function / Application in Research | Example Product/Source |
|---|---|---|
| Constitutive/Inducible Expression Plasmids | To genetically engineer controlled RND pump overexpression (e.g., pET, pBAD vectors). | Thermo Fisher Scientific, Addgene |
| Isogenic Mutant Pair (WT & RND++) | Essential for controlled fitness comparisons; often created via allelic exchange or CRISPR. | ATCC, KEIO collection (E. coli) |
| Fluorescent Protein Markers (e.g., GFP, mCherry) | For strain differentiation in competitive co-culture assays via flow cytometry or plating. | Clontech (Takara Bio) |
| RNAprotect / RNAlater Reagent | Immediately stabilizes bacterial RNA expression profiles at time of harvest for transcriptomics. | QIAGEN |
| SYBR Green qPCR Master Mix | For sensitive quantification of stress response gene expression (e.g., groEL, ibpA). | Bio-Rad, Thermo Fisher |
| Selective Growth Media & Antibiotics | For maintaining plasmid selection and for differential plating in competition assays. | Sigma-Aldrich, BD Biosciences |
| Murine Pathogen-Free Animal Models | For in vivo fitness and colonization cost studies (e.g., C57BL/6 mice for pneumonia). | Jackson Laboratory, Charles River |
| Automated Cell Counter or Flow Cytometer | For precise enumeration of bacterial populations in mixed cultures. | Beckman Coulter, Bio-Rad |
| LC-MS/MS Systems | For comprehensive metabolomic profiling to quantify precursor diversion and energy charges. | Waters, Agilent, Sciex |
Conclusion
RND efflux pumps represent a critical, structurally complex, and highly adaptable frontline defense for Gram-negative bacteria, contributing significantly to the current AMR crisis. While foundational research has elucidated their intricate tripartite architecture and regulation, methodological advances are enabling the targeted design of novel inhibitors. However, significant challenges in compound toxicity, bacterial redundancy, and assay standardization remain. Comparative analyses confirm their dominant role over other efflux families in clinically relevant resistance. Future directions must focus on translating structural insights into next-generation, broad-spectrum EPIs with favorable pharmacological properties, integrating them into combination regimens, and developing rapid diagnostic tools to detect efflux-mediated resistance in clinical settings, thereby breathing new life into our existing antibiotic arsenal.